Abstract
The survival and proliferation of follicular lymphoma (FL) cells are strongly dependent on macrophages, because their presence is necessary for the propagation of FL cells in vitro. To this regard, as also shown for the majority of solid tumors, a high tissue content of tumor-associated macrophages (TAMs), particularly if showing a protumoral phenotype (also called M2), is strongly associated with a poor outcome among patients with FL treated with chemotherapy. The introduction of rituximab, an anti-CD20 antibody that can be used by TAMs to facilitate antibody-dependent cellular cytotoxicity and antibody-dependent cellular phagocytosis, has challenged this paradigm. In the rituximab era, clinical studies have yielded conflicting results in FL, showing variable outcomes based on the type of regimen used. This highlighted, for the first time, that the impact of TAMs on the prognosis of patients with FL may depend on the administered treatment, emphasizing the need to better understand how currently available therapies affect macrophage function in FL. We summarize the impact of approved and novel therapies for FL, including radiation therapy, chemotherapy, anti-CD20 monoclonal antibodies, lenalidomide, and targeted agents, on the biology of TAMs and describe their effects on macrophage phagocytosis, polarization, and function. Although novel agents targeting the CD47/SIRPα axis are being developed and show promising activity in FL, a deeper understanding of macrophage biology and their complex pathways will help to develop novel and safer therapeutic strategies for patients with this type of lymphoma.
Introduction
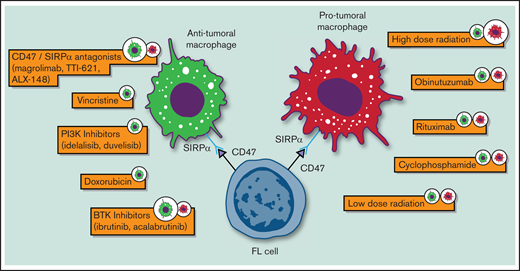
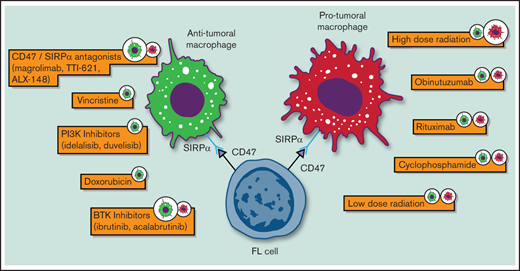
Follicular lymphoma (FL) is an indolent B-cell non-Hodgkin lymphoma (NHL) with a highly variable clinical course. Since the introduction of immunohistochemistry (IHC), pathologists have noticed the presence of significant nonmalignant cells, including macrophages, in its tumor milieu that are able to influence tumor growth and survival.1-3 To this regard, the in vitro propagation of FL primary cells, for even short-term survival, requires signals from nurse-like cells, highlighting its substantial degree of dependence on the tumor myeloid microenvironment.4,5 In addition, gene-expression profiling studies have shown that certain nonneoplastic cells (ie, macrophages) conferred poor prognosis in patients with FL who were treated with chemotherapy.6 As the therapeutic scenario of FL evolves, understanding macrophage biology and how current therapies affect their function becomes increasingly relevant for this lymphoma subtype (Table 1 and Figure 1).
