Key Points
Pharmacologic TP in hospitalized adolescents with SCD increased from 1.3% of admissions in 2010 to 14.4% in 2021.
Prophylactic direct oral anticoagulants are increasingly used in adolescents with SCD.
Abstract
Patients with sickle cell disease (SCD) are predisposed to a hypercoagulable state. Despite the increased risk of venous thromboembolism in the SCD population, there is limited evidence available to guide thromboprophylaxis (TP) practices in these patients. This study aimed to assess the use of pharmacologic and nonpharmacologic TP in adolescent patients with SCD using the Pediatric Health Information System (PHIS). We hypothesized that TP was increasingly used in hospitalized adolescent patients with SCD. The study included patients with SCD aged 13 to 21 years, admitted to a PHIS hospital between 1 January, 2010, and 30 June, 2021. A total of 7202 unique patients consisting of 34 094 unique admissions were included for analyses. Pharmacologic or mechanical TP was used in 2600 (7.6%) admissions, with 3.6% of admissions (n = 1225) receiving pharmacologic prophylaxis and 4.3% (n = 1474) receiving mechanical prophylaxis. Pharmacologic TP increased in use from 1.3% of admissions in 2010 to 14.4% in the first half of 2021. Enoxaparin was the most commonly prescribed anticoagulant, used in 87% of admissions in which pharmacologic TP was used. The use of prophylactic direct oral anticoagulants was first documented in 2018 and increased to 25% of admissions with pharmacologic TP by 2021. This study demonstrates a steady increase in TP use in adolescent patients with SCD admitted to the hospital. Prospective cohort studies are needed to determine VTE risk factors in adolescents and children with SCD and the efficacy and safety of prophylactic regimens.
Introduction
Sickle cell disease (SCD) is associated with a chronic prothrombotic state that increases the risk of venous thromboembolism (VTE). Adults with SCD have a 4- to 100-fold increased risk for VTE and a 2 to fourfold increased risk of VTE-related death as compared with the general population.1-3 A large cohort study that examined 1.8 million SCD admissions from 1979 to 2003 found that the prevalence of pulmonary embolism in hospitalized patients (<40 years of age) with SCD was ∼3.5 times higher than in African American controls.4 Notably, the mean age at development of both pulmonary embolism and deep vein thrombosis was less in hospitalized adults with SCD (28 and 31 years, respectively) than African American patients comprising the control group (57 and 54 years, respectively).4
The mechanisms of hypercoagulability in SCD are diverse, including enhanced platelet function,5-7 activation of the coagulation cascade,8-10 and impaired fibrinolysis.9 In addition, the risk of VTE in patients with SCD is influenced by traditional risk factors outside the disease itself, such as hospitalization and the presence of a central venous catheter (CVC).11,12 In children and adolescents with SCD, case reports, case series, and combined adult and adolescent cohort studies suggest an increased prevalence of VTE in children who undergo CVC placement.11-14
Despite the increased risk of VTE in the SCD population, there is limited evidence available to guide thromboprophylaxis (TP) practices in these patients. In 2018, the American Society of Hematology published guidelines recommending that pharmacologic VTE prophylaxis be provided for acutely or critically ill hospitalized adults with acceptable bleeding risk, and use of mechanical prophylaxis as an alternative when bleeding risk is unacceptable.15 SCD was not explicitly mentioned in these guidelines. Nonetheless, hematologists acknowledge the risk of VTE in patients with SCD and have been using TP despite the lack of standardized guidelines.16,17
This retrospective cohort study aims to assess the use of pharmacologic and nonpharmacologic TP in adolescent patients with SCD who are hospitalized at freestanding children's hospitals in the United States using a large administrative pediatric hospital database. Given the increasing awareness of VTE as preventable harm during hospitalization18 and the growing use of direct oral anticoagulants (DOACs) in adolescents allowing for a noninjectable option,19 we hypothesized that the use of prophylaxis would increase over time.
Methods
Data source
The data source for this multicenter cohort study was the Pediatric Health Information Systems (PHIS). Managed by the Children's Hospital Association, PHIS is an administrative database that contains clinical and resource utilization data for inpatient, ambulatory surgery, emergency department, and observation unit patient encounters for 50 of the largest freestanding children’s hospitals in the United States. All data are deidentified and assigned a unique identifier which allows patients to be tracked across admissions over time. The Children's Hospital Association and the participating institutions routinely check for data quality to ensure reliability and validity.
