Key Points
Gata2 +9.5 enhancer regulates adult hematopoietic stem cell self-renewal.
Gata2 +9.5 enhancer regulates T-cell development at the lymphoid-primed MPP3 stage.
Abstract
Mammalian GATA2 gene encodes a dual zinc finger transcription factor, which is essential for hematopoietic stem cell (HSC) generation in the aorta, gonad, mesonephros (AGM) region, HSC self-renewal, and specification of progenitor cell fates. Previously, we demonstrated that Gata2 expression in AGM is controlled by its intronic +9.5 enhancer. Gata2 +9.5 deficiency removes the E-box motif and the GATA site and depletes fetal liver HSCs. However, whether this enhancer has an essential role in regulating adult hematopoiesis has not been established. Here, we evaluate Gata2 +9.5 enhancer function in adult hematopoiesis. +9.5+/− bone marrow cells displayed reduced T cell reconstitution in a competitive transplant assay. Donor-derived analysis demonstrated a previously unrecognized function of the +9.5 enhancer in T cell development at the lymphoid-primed multipotent progenitor stage. Moreover, +9.5+/− adult HSCs displayed increased apoptosis and reduced long-term self-renewal capability in comparison with wild-type (WT) HSCs. These phenotypes were more moderate than those of Gata2+/− HSCs. Consistent with the phenotypic characterization, Gata2 expression in +9.5+/− LSKs was moderately higher than that in Gata2+/− LSKs, but lower than that in WT LSKs. Our data suggest that +9.5 deficiency compromises, without completely abrogating, Gata2 expression in adult HSCs.
Introduction
Mammalian GATA2 gene encodes a dual zinc finger transcription factor, which is essential for hematopoietic stem cell (HSC) generation from hemogenic endothelium in the aorta, gonad, mesonephros (AGM) region and HSC self-renewal.1 -3 Previously, we demonstrated that Gata2 expression in AGM is controlled by part of its intronic +9.5 enhancer.2,4,5 Gata2 +9.5 deficiency removes the E-box motif and the GATA site and depletes fetal liver HSCs. +9.5−/− mouse embryos died at E13-14, revealing the important +9.5 enhancer function in embryonic HSC generation.2,6,7 Furthermore, a +9.5 Ets motif mediates hematopoietic regeneration in response to myeloablation.8 However, whether this enhancer has broad functions to regulate adult hematopoiesis has not been established. Here, we report that Gata2 +9.5 enhancer regulates adult HSC self-renewal and its haploinsufficiency impacts T cell development starting at the multipotent progenitor 3 (MPP3) stage.
Methods
Mice
Gata2 germline heterozygous deletion mice (Gata2+/−)3 and Gata2 +9.5 cis-element germline heterozygous deletion mice (+9.5+/−)6 were maintained in a pure C57BL/6J genetic background (>N10). CD45.1+ isogenic recipient mice were purchased from Jackson Laboratories. All animal experiments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals and approved by an Animal Care and Use Committee at University of Wisconsin-Madison. The program is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care.
Additional methods are described in supplemental Methods.
Results and discussion
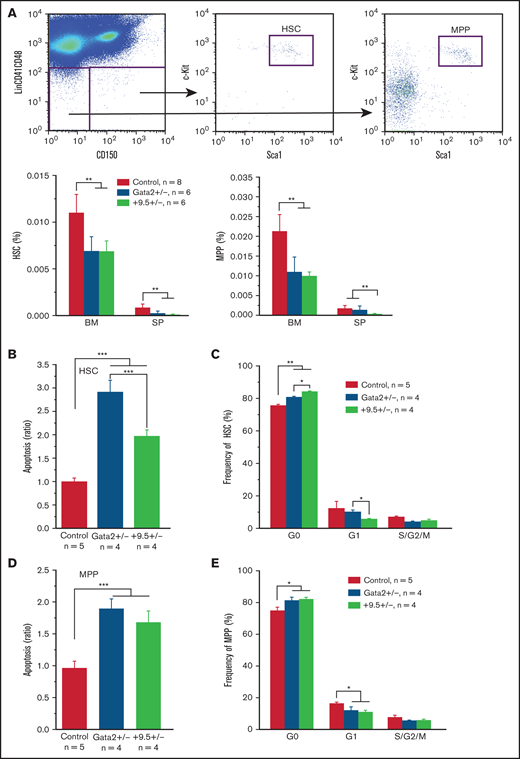