Summary of biological impact of therapeutic agents on macrophages in FL
| Agent . | Molecular mechanism . | Biological impact . | References . |
|---|---|---|---|
| Low-dose radiation | Induction of TLR4, STAT1, ICGR1A, C1q, and APOE in macrophages | Increased macrophage phagocytosis | 26 |
| High-dose radiation | Induction of CD80, CD86, and HLA-DR in macrophages Downregulation of CD163, MRC1, VCAN, and IL-10 Preservation of MMP-2 and MMP-9 activity | Increased macrophage phagocytosis but decreased macrophage production of proinflammatory (antitumoral) cytokines | 25 |
| CTX | Induction of ATF4-mediated paracrine secretion of VEGF-A in lymphoma cells CD47 downregulation | Increased macrophage infiltration and phagocytosis | 28 |
| DOX | Increased tumor immunogenicity Macrophage sparing | Increased macrophage production of proinflammatory (antitumoral) cytokines | 32,33 |
| Vincristine | NLRP3 inflammasome induction Increased production of proinflammatory cytokines (IL-1β, IL-6, and CXCL1) | Increased macrophage production of proinflammatory (antitumoral) cytokines | 34 |
| Bendamustine | Unknown | Unknown | |
| Rituximab | Interaction with macrophage’s FcγRIV, FcγRIII, and FcγRI | Increased ADCC and ADCP | 45,46 |
| Obinutuzumab | Interactions with FcγRIIIa | Increased ADCC and ADCP | 48,51–53 |
| Lenalidomide | Unknown in FL | Unknown in FL | 58,95 |
| BTK inhibitors (ibrutinib) | Downregulation of CXCL12, CXCL13, CCL19, and VEGF Dephosphorylation of ERK1/2, AKT, and PLCγ2 Inhibition of JAK2 | Decreased interaction between protumoral macrophages (M2) and FL cells Increased ADCP | 63,66–68 |
| PI3K inhibitors (idelalisib and duvelisib) | Reduction of BAD phosphorylation via Ser136 Downregulation of BFL-1 Stimulation of NF-κB activation leading to C/EBPβ inactivation | Restored FL dependence on BCL-2 Increased macrophage polarization to an M1 phenotype | 76–79 |
| CD47/SIRPα-blocking agents | Decreased SIRPα signaling | Increased macrophage infiltration and phagocytosis | 87 |
| Increased macrophage production of proinflammatory (antitumoral) cytokines | |||
| Increased macrophage polarization to an M1 phenotype | |||
| Increased ADCC and ADCP |
| Agent . | Molecular mechanism . | Biological impact . | References . |
|---|---|---|---|
| Low-dose radiation | Induction of TLR4, STAT1, ICGR1A, C1q, and APOE in macrophages | Increased macrophage phagocytosis | 26 |
| High-dose radiation | Induction of CD80, CD86, and HLA-DR in macrophages Downregulation of CD163, MRC1, VCAN, and IL-10 Preservation of MMP-2 and MMP-9 activity | Increased macrophage phagocytosis but decreased macrophage production of proinflammatory (antitumoral) cytokines | 25 |
| CTX | Induction of ATF4-mediated paracrine secretion of VEGF-A in lymphoma cells CD47 downregulation | Increased macrophage infiltration and phagocytosis | 28 |
| DOX | Increased tumor immunogenicity Macrophage sparing | Increased macrophage production of proinflammatory (antitumoral) cytokines | 32,33 |
| Vincristine | NLRP3 inflammasome induction Increased production of proinflammatory cytokines (IL-1β, IL-6, and CXCL1) | Increased macrophage production of proinflammatory (antitumoral) cytokines | 34 |
| Bendamustine | Unknown | Unknown | |
| Rituximab | Interaction with macrophage’s FcγRIV, FcγRIII, and FcγRI | Increased ADCC and ADCP | 45,46 |
| Obinutuzumab | Interactions with FcγRIIIa | Increased ADCC and ADCP | 48,51–53 |
| Lenalidomide | Unknown in FL | Unknown in FL | 58,95 |
| BTK inhibitors (ibrutinib) | Downregulation of CXCL12, CXCL13, CCL19, and VEGF Dephosphorylation of ERK1/2, AKT, and PLCγ2 Inhibition of JAK2 | Decreased interaction between protumoral macrophages (M2) and FL cells Increased ADCP | 63,66–68 |
| PI3K inhibitors (idelalisib and duvelisib) | Reduction of BAD phosphorylation via Ser136 Downregulation of BFL-1 Stimulation of NF-κB activation leading to C/EBPβ inactivation | Restored FL dependence on BCL-2 Increased macrophage polarization to an M1 phenotype | 76–79 |
| CD47/SIRPα-blocking agents | Decreased SIRPα signaling | Increased macrophage infiltration and phagocytosis | 87 |
| Increased macrophage production of proinflammatory (antitumoral) cytokines | |||
| Increased macrophage polarization to an M1 phenotype | |||
| Increased ADCC and ADCP |
Biological impact of standard and novel therapeutic agents on macrophages in FL. Only agents for which preclinical data were available are depicted.
Biological impact of standard and novel therapeutic agents on macrophages in FL. Only agents for which preclinical data were available are depicted.