Study population and definitions
The study included patients with SCD, aged 13 to 21 years, admitted to a PHIS hospital between 1 January 2010 and 30 June 2021. International Classification of Diseases, ninth (ICD-9-CM) and 10th revision (ICD-10-CM) diagnosis codes were used to identify SCD genotype (SS, SC, and Sβ thal) (Table 1). For patients with diagnoses of multiple genotypes throughout the study period, the most frequently used code was used to determine SCD genotype. In the instance of a tie for the most frequent genotype code, a nonspecific SCD was assigned for the patient's genotype.
Adolescents with SCD admitted to US children’s hospitals, PHIS, 2010-2021
| Characteristic | |
| Unique patients | 7202 |
| Unique admissions | 34 094 |
| Age at first admission median (range) | 15.6 y (13.0-21.9) |
| Male sex, n (%) | 3549 (49.3) |
| Race, n (%) | |
| Black | 6507 (90.3) |
| White | 184 (2.6) |
| Other/Unknown | 511 (7.1) |
| Ethnicity, n (%) | |
| Non-Hispanic/Latino | 6418 (89.1) |
| Hispanic/Latino | 328 (4.6) |
| Unknown | 456 (6.3) |
| Insurance type, n (%) | |
| Public | 4829 (67.1) |
| Private | 2061 (28.6) |
| Other (self-pay, charity) | 255 (3.5) |
| Unknown | 56 (0.8) |
| Region, n (%) | |
| Midwest | 1604 (22.3) |
| Northeast | 1138 (15.8) |
| South | 3723 (51.7) |
| West | 737 (10.2) |
| Sickle Cell Genotype∗, n (%) | |
| SS | 4951 (68.7) |
| SC | 1068 (14.8) |
| Sβ thalassemia | 663 (9.2) |
| Nonspecific/other† | 520 (7.2) |
| Characteristic | |
| Unique patients | 7202 |
| Unique admissions | 34 094 |
| Age at first admission median (range) | 15.6 y (13.0-21.9) |
| Male sex, n (%) | 3549 (49.3) |
| Race, n (%) | |
| Black | 6507 (90.3) |
| White | 184 (2.6) |
| Other/Unknown | 511 (7.1) |
| Ethnicity, n (%) | |
| Non-Hispanic/Latino | 6418 (89.1) |
| Hispanic/Latino | 328 (4.6) |
| Unknown | 456 (6.3) |
| Insurance type, n (%) | |
| Public | 4829 (67.1) |
| Private | 2061 (28.6) |
| Other (self-pay, charity) | 255 (3.5) |
| Unknown | 56 (0.8) |
| Region, n (%) | |
| Midwest | 1604 (22.3) |
| Northeast | 1138 (15.8) |
| South | 3723 (51.7) |
| West | 737 (10.2) |
| Sickle Cell Genotype∗, n (%) | |
| SS | 4951 (68.7) |
| SC | 1068 (14.8) |
| Sβ thalassemia | 663 (9.2) |
| Nonspecific/other† | 520 (7.2) |
This table excludes 427 patients who were documented as having VTE.
Determined by ICD coding. For patients who had multiple genotypes coded throughout the study period, the genotype that was used most frequently was chosen.
Includes nonspecific and not elsewhere classifiable SCD diagnosis codes as well as instances of a tie for the most frequently used genotype and the method described in the previous footnote could not be applied.
Anticoagulant medications (enoxaparin, fondaparinux, apixaban, rivaroxaban, and dabigatran) were identified by pharmacy billing codes. Unfractionated heparin as a TP was not investigated because we could not delineate whether heparin was used for TP or as a line flush. In an effort to focus on anticoagulant use for prophylaxis, patients with a VTE diagnosis at any time during the study period were excluded. In addition, admissions with a length of stay of ≤2 days were excluded because a full assessment of VTE risk may not occur until 24 hours after hospital admission or may not be considered in patients known to be admitted solely for overnight observation. Supply billing codes were used to determine CVC placement and the use of mechanical prophylaxis. Mechanical prophylaxis included billing codes for compression stockings, sleeves, and related compression devices. We also extracted information on the subspecialty of attending and consulting physicians during admission to determine whether a hematologist was involved in the patient’s care. Hospitals give information on the treating/consulting physicians which includes the subspecialties the physician is board certified in; therefore, we used physician identifiers rather than just billing codes to determine hematology involvement.