We analyzed adult hematopoietic stem and progenitor cells (HSPCs) in 6-week-old wild-type (WT) control, +9.5+/−, and Gata2+/− mice. Gata2+/− mice served as a positive control for reducing Gata2 expression in adult hematopoietic compartment.9,10 In contrast to normal numbers of fetal liver HSCs and MPPs in E12.5 +9.5+/− embryos,6 both HSC and MPP compartments were significantly decreased in +9.5+/− bone marrow (BM) and spleen (SP) compared with those in WT, but largely comparable to those in Gata2+/− mice (Figure 1A). The +9.5+/− HSC apoptosis was at a level intermediate to those of control and Gata2+/− HSCs (Figure 1B), whereas their quiescence was higher (Figure 1C). By contrast, both +9.5+/− and Gata2+/− MPPs displayed increased apoptosis (Figure 1D) and quiescence (Figure 1E) in comparison with WT MPPs. Quantification of Lin−Sca1+ cKit+ (LSK) cells revealed that similar to HSCs and MPPs, +9.5+/− and Gata2+/− LSK cells decreased in BM (supplemental Figure 1A), whereas the MP compartment was largely normal in +9.5+/− and Gata2+/− mice (supplemental Figure 1B). In addition, consistent with the prior report,10 Gata2+/− LSKs exhibited increased apoptosis (supplemental Figure 1C) and quiescence (supplemental Figure 1D). The +9.5+/− LSKs showed a similar apoptotic rate and elevated quiescence compared with Gata2+/− LSKs. These analyses revealed phenotypes in +9.5+/− adult HSCs and LSKs resembling those in Gata2+/− cells but with significant quantitative differences.10
Increased apoptosis in Gata2 +9.5+/− and Gata2+/− HSPCs. Six-week-old WT C57BL/6 (Control), Gata2 +9.5+/− (+9.5+/−), and Gata2+/− (Gata2+/−) mice were analyzed. (A) Gating strategy for HSCs and MPPs. HSCs and MPPs are defined based on LSK SLAM markers as described in Methods. Their frequencies were quantified in BM and SP. (B,D) Quantification of apoptotic BM HSCs (B) and MPPs (D) using Annexin V and 4′,6-diamidino-2-phenylindole (DAPI). (C,E) Cell cycle profiling of BM HSCs (C) and MPPs (E) using Ki67 and DAPI. The results are presented as mean ± standard deviation (SD). *P < .05; **P < .01; ***P < .001.
Increased apoptosis in Gata2 +9.5+/− and Gata2+/− HSPCs. Six-week-old WT C57BL/6 (Control), Gata2 +9.5+/− (+9.5+/−), and Gata2+/− (Gata2+/−) mice were analyzed. (A) Gating strategy for HSCs and MPPs. HSCs and MPPs are defined based on LSK SLAM markers as described in Methods. Their frequencies were quantified in BM and SP. (B,D) Quantification of apoptotic BM HSCs (B) and MPPs (D) using Annexin V and 4′,6-diamidino-2-phenylindole (DAPI). (C,E) Cell cycle profiling of BM HSCs (C) and MPPs (E) using Ki67 and DAPI. The results are presented as mean ± standard deviation (SD). *P < .05; **P < .01; ***P < .001.
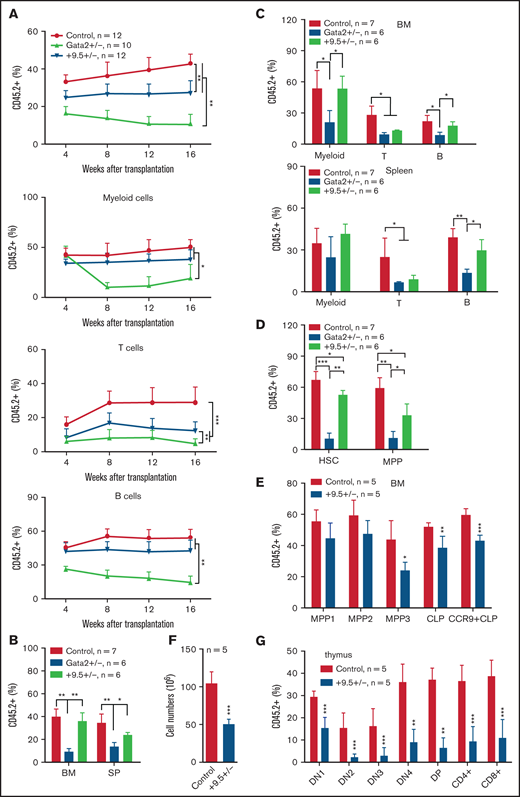
We performed a serial competitive reconstitution analysis of +9.5+/− HSC functions in vivo. WT, +9.5+/−, and Gata2+/− BM cells (CD45.2+) were mixed with competitor cells (CD45.1+) at a 1:1 ratio and transplanted into lethally irradiated recipients (CD45.1+). Consistent with what we found with +9.5+/− fetal liver cells, +9.5+/− cells in peripheral blood (PB) were significantly decreased (P < .01) in comparison with WT cells (Figure 2A). Interestingly, this reduced reconstitution mainly resulted from decreased donor-derived T cells, whereas donor-derived myeloid and B cells were comparable to those in recipients transplanted with WT cells. We killed recipients 16 