Impact of macrophages in patients with FL treated in the prerituximab era
Tumor-associated macrophages (TAMs) have been studied across various cancers, including breast, prostate, ovarian, and cervical carcinoma; across all tumor subtypes, increased numbers of TAMs have been associated with widespread metastasis and inferior survival in patients treated with traditional chemotherapy.7-9 A landmark analysis by Dave et al evaluated the impact of TAMs in 191 patients with previously untreated FL in the prerituximab era.6 Gene-expression profiling was performed on tumor biopsies obtained from patients with FL, and 2 gene expression signatures (immune response 1 and immune response 2) that were strongly associated with clinical outcome were identified. Although immune response 1 was enriched for genes expressed primarily in T cells (eg, CD7, CD8B1, ITK, LEF1, STAT4) and associated with longer survival (relative risk of death = 0.15), immune response 2 was enriched for genes expressed primarily in macrophages and follicular dendritic cells (eg, ACTN1 , TNFSF13B, TLR5, FCGR1A, SEPT10, LGMN, and C3AE1) and was associated with shorter survival (relative risk of death = 9.35), highlighting the negative prognostic impact of macrophages in patients with FL. Similar data were reported in a retrospective study by Farinha et al that analyzed the impact of TAMs in 99 patients with advanced-stage FL that was treated with frontline bleomycin, cisplatin, etoposide, doxorubicin (DOX), cyclophosphamide (CTX), vincristine, and prednisone, followed by involved site radiation, using IHC for CD68 on pretreatment tissue biopsies.10 In this analysis, a significantly shorter median overall survival was observed for patients with >15 TAMs per high-power field compared with those with a lower content (5 years vs 16 years). The association was maintained in a multivariate proportional hazard model including TAM content, International Prognostic Index score, and histologic grade, with a hazard ratio (HR) of 2.4.
The consistently poorer outcome observed in these patients raised the hypothesis that protumoral TAMs, also called M2, may be enriched in patients with FL.11 To this regard, Guilloton and colleagues showed that bone marrow mesenchymal stromal cells obtained from patients with FL expressed a specific gene signature and produced significantly higher levels of CCL2 compared with healthy donors when cocultured with FL cells.12 Through CCL2, mesenchymal stromal cells were able to recruit monocytes and drive their differentiation into a proangiogenic and lipopolysaccharide-unresponsive phenotype, as typically observed for M2.
Impact of macrophages in patients with FL treated in the rituximab era
The conflicting role of TAMs in patients with FL treated in the rituximab era was initially reported by Kridel and colleagues, using IHC for CD163 as a marker of M2 phenotype on pretreatment tissue biopsies.13 The study included 2 large FL cohorts: 186 patients treated with frontline R-CVP (rituximab, CTX, vincristine, and prednisone) and 395 patients treated with R-CHOP (rituximab, CTX, DOX, vincristine, and prednisone) who were subsequently randomized to rituximab maintenance or observation. Similar to what was reported in studies performed in the prerituximab era, an increased number of CD163+ cells was associated with poor outcomes in patients who were treated with R-CVP, with a 5-year progression-free survival (PFS) rate of 38% vs 72% (P = .004) for patients with a low number of CD163+ cells. Unexpectedly, however, an increased number of CD163+ cells was associated with a favorable outcome in patients treated with R-CHOP, with a 5-year PFS rate of 60% vs 44% (P = .011) in patients with a low number of CD163+ cells, independent of the subsequent use of rituximab maintenance. These findings highlighted, for the first time, that the impact of TAMs on the prognosis of patients with FL may not depend exclusively on their phenotype, but also on the administered treatment (specifically DOX and rituximab in this case), emphasizing the need to better understand how currently available therapies affect macrophage function in FL.
Impact of TAMs on FL transformation and POD24
Progression of disease within 24 months (POD24) from frontline chemoimmunotherapy and transformation to large B-cell lymphoma have been associated with worse outcome in FL.14,15 Blaker and colleagues compared tissue biopsies obtained from 52 patients with transformed FL with those from 40 patients with FL without signs of transformation.16 Using IHC for CD68 (as a general macrophage marker) and PD-L1, the investigators observed that a higher degree of intrafollicular (IF) infiltration of CD68+ and PD-L1+ macrophages was associated with a shorter time to transformation, with an HR of 2.1 for CD68 and an HR of 1.5 for PD-L1. It is important to note that, even if PD-L1+ macrophages suppress T-cell activation through the PD-1 receptor, suggesting an M2 phenotype, its expression is upregulated by interferon-γ (IFN-γ), a chemokine that typically induces an M1 phenotype instead.17,18 In addition, Stevens and colleagues from the Lunenburg Lymphoma Biomarker Consortium analyzed biological predictive markers of POD24 in 122 patients with FL who were treated with R-CHOP or R-CHOP–like regimens, followed by IFN-α maintenance for 2 years or observation, using IHC for CD163 on pretreatment tissue biopsies.19 An increased number of CD163+ macrophages was associated with a shorter PFS, as previously shown by Kridel et al,13 as well as an increased risk for POD24 (P = .038).