Statistical analysis
All data were summarized using standard descriptive statistics, frequency and percentage for categorical variables, and median and range for quantitative variables. The proportion of prophylaxis use between groups or over time was completed using chi-square tests. P values are 2-sided, and those <.05 were considered statistically significant. All analyses were completed using SAS software, version 9.4 (SAS Institute, Cary, NC).
Results
From January 2010 to June 2021, a total of 7629 unique adolescent patients with SCD were admitted to 48 PHIS hospitals. A VTE diagnosis was documented in 427 patients (5.6%) who were subsequently excluded from analyses, leaving 7202 unique patients (49.3% male) with 34 094 unique admissions included for analyses. Patients were primarily Black (90.3%) and non-Hispanic (89.1%) (Table 1). The median age at first admission was 15.6 years (13.0-21.9 years). The SCD genotypes included HbSS (68.7%), HbSC (14.8%), HbSβ thalassemia (9.2%), and nonspecific/other sickle cell (7.2%) (Table 1).
Pharmacologic or mechanical TP was used in 2600 (7.6%) admissions (Table 2), with 3.6% of admissions (n = 1225) receiving pharmacologic prophylaxis and 4.3% (n = 1474) receiving mechanical prophylaxis. A small percentage (n = 99, 0.3%) of admissions were documented as receiving both pharmacologic and mechanical prophylaxis. Enoxaparin was the most commonly used anticoagulant, prescribed in 87.0% of admissions in which pharmacologic anticoagulation was used. Overall, apixaban and rivaroxaban were prescribed in 13.7% and 0.7% of admissions using pharmacologic prophylaxis, respectively. Patients receiving apixaban or rivaroxaban ranged from 13 to 21 years of age but were predominately in the 16 to 21 age range (92%).
Frequency of pharmacologic or mechanical thromboprophylaxis use by admission per age group
| . | Age 13-14 . | Age 15-17 . | Age 18-21 . |
|---|---|---|---|
| N = 7256 . | N = 14172 . | N = 12666 . | |
| Prophylaxis (pharmacological or mechanical) | 362 (5.0) | 865 (6.1) | 1373 (10.8) |
| Pharmacologic prophylaxis | 119 (1.6) | 333 (2.3) | 773 (6.1) |
| Enoxaparin | 113 (95.0) | 282 (84.7) | 670 (86.7) |
| Apixaban | 4 (3.4) | 55 (16.5) | 109 (14.1) |
| Rivaroxaban | 1 (0.8) | 3 (0.9) | 4 (0.5) |
| Fondaparinux | 0 (−) | 1 (0.3) | 0 (−) |
| Dabigatran | 1 (0.8) | 0 (−) | 0 (−) |
| Mechanical prophylaxis | 253 (3.5) | 569 (4.0) | 652 (5.1) |
| Compression sleeves for sequential device | 224 (88.5) | 495 (87.0) | 589 (90.3) |
| Sequential compression device | 29 (11.5) | 70 (12.3) | 58 (8.9) |
| Compression stockings | 5 (2.0) | 27 (4.7) | 29 (4.4) |