weeks after transplant and analyzed donor-derived hematopoiesis. The +9.5+/−-derived cells were comparable in BM but lower in SP of primary recipients (Figure 2B). As with PB, +9.5+/−-derived T cells were reduced in BM and SP (Figure 2C). Moreover, we observed a moderate but significant reduction of +9.5+/−-derived HSCs and MPPs in comparison with WT recipients (Figure 2D). Consistent with reduced donor-derived HSCs and MPPs in primary recipients, +9.5+/−-derived cells displayed a decrease in multilineage reconstitution in secondary recipients (supplemental Figure 2A). Similar to the primary transplantation, +9.5+/−-derived HSCs and MPPs were moderately decreased in BM of secondary recipients (supplemental Figure 2B). Consistent with previous studies,9,10 Gata2+/− BM cells exhibited dramatically reduced multilineage reconstitution (P < .01) in both primary and secondary recipients (Figure 2; supplemental Figure 2).
Gata2 +9.5+/− BM cells display decreased reconstitution in primary recipients. A total of 1 × 106 CD45.2+ BM cells from 6-week-old WT C57BL/6J (Control), Gata2 +9.5+/− (+9.5+/−), and Gata2+/− (Gata2+/−) mice were mixed with 1 × 106 CD45.1+ competitor BM cells and injected into lethally irradiated recipients (CD45.1+). (A) Quantification of donor-derived myeloid, T, and B cells in recipient peripheral blood every 4 weeks after transplantation. (B-D) Terminal evaluation was performed in primary recipients 16 weeks after transplant. (B) Quantification of donor-derived cells in BM and SP of primary recipients. (C) Quantification of donor-derived myeloid, T, and B cells in BM and SP of primary recipients. (D) Quantification of donor-derived HSCs and MPPs in BM of primary recipients. The results are presented as means ± SD. *P < .05; **P < .01; ***P < .001. (E-G) Recipients were killed at 8 weeks after transplant. (E) Quantification of donor-derived MPP1, MPP2, and MPP3; CLPs; and CCR9+ CLPs in BM. (F) Quantification of thymic cell numbers. (G) Quantification of donor-derived T cells in thymus. The results are presented as means + SD. *P < .05; **P < .01; ***P < .001.
Gata2 +9.5+/− BM cells display decreased reconstitution in primary recipients. A total of 1 × 106 CD45.2+ BM cells from 6-week-old WT C57BL/6J (Control), Gata2 +9.5+/− (+9.5+/−), and Gata2+/− (Gata2+/−) mice were mixed with 1 × 106 CD45.1+ competitor BM cells and injected into lethally irradiated recipients (CD45.1+). (A) Quantification of donor-derived myeloid, T, and B cells in recipient peripheral blood every 4 weeks after transplantation. (B-D) Terminal evaluation was performed in primary recipients 16 weeks after transplant. (B) Quantification of donor-derived cells in BM and SP of primary recipients. (C) Quantification of donor-derived myeloid, T, and B cells in BM and SP of primary recipients. (D) Quantification of donor-derived HSCs and MPPs in BM of primary recipients. The results are presented as means ± SD. *P < .05; **P < .01; ***P < .001. (E-G) Recipients were killed at 8 weeks after transplant. (E) Quantification of donor-derived MPP1, MPP2, and MPP3; CLPs; and CCR9+ CLPs in BM. (F) Quantification of thymic cell numbers. (G) Quantification of donor-derived T cells in thymus. The results are presented as means + SD. *P < .05; **P < .01; ***P < .001.