However, this paradigm was recently challenged by Tobin et al, using targeted gene sequencing and tissue microarray immunofluorescence to characterize the association between pretreatment tumor microenvironment (TME) features and POD24 in 132 patients with FL treated with frontline chemoimmunotherapy.20 In this study, gene-expression profiling revealed distinct clustering of FL samples based on high or low expression of immune infiltrating cells. The latter was associated with a higher risk for POD24, with an odds ratio of 4.3, despite the low expression of macrophage markers (eg, CD68) and M2 markers (eg, PD-L2), suggesting that a “cold microenvironment” may be more detrimental than a protumoral milieu.
Effects of radiation on TAMs in FL
Radiation therapy is the mainstay of treatment for early-stage FL, with observational studies suggesting that approximately half of these patients may be cured with a standard dose of 24 Gy.21 In addition, very-low-dose involved-field radiotherapy (4 Gy) has emerged as an effective and less toxic alternative, with a response rate of 92% and a median time to progression of 14 months.22,23 Radiation has been shown to produce DNA fragmentation of lymphoma cells, with subsequent release of damage-associated molecular patterns that are recognized by various immune cells, including macrophages. Upon stimulation by damage-associated molecular patterns, macrophages recruit T cells for antigen presentation and ultimately exert an antitumoral or protumoral activity, depending on the magnitude and durability of the T-cell response.24 To this regard, Pinto et al analyzed phenotypic changes occurring in primary human macrophages exposed to cumulative ionizing radiation doses of 2 Gy each, up to 10 Gy.25 Although macrophages exposed to high-dose radiation demonstrated high DNA damage, they remained viable and metabolically active and had enhanced phagocytosis, suggesting an antitumoral phenotype. Macrophages also showed downregulated or undetectable proinflammatory cytokines, such as interleukin-1β (IL-1β), tumor necrosis factor, IL-6, and CCR7, and were able to promote cancer cell invasion and cancer cell–induced angiogenesis. More recently, Knoops et al showed that a lower dose of radiation may induce a more consistent antitumoral phenotype in this immune cell subset.26 Gene-expression profiling was performed in 20 patients with FL who were treated with ultralow-dose radiation, and tissue core biopsies were collected before and 24 hours after treatment. Matched samples showed that genes associated with macrophage and/or dendritic cell activation, such as TLR4, STAT1, and ICGR1A, were highly induced, along with the genes necessary for lysosomal activity and macrophage-dependent apoptotic clearance, such as C1q and APOE. Of interest, IHC for CD68 did not show any increase in the absolute number of TAMs, suggesting that ultra-low dose radiation may be able to induce an antitumoral phenotype only in macrophages that are already present in the irradiated tissue, rather than favoring local recruitment.
Effects of chemotherapy on TAMs in FL
CTX
CTX is an alkylating agent that is commonly used for the treatment of patients with FL as part of regimens such as R-CVP and R-CHOP.27 Preclinical studies have demonstrated that “superphagocytic” macrophages could be the primary effectors mediating the activity of single-agent CTX in these patients. In fact, CTX has been shown to induce an endoplasmic reticulum stress response in preclinical lymphoma models, leading to upregulation of ATF4 and VEGF-A transcription, in addition to decreased CD47 expression. This, in turn, can increase macrophage recruitment with subsequent lymphoma clearance.28 VEGF-A plays a critical role in this mechanism, as demonstrated by the abrogation of macrophage phagocytosis through inhibition of SYK, an important mediator of the VEGF-A pathway. Interestingly, single-cell RNA analyses showed that 2 specific clusters of macrophages, designated as C2 and C4, were responsible for CTX-induced antibody-dependent cellular phagocytosis (ADCP). C2 and C4 macrophages were enriched for genes expressing Fc receptor–mediated phagocytosis and phagosome maturation. In addition, the C4 subtype, which expressed FCGR4 and CD36, accounted for 20% to 25% of bone marrow macrophages after CTX treatment and exhibited the highest level of phagocytosis, suggesting its role as the primary effector of single-agent CTX.