| . | Age 13-14 . | Age 15-17 . | Age 18-21 . |
|---|---|---|---|
| N = 7256 . | N = 14172 . | N = 12666 . | |
| Prophylaxis (pharmacological or mechanical) | 362 (5.0) | 865 (6.1) | 1373 (10.8) |
| Pharmacologic prophylaxis | 119 (1.6) | 333 (2.3) | 773 (6.1) |
| Enoxaparin | 113 (95.0) | 282 (84.7) | 670 (86.7) |
| Apixaban | 4 (3.4) | 55 (16.5) | 109 (14.1) |
| Rivaroxaban | 1 (0.8) | 3 (0.9) | 4 (0.5) |
| Fondaparinux | 0 (−) | 1 (0.3) | 0 (−) |
| Dabigatran | 1 (0.8) | 0 (−) | 0 (−) |
| Mechanical prophylaxis | 253 (3.5) | 569 (4.0) | 652 (5.1) |
| Compression sleeves for sequential device | 224 (88.5) | 495 (87.0) | 589 (90.3) |
| Sequential compression device | 29 (11.5) | 70 (12.3) | 58 (8.9) |
| Compression stockings | 5 (2.0) | 27 (4.7) | 29 (4.4) |
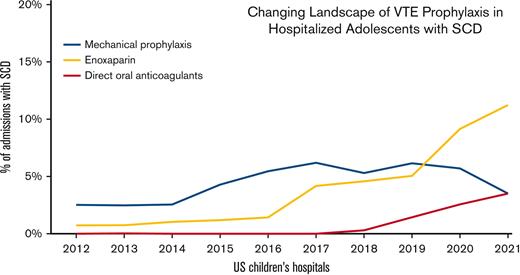
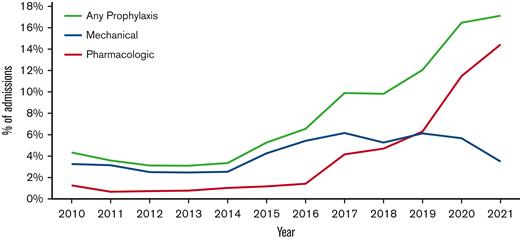
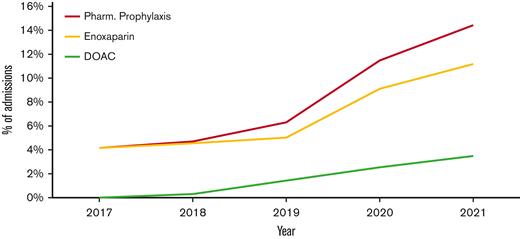
The use of TP was noted to change over the study period, with significant increases observed in both pharmacologic and mechanical prophylaxis (P<.0001; Figure 1). Pharmacologic TP increased from 1.3% of admissions in 2010 to 4.2% in 2017 and 14.4% in the first half of 2021. The large increase in TP from 2017 to mid-2021 was primarily caused by the increased use of enoxaparin rather than the increased use of DOACs. (Figure 2) Enoxaparin remained the most commonly used anticoagulant throughout the study period. However, the use of prophylactic DOACs was first documented in 2018 and by 2021 it increased to 25% of all admissions in which pharmacologic TP was used. Mechanical prophylaxis was used in 3.3% of admissions in 2010, increasing to 6.2% by 2017; however, we did not observe any notable increase from 2017 to 2021, with only 3.5% of admissions using mechanical prophylaxis in the first half of 2021.
Thromboprophylaxis use per year in admissions for adolescent patients with SCD at US children's hospitals. Line graph showing the use of mechanical and pharmacologic thromboprophylaxis per year in adolescent patients with SCD admitted in US children’s hospitals.
Thromboprophylaxis use per year in admissions for adolescent patients with SCD at US children's hospitals. Line graph showing the use of mechanical and pharmacologic thromboprophylaxis per year in adolescent patients with SCD admitted in US children’s hospitals.
Pharmacologic thromboprophylaxis use from 2017-2021. Line graph showing the use of pharmacologic prophylaxis with either enoxaparin or a DOAC from 2017 to mid-2021.
Pharmacologic thromboprophylaxis use from 2017-2021. Line graph showing the use of pharmacologic prophylaxis with either enoxaparin or a DOAC from 2017 to mid-2021.