Our results demonstrated a previously unrecognized +9.5 enhancer function in T-cell development. To identify the relevant T-cell developmental stage(s), we analyzed donor-derived linage-biased MPPs and the downstream common lymphoid progenitors (CLPs) in control and +9.5+/− recipients 8 weeks after transplant as previously described.11 MPP1 can generate all lineages of blood cells, MPP2 loses megakaryocyte/erythroid potential, whereas MPP3 is responsible for generating downstream CLPs and lymphocytes. We found that donor-derived MPP1 and MPP2 were comparable between control and +9.5+/− recipients (Figure 2E). By contrast, donor-derived MPP3 and CLP compartments were reduced in +9.5+/− recipients compared with control (Figure 2E). In particular, donor-derived CCR9+ CLPs, which migrate to the thymus and initiate thymic T-cell development, were also reduced in +9.5+/− recipients (Figure 2E). Not surprisingly, thymic T-cell cellularity in +9.5+/− recipients was decreased to ∼50% of that in control (Figure 2F). We analyzed thymic T-cell development from the Thy1.2+CD4−CD8− double-negative (DN) cells to CD4+CD8+ double-positive cells and to CD4+ and CD8+ T cells. DN thymocytes can be further divided into 4 successive subpopulations depending on the status of CD44 and CD25 expression: CD44+CD25− (DN1), CD44+CD25+ (DN2), CD44−CD25+ (DN3), and CD44−CD25− (DN4). Donor-derived thymic T cells were reduced at all stages in +9.5+/− recipients compared with control (Figure 2G). These results indicate that +9.5 regulates T-cell development at the MPP3 stage.
In addition, our data demonstrated that +9.5+/− HSCs and MPPs displayed more moderate phenotypes in self-renewal compared with Gata2+/− cells, suggesting that in addition to the +9.5 partial enhancer we characterized, there is additional element(s) regulating Gata2 expression in these cells. In support of our conclusion, heterozygous mutations in the Ets motif of +9.5 enhancer reduce GATA2 expression and cause GATA2 deficiency syndromes in human.5,6 These mutations impair hematopoietic regeneration in adult mice after myeloablation.8 To determine the impact of +9.5 enhancer deletion on Gata2 expression, we quantified Gata2 mRNA levels in WT, +9.5+/−, and Gata2+/− LSK and MP cells using quantitative reverse transcriptase-polymerase chain reaction. Corroborating the prior study,10 Gata2 expression was decreased in Gata2+/− LSKs and MPs. As expected, Gata2 expression in +9.5+/− MP populations was comparable to that in Gata2+/− MPs, whereas Gata2 expression in +9.5+/− LSKs was moderately higher than that in Gata2+/− LSKs but still lower than that of WT LSKs (supplemental Figure 3). Thus, the reduced self-renewal in +9.5+/− HSPCs is associated with Gata2 downregulation.
In summary, we evaluated Gata2 +9.5 enhancer function in adult hematopoiesis and demonstrated a +9.5 enhancer function to regulate adult HSC self-renewal. The impact of +9.5+/− on HSCs is less than Gata2+/−, suggesting that +9.5 deficiency compromises, but does not completely abrogate, Gata2 expression in adult HSCs. In sharp contrast to the +9.5 enhancer, Gata2 −77 enhancer regulates adult HSC survival and the impact of −77+/− on HSCs is opposite to that of Gata2+/−.12 Moreover, Gata2 +9.5 enhancer regulates T-cell development at the lymphoid-primed MPP3 stage, and T-cell development has not been analyzed in −77 enhancer mutant mice.
Acknowledgments
The authors thank Stuart Orkin for generously providing Gata2+/− mice and the University of Wisconsin Carbone Comprehensive Cancer Center (UWCCC) for use of its Shared Services (Flow Cytometry Laboratory, Genome Editing and Animal Models Shared Resource, and Experimental Pathology Laboratory).
The Flow Cytometry Laboratory was supported by National Institutes of Health Shared Instrument Grants 1S10RR025483-01 (BD FACS AriaII BSL-2 Cell Sorter) and 1S100OD018202-01 (BD LSR Fortessa). This work was supported by ASH Scholar and LLS Special Fellow awards to AAS, National Institute of Health grants CA152108 to J.Z. and DK68634 to E.H.B, Edward Evans MDS Foundation to E.H.B., and National Institutes of Health/National Cancer Institute P30 CA014520-UW-Comprehensive Cancer Center (UWCCC) Support.
Authorship
Contribution: X.Y. provided experimental design and execution and wrote the manuscript; Y.Z., Y.-I.C., and G.K. executed the experiments; E.A.R. provided histopathology analysis and wrote the manuscript; Y.Z., K.D.J., and A.A.S. provided technical or material support; and E.H.B. and J.Z. provided experimental design and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Jing Zhang, Room 7453, WIMR II, McArdle Laboratory for Cancer Research, 1111 Highland Ave, University of Wisconsin–Madison, Madison, WI 53705; e-mail: zhang@oncology.wisc.edu.
References
Author notes
For data sharing, contact the corresponding author: zhang@oncology.wisc.edu.
The full-text version of this article contains a data supplement.