DOX
DOX is an anthracycline that is commonly used for the treatment of patients with FL as part of regimens, such as R-CHOP, and is typically reserved for patients with confirmed or suspected transformation.29-31 Preclinical studies show that the antitumoral activity of DOX is partially based on increased tumor immunogenicity. Mantovani and colleagues demonstrated that, in mouse xenografts, the efficacy of DOX was limited to those inoculated with immunogenic lymphoma cell lines. Subsequently, macrophages obtained from these models showed remarkable activity and nonspecifically inhibited growth and DNA synthesis of lymphoma cells in vitro.32 Additionally, antimacrophage agents, such as silica and carrageenan, abrogated the efficacy of DOX in immunogenic mouse models, whereas promacrophage agents, such as Cryptosporidium parvum, exhibited significant therapeutic synergism, suggesting that macrophages modulate the antitumor activity of DOX. Interestingly, although typically eliminating most lymphocyte subsets, DOX spares macrophages, further supporting the concept that TAMs are the main cellular subtype that is responsible for the immune-mediated antitumoral effects of DOX.33
Vincristine
Vincristine, a vinca alkaloid with antitubulin properties, is typically used for the treatment of FL in combination withCVP or CHOP. Wong et al showed that vincristine and DOX synergistically increase the expression of proinflammatory cytokines, such as IL-1β, IL-6, and CXCL1, by activation of the NLRP3 inflammasome in bone marrow–derived macrophages.34 Although these cytokines have been implicated in the cytotoxic side effects observed with traditional chemotherapy, including CHOP , they also exert antitumoral activity, emphasizing the role of macrophage-mediated cytotoxicity in patients with FL treated with vincristine.35
Bendamustine
Bendamustine is an alkylating agent that is commonly used as frontline therapy for patients with FL, typically in combination with a monoclonal anti-CD20 antibody.36 A retrospective study performed by Shimono et al showed that high monocyte content relative to lymphocyte content was associated with longer PFS in 87 patients with FL treated with frontline bendamustine + rituximab (BR).37 Although it could be speculated that circulating monocytes reflect the intratumoral content of tissue macrophages,38 the biological mechanisms responsible for the favorable interaction between this drug and macrophages remain largely unknown.
Effects of anti-CD20 monoclonal antibodies on TAMs in FL
Rituximab
The introduction of rituximab has significantly improved the outcome of patients with FL.39–41 One of the key features of this anti-CD20 monoclonal antibody is the lack of internalization after treatment, favoring its sustained interaction with effector immune cells, particularly macrophages.42 Although, as outlined above, the presence of TAMs was consistently associated with worse outcome in the prerituximab era, favorable associations have been reported in the rituximab era, particularly in combination with anthracycline-based regimens.10 In a retrospective study, Taskinen and colleagues analyzed the impact of TAMs, assessed via IHC for CD68.43 Pretreatment tissue biopsies were obtained from 96 patients with previously untreated or relapsed FL treated with R-CHOP; significantly longer median PFS (not reached vs 45 months, P = .006) and 5-year overall survival rates (97% vs 90%, P = .116) were observed in cases with high TAM content compared with those with low TAM content. The favorable impact of rituximab on TAMs was investigated further by Canioni and colleagues in a retrospective study in which the IF and extrafollicular content of TAMs was assessed by IHC for CD68.44 Among 194 previously untreated patients with high tumor burden FL who were randomized to receive CTX, DOX, etoposide, prednisolone, and IFN (CHVP-I) or rituximab + CHVP-I, high IF (risk ratio, 1.7; P = .01) and high extrafollicular (risk ratio, 1.6; P = .02) TAM content were significantly associated with inferior event-free survival in patients who received CHVP-I but not among those who were treated with rituximab + CHVP-I, suggesting that macrophages may mediate the antitumoral activity of rituximab.
To this regard, Minard-Colin et al analyzed the interaction between macrophages and anti-CD20 monoclonal antibodies in an FcRγ−/− mouse model and demonstrated that antibody-dependent cellular cytotoxicity (ADCC) mediated by FcγR-bearing macrophages was required for anti-CD20 monoclonal antibody–induced lymphoma cell depletion.45 The latter was abolished when clodronate-mediated depletion of macrophages was performed, resulting in significant lymphoma growth. Of interest, in vitro data show that M2 macrophages have a twofold to threefold increase in phagocytosis of rituximab-opsonized targets compared with M1 macrophages.46 This could be particularly relevant in FL, where IL-10 is typically overexpressed and has been shown to increase the content of M2 macrophages, as well as to also attenuate their phagocytic activity.47
Obinutuzumab
Obinutuzumab is a type II glycoengineered anti-CD20 monoclonal antibody that was shown to have lower complement-dependent cytotoxicity than rituximab, but greater ADCC and phagocytosis, with direct B-cell killing effects, resulting from the targeting of a different CD20 epitope.48,49 This has translated to a greater clinically efficacy in FL, with randomized clinical trials showing significantly prolonged PFS with the use of frontline obinutuzumab-based chemoimmunotherapy compared with rituximab-based chemoimmunotherapy.50
In vitro coculture studies show that macrophages exhibit superior ADCC and ADCP in the presence of obinutuzumab compared with other anti-CD20 monoclonal antibodies.51 Interestingly, similarly to what was shown for rituximab, obinutuzumab-opsonized lymphoma cells are better phagocytized by M2 macrophages than by M1 macrophages.