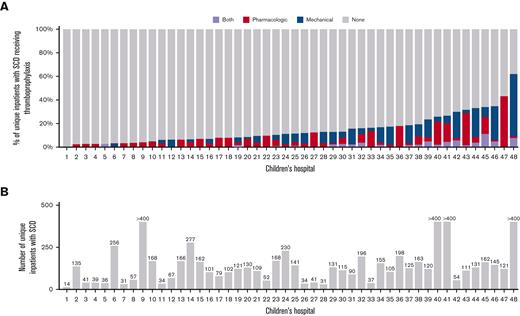
Prophylaxis was more likely to be used in admissions that included time in the intensive care unit (20.4% vs 6.8%, P<.001), operating room procedures (18.4% vs 6.8%, P<.001), and CVC placement (13.6% vs 7.3%, P<.001). TP was more likely to be used in patients aged 18 to 21 years (10.8% of admissions, P<.001) compared with patients aged 15 to 17 years and 13 to 14 years (6.1% and 5.0% of admissions, respectively). Most admissions (n = 29 775, 87.3%) had a hematology provider involved in their medical care (admitting, attending or consulting physician). Admissions without hematology involvement were more likely to have pharmacologic (4.6% vs 3.4%, P<.001) or mechanical (5.4% vs 4.2%, P<.001) prophylaxis prescribed compared with admissions in the hematology service. However, DOACS were more likely to be prescribed in admissions that involved a hematology/oncology provider (0.1% vs 0.6%, P<.0001.) The type of TP used and the percentage of unique patients receiving TP varied by participating PHIS institutions, ranging from 0% to 62% of patients receiving TP during their hospitalization (Figure 3). In our cohort, the percentage of patients with the ICD-10 code for coronavirus disease 2019 (COVID-19) in 2020 and 2021 was 2.75% and 3.73%, respectively.
Frequency and type of thromboprophylaxis use in adolescents with SCD at 48 US children’s hospitals (PHIS, 2010-2021). (A) Bar graph showing thromboprophylaxis practices of 48 children’s hospitals in hospitalized adolescent patients with SCD. (B) Bar graph showing patients included in the study by hospital. Hospitals with more than 400 patients were capped/censored at N = 400 in order to preserve anonymity.
Frequency and type of thromboprophylaxis use in adolescents with SCD at 48 US children’s hospitals (PHIS, 2010-2021). (A) Bar graph showing thromboprophylaxis practices of 48 children’s hospitals in hospitalized adolescent patients with SCD. (B) Bar graph showing patients included in the study by hospital. Hospitals with more than 400 patients were capped/censored at N = 400 in order to preserve anonymity.
Discussion
In this large multi-institutional study of US children’s hospitals, the use of TP in adolescents with SCD was documented in 7.6% of hospitalizations and increased over the study period. These findings imply that despite the lack of evidence-based data supporting the use of pharmacologic TP in children with SCD, pediatricians are likely to identify patients with SCD at an increased risk for thrombosis and to provide TP in a high-risk setting such as hospital admission. A PHIS study of hospitalized patients (2008-2015) aged 10 to 18 years with various medical issues demonstrated that 1% of patients received prophylactic anticoagulation and 5% received mechanical prophylaxis.20 Similarly, in 2 studies looking at pharmacologic TP in pediatric trauma patients aged 0 to 18 years, 1.1% and 6.2% of patients were given VTE prophylaxis, respectively.21,22 Our study did include more current data (up to 2021), and the use of TP has likely increased in other pediatric and adolescent populations as well given the increasing recognition of VTE as preventable harm in children’s hospitals.18
We found that enoxaparin was the most commonly used anticoagulant throughout the time frame of our study. The use of apixaban and rivaroxaban in this population has steadily increased since 2017, although seen primarily in patients >16 years of age. It is not surprising that DOACs were used mainly in older adolescents and young adults, given that for the majority of the study time frame, there were no Food and Drug Administration–approved DOACs for patients <18 years old. In addition, the 2018 American Society of Hematology guidelines recommend using low-molecular-weight heparin over DOACs for VTE prophylaxis in acutely ill medical inpatients because of the increased bleeding risk associated with DOAC use.15 To date, there have been 4 retrospective and 1 prospective study reporting the use of DOACs in adults with SCD and VTE23-27; there are none in children with SCD. In addition, dabigatran and rivaroxaban were both recently approved by the Food and Drug Administration for pediatric use in 2021,28,29 albeit for the treatment of VTE, not primary TP. As clinical experience and research studies in these areas continue to grow, we anticipate the use of DOACs will continue to increase in hospitalized patients with SCD.
It is also important to consider the unique characteristics of SCD that influence a practitioner’s decision to use TP. For example, first line treatment for vaso-occlusive crisis in SCD is pharmacologic therapy with nonsteroidal anti-inflammatory drugs (NSAIDs) and opioids.30 Often, patients with vaso-occlusive crisis are on a course of NSAIDs for days before admission. Thus, initiating TP in this setting may increase the risk of bleeding. The risk-benefit ratio of TP is further complicated when the patient has underlying intracranial pathology, such as moyamoya disease or a history of stroke. Future work should include evaluating the safety of using pharmacologic TP in patients with SCD who are on aspirin or NSAIDs, as well as those with moyamoya syndrome who are at an increased risk of intracranial hemorrhage.
Our work highlights the variability in VTE prophylaxis in pediatric hospitals. We found that among the 48 hospitals included in this study, the percentage of hospitalized patients with SCD who received TP ranged from 0% to as high as 62% (Figure 3). In addition, even in hospitals where TP was more frequently prescribed (Figure 3, Hospital ID 42-48), the type of prophylaxis varied notably between institutions. Despite the dissimilarity in TP practices across institutions, we found that the trend in the use of pharmacologic TP in hospitalized adolescent patients with SCD steadily increased from 2010 to 2021, with the use of mechanical prophylaxis leveling off and decreasing in the last 5 years (Figure 1). The continued increase of TP through 2020 and 2021 was likely influenced by the COVID-19 pandemic as the current guidance identifies TP during hospitalization to be of major importance for VTE prevention.31-33 In the admissions in our analyses from January 2020 to June 2021, ∼3% to 4% of unique admissions encounters included a diagnostic code for COVID-19.
There are inherent limitations of retrospective and administrative database studies that should be considered when interpreting our results. The data are dependent on the accurate coding of the diagnosis on admission and the reported pharmacy code. The use of ICD-9 and ICD-10 codes from hospital databases for determining specific SCD genotypes is problematic because there may be miscoding.34 Because we included patients with only 1 SCD-related admission in the study, it is plausible that a patient may be coded as SCD in that singular admission when they actually have traits. Our rationale for allowing this is that some patients with mild SCD may not be hospitalized as often and so including patients with 1 SCD-related admission would give us a better representation of the SCD population. There is also a lack of granular information regarding the patient's medical history at the time of admission or preexisting medical conditions. The patient may have had any number of underlying comorbidities that contributed to the use of mechanical or pharmacologic TP during their admission. Another limitation is the inability of the administrative database to differentiate between therapeutic and prophylactic dosing of anticoagulation. Our best estimate of TP use in this population is for patients who were placed on TP in the absence of a diagnosis code of VTE. Our approach, however, almost certainly underestimates the use of TP because it would not have included patients with a history of VTE who completed their initial anticoagulant therapy but were now receiving TP due to hospitalization. Finally, our methodology and data source do not allow us to evaluate the safety or efficacy of TP in this clinical setting.
Hospital-acquired VTE continues to be a common preventable harm in children’s hospitals. Children and adolescents with SCD are particularly at risk. This study demonstrates a steady increase in the use of TP in adolescent patients with SCD admitted to the hospital. However, TP overall was still only used in the minority of admissions. Prospective cohort studies are needed to determine VTE risk factors in adolescents and children with SCD and the efficacy and safety of prophylactic regimens.
Authorship
Contribution: J.D. designed the study, drafted the abstract, introduction, results, and discussion; J.S. helped design the study, queried and cleaned the data, composed methods and statistical analyses, and revised and edited abstract, introduction, results, and discussion; S.O.B. is a subject matter expert in thrombosis and assisted in designing the study, editing the abstract, introduction, methods, results, and discussion.
Conflict-of-interest disclosure: J.D. has served on advisory boards for Genentech, Octapharma, and Boehringer Ingelheim. S.H.O. serves on an advisory board and as study chair for a clinical trial of direct oral anticoagulants for Bristol Myers Squibb. J.S. declares no competing financial interests.
Correspondence: Jennifer Davila, 3415 Bainbridge Ave, Bronx, NY 10467; e-mail: jedavila@montefiore.org.
References
Author notes
Presented in abstract form at the 62nd American Society of Hematology Annual Meeting in December 2020.
Data for this project are not publicly available and were queried from the PHIS database of www.childrenshospital.org. Researchers from the participating PHIS institutions can request training to gain access to the database or collaborate with an analyst with access to the database.
The full-text version of this article contains a data supplement.