The favorable interaction between obinutuzumab and macrophages in FL can be further improved by its combination with agents that are able to stimulate macrophage antitumoral activity. In fact, in preclinical lymphoma models, the combination of obinutuzumab and a TLR7 agonist resulted in significant improvement in macrophage effector mechanisms and a 10-day increase in survival over negative controls.52 In addition, its combination with a bispecific antibody that is able to target CD20 and CD47 resulted in a significant increase in macrophage-mediated phagocytosis compared with rituximab in lymphoma cell lines and patient-derived xenografts.53 These data suggest that obinutuzumab may be a better option than rituximab for future combination strategies aimed at improving macrophage activity in patients with FL.
Effects of lenalidomide on TAMs in FL
Lenalidomide is an immunomodulatory agent, with pleomorphic activity on the TME, that is typically used in combination with an anti-CD20 monoclonal antibody for the treatment of previously untreated and relapsed patients with FL.54-56 Although the majority of translational studies in FL have focused on characterizing the effects of lenalidomide on T cells, its effects on TAMs are better described in chronic lymphocytic leukemia and multiple myeloma, with favorable effects on ADCC and ADCP through regulation of the vitamin D pathway, as well as on macrophage phenotype through activation of Rap1 GTPase.57-61 In preclinical lymphoma models, lenalidomide was shown to enhance macrophage-mediated ADCC of rituximab-opsonized lymphoma cells, with a 60% killing of Raji and Farage lymphoma cell lines compared with a 30% killing with rituximab alone.62 Studies aimed at determining the impact of lenalidomide on TAMs in FL and the mechanisms of resistance in this disease subtype are greatly needed.
Effects of targeted agents on TAMs in FL
BTK inhibitors
Although BTK inhibitors are not approved by the US Food and Drug Administration for treatment of FL, they have been investigated extensively in this disease in light of the enhanced B-cell receptor activation observed in FL.63 Despite promising preclinical data, the activity of single-agent ibrutinib has been shown to be limited in FL, with an overall response rate of 37% to 54% and a median PFS of only 11 to 14 months in patients with relapsed disease, emphasizing the need to better understand its biological effects on the FL TME, including macrophages.64,65 Of interest, 1 of the proposed mechanisms of FL genesis includes aberrant activation of B-cell receptor signaling based on the direct interaction between endogenous lectin expressed on macrophages and atypical sugar moieties expressed on the surface immunoglobulin of FL cells, leading to constitutive activation of ERK1/2, AKT, and PLCγ2. In vitro data show that this interaction is primarily mediated by M2 macrophages and can be effectively abrogated by the use of ibrutinib via dephosphorylation of the above targets.63,66 Additional in vitro data show that inhibition of BTK in macrophages abrogates the secretion of homeostatic and angiogenic chemokines, such as CXCL12, CXCL13, CCL19, and VEGF, and the supernatant collected from ibrutinib-treated macrophages can significantly compromise adhesion, invasion, and migration of lymphoid malignant cells, as well as endothelial cell tube formation.67
More recently, ibrutinib was shown to synergize with anti-CD20 monoclonal antibodies to increase macrophage-mediated phagocytosis. By utilizing an ADCP coculture system, Barbarino and colleagues were able to demonstrate that ibrutinib enhanced the phagocytosis of rituximab- and obinutuzumab-opsonized lymphoma cells, independent of BTK inhibition, as a result of its off-target effect against JAK2.68 This may explain the promising clinical activity of the combination of ibrutinib and rituximab as frontline therapy that was reported recently for patients with FL.69
Acalabrutinib is a more selective and potent BTK inhibitor than ibrutinib, with proven clinical efficacy as a single agent or in combination with rituximab in patients with FL.70 Likely as a result of the lack of an off-target effect, chronic lymphocytic leukemia preclinical models have shown, by flow cytometry or direct phagocytosis assays, that acalabrutinib does not impact macrophage phagocytosis compared with ibrutinib, at any dose.71 Preclinical data regarding the impact of acalabrutinib on TAMs in FL are missing and are eagerly awaited to guide the next generation of clinical trials in this space.
PI3K inhibitors
The phosphatidylinositol 3-kinase (PI3K)/Akt/mTOR pathway is constitutively active in many B-cell malignancies, including FL, and its targeting with idelalisib (a δ inhibitor), duvelisib (a ɣδ inhibitor), copanlisib (a αδ inhibitor), and umbralisib (a CK1/δ inhibitor) has proven clinical efficacy in patients with relapsed or refractory FL.72-75 Preclinical data suggest that the therapeutic effects of PI3K inhibitors can be explained, in part, by modulation of the TME. In coculture assays of patient-derived FL cells and follicular dendritic cells, PI3Kδ inhibition through idelalisib upregulated follicular dendritic cell–induced genes related to angiogenesis, extracellular matrix formation, and transendothelial migration.76 Furthermore, macrophages protected FL cells from BCL-2–associated agonist of cell death (BAD), and the addition of idelalisib restored FL dependence on BCL-2 primarily through reduction of BAD serine phosphorylation on the direct PI3K target Ser136 and downregulation of BFL-1 expression, suggesting a potential synergy with BCL-2 inhibitors, such as venetoclax (VTX) (refer to BCL-2 inhibitors).
Mouse models have demonstrated that PI3Kγ signaling functions through CCAAT/enhancer binding protein (C/EBPβ) as a key inhibitor of phagocytosis in TAMs, inducing an immunosuppressive transcriptome.77 PI3Kγ inhibition, in fact, stimulates and prolongs NF-κB activation, which, in turn, inhibits C/EBPβ activation, leading to TAM polarization toward an M1 phenotype. These findings have been confirmed in multiple preclinical lymphoma models, showing a consistent polarization of TAMs from an M2 to an M1 phenotype with the use of PI3Kγ inhibitors.78,79
EZH2 inhibitors
EZH2 is a histone methyltransferase that supports germinal center formation in B cells and whose gain-of-function mutations have been reported in ∼25% of FL cases.80 Tazemetostat is an oral selective inhibitor of mutant or wild-type EZH2 with proven clinical efficacy in patients with relapsed or refractory FL.81 Transcriptomics and epigenomics analyses showed that IFN-γ can recruit EZH2 at promoters of select anti-inflammatory genes, such as MERTK, PPARG, and RANK, resulting in their silencing and inducing an activated macrophage phenotype.82 Although FL germinal centers are enriched with follicular helper T cells, which is characterized by a low production of IFN-γ, EZH2 expression in FL-derived macrophages has not been investigated and could be independent of IFN-γ.83 Data regarding the impact of EZH2 inhibitors on TAMs in FL are missing and may inform future combination strategies.
BCL-2 inhibitors
Despite BCL-2 translocation being the hallmark of FL, limited clinical activity has been reported for VTX in patients with relapsed FL, including a median PFS of 11 months and with only 14% achieving a complete response.84 In addition, significant synergy has not been observed when combining this BCL-2 inhibitor with rituximab or BR , highlighting the need to shed light on its activity on the TME, including macrophages.85 To this regard, in vitro data suggest that the immune microenvironment could modulate FL dependence on BCL-2. In fact, BH3 profiling has shown that, when cultured alone, FL cells are strongly dependent on BCL-2 and are highly sensitive to VTX. However, FL coculture with macrophages displayed lower response to BAD peptide and increased dependence on BFL-1, with subsequent decreased sensitivity to VTX. Interestingly, as outlined above, the addition of idelalisib to FL cocultures downregulated BFL-1 expression, restoring proapoptotic BAD activity and VTX efficacy.76
Future directions: CD47/SIRPα-blocking agents in FL
Malignant cells, including FL, upregulate CD47, a widely expressed transmembrane protein that interacts with its macrophage receptor SIRPα to avoid phagocytosis.86,87 A growing number of studies have demonstrated that inhibiting the CD47-SIRPα signaling pathway enhances macrophage-mediated phagocytosis of tumor cells. Chen et al demonstrated that SIRPα-expressing monocytes/macrophages in the TME have a functional and prognostic impact in FL.88 Three key intratumoral macrophage subsets (CD14+SIRPαhi, CD14SIRPαlow, and CD14−SIRP αneg) were identified using tumor biopsies from patients with FL, followed by in vitro enrichment for SIRPα-expressing cells. SIRPα-CD47 blockade using SIRPα-Fc alone or in combination with rituximab enhanced phagocytosis across all 3 subsets, with CD14+SIRPαhi cells displaying the greatest degree of phagocytosis. The prognostic impact of SIRPα-defined subsets on a cohort of 83 previously untreated patients with FL revealed that CD14+SIRPαhi cells were significantly (P < .03) associated with poorer survival, consistent with findings from the prerituximab era.6,44 The CD14−SIRPαlow subset appeared to be associated with better outcomes, although statistical significance was not reached, whereas CD14−SIRPαneg cells did not have a prognostic impact.88
In vitro phagocytosis assays using human and mouse macrophages demonstrated that CD47 blockade can significantly improve phagocytosis of lymphoma cells, particularly in combination with rituximab, as a result of increased ADCP.87 The efficacy of this combination was also confirmed in vivo in a xenotransplantation mouse model injected with Raji cells, with a 86% reduction in tumor volume. Even better results were reported in a xenograft mouse model transplanted with patient-derived FL cells, with complete tumor resolution. Interestingly, the efficacy of this combination was similar in a complement- and natural killer cell–deficient lymphoma xenotransplantation model compared with a complement and natural killer cell–competent mouse model, whereas no efficacy was observed after macrophage depletion, suggesting that the latter alone is sufficient to mediate its therapeutic effect.
Such promising preclinical data have translated into significant clinical activity in FL, with an overall response rate of 71% and a complete response rate of 43% in 7 patients with relapsed refractory FL who were treated with a combination of an anti-CD47 antibody (5F9 or magrolimab) and rituximab.89 Similarly encouraging results were reported in FL with TTI-621, a recombinant soluble fusion protein consisting of the CD47-binding domain of human SIRPα and the Fc region of human immunoglobulin G1, and ALX-148, a fusion protein consisting of a high-affinity CD47 blocker linked to an inactive human immunoglobulin Fc region.90,91 Although these and other agents that block CD47-SIRPα cross talk show increasing activity in FL, research aimed at minimizing their toxicity (primarily represented by cytopenia) and increasing their efficacy is ongoing.92
Conclusions
Macrophages play a crucial role in the pathophysiology of FL, and their increased content was associated with worse outcomes among patients treated with chemotherapy in the prerituximab era. However, approved and novel therapies that are available for the treatment of FL differentially impact macrophage phenotype, function, and concentration, challenging this established paradigm. In addition, clinical translational studies demonstrated the limitations of techniques that are available for the characterization of macrophages. In fact, single-cell analysis is a highly effective technique for the characterization of the lymphoid TME ; the tissue manipulation required for its use may limit its application to the characterization of tissue macrophages. In addition, it does not provide any information about the complex spatial cellular interactions occurring within the immune microenvironment. Conventional IHC is a widely used diagnostic technique in tissue pathology, but it is associated with a number of limitations, including high interobserver variability and the ability to label only 1 marker per tissue section. Therefore, highly multiplexed techniques have emerged to circumvent these constraints93 ; they have been applied to tissue biopsies obtained from patients with lymphoma treated with immunotherapy and resulted in the identification of 30 cell clusters with spatial information, including an M2 macrophage cluster that was enriched in nonresponders before therapy.94 A more consistent use of these technologies may help to address the conflicting translational data outlined above and provide better insight into the impact of approved and novel agents on macrophage phenotype and function. A deeper understanding of macrophage biology and their complex pathways will help to develop novel and safer therapeutic strategies for patients with FL, potentially impacting the ultimate paradigm, a lack of cure.
Acknowledgments
The authors thank David Aten for graphical support.
P.S. is supported by a Lymphoma Research Foundation Career Development Award.
Authorship
Contribution: S.G., M.L.M.-P., and P.S. critically reviewed the literature and wrote the manuscript.
Conflict-of-interest disclosure: P.S. is a consultant for Genentech-Roche and has received research support from AstraZeneca. The remaining authors declare no competing financial interests.
Correspondence: Paolo Strati, The University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd, Unit 429, Houston, TX 77030; e-mail: pstrati@mdanderson.org.
References
Author notes
Data sharing requests should be sent to Paolo Strati (pstrati@mdanderson.org).