Abstract
Because abnormal erythroid differentiation is the most common manifestation of the myelodysplastic syndromes (MDS), it was hypothesized that erythroid gene expression may be used to illustrate myelodysplastic transcription patterns. Ten normal bone marrow aspirates (NBM) were first analyzed using an erythroid-focused cDNA array to define steady-state transcription levels. Proliferation and differentiation gene subsets were identified by statistically significant differences between NBM and erythroleukemia gene expression. Next, cDNAs from 5 separate MDS aspirates were studied: refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation (RAEB-T), and RAEB-T/secondary MDS. A distinct pattern of significantly increased proliferation-associated and reduced differentiation-associated gene activity was established for MDS.
Introduction
The myelodysplastic syndromes (MDS) have shared features of refractory cytopenia, dysplastic cellular morphology, and a propensity toward malignant transformation.1 The biologic basis of these features appears to be the uncoupling of proliferation and differentiation during hematopoiesis.2 Changes in the level of programmed cell death during differentiation also contribute to the ineffective hematopoiesis associated with MDS.3Leukemic transformation is thought to arise from cumulative genetic damage in the dysplastic stem cells. The nature of these insults can often be traced to growth-related proteins,4 and study of the genetic mutations associated with myelodysplasia provides insight into the disease pathophysiology. Despite these conceptual advances, supportive care has remained the mainstay of MDS clinical care for decades. Problems with clinical management of MDS relate to the therapeutic risks inherent in treating elderly patients and the diversity of the disease. Diagnosis of MDS can be difficult, with potential for confusion between MDS and aplastic anemia5and myeloproliferative disease.6 Although not ideal, the French-American-British classification system for MDS7 has remained a diagnostic standard in clinical practice for almost 2 decades. However, this system does not take genetic pathways into account. An international prognostic scoring system for MDS has been developed more recently that includes cytogenetic data.8Nevertheless, existing clinical tools are limited in their ability to confirm the diagnosis, predict disease progression, or gauge treatment responses in individual MDS patients.
Although cytogenetic abnormalities have become a hallmark of MDS, many of those abnormalities are complex and not fully understood. Both loss-of-function and gain-of-function mutations may result in myelodysplasia. Some genes, such as N-RAS andp53, are more commonly mutated.9,10Genetic abnormalities on the long arm of chromosome 5 have been positively and negatively associated with survival.11Despite large numbers of structural genomic mutations that are associated with myelodysplasia, only a small number of morphologic changes result. By determining the pattern of gene expression that is normally involved in hematopoiesis, it should therefore be possible to improve our ability to assess the biologic effects of MDS-related genetic mutations. A pattern of overexpressed growth-related and underexpressed differentiation-related genes would be predicted. To test this hypothesis, we selected the erythroid lineage for study because it is commonly involved in all MDS syndromes and measured bone marrow expression of genes known to be transcribed in normal human proliferating erythroid cells. Gene expression patterns identified in normal marrow were compared with those in myelodysplastic marrow for the purpose of identifying general and patient-specific transcriptional abnormalities.
Materials and methods
Cells and cDNA
Bone marrow, including that from patients with myelodysplasia, and peripheral blood mononuclear cells were obtained with clinical review and approval. Commercially available human bone marrow was also used (Poietics, Rockville, MD). Cell lines were obtained from laboratory stocks or purchased (American Type Culture Collection, Rockville, MD). All cDNA was chosen from clones derived from proliferating human erythroid cells (http://hembase.niddk.nih.gov/). In addition to clones identified by the suppression subtraction technique,12 a separate normalized library called “AX” was constructed. The AX library was generated as follows. First, 65 000 proliferating erythroid cells from the buffy coat of a blood donation were obtained by flow cytometric separation after a 5-day culture period in the presence of erythropoietin. Total RNA was purified from the sorted cell population using TRIzol reagent. RNA (0.3 μg) was converted into double-stranded cDNA using Clontech's CapFinder cDNA Library Construction Kit (Clontech, Palo Alto, CA), according to the manufacturer's protocol, and cloned intoEcoRI-digested Lambda Zap II vector (Stratagene, La Jolla, CA). The phage library was amplified once before in vivo excision in SOLR cells. Individual colonies were grown, and the cDNA inserts were sequenced in high throughput (National Institutes of Health intramural sequencing center http://www.nisc.nih.gov/). Clones chosen for the array were regrown and resequenced in a separate laboratory to confirm their identities. The plasmid inserts were amplified by polymerase chain reaction (PCR) using the forward primer 5′-TCGAGCGGCCGCCCGGGCAGGT-3′ and reverse primer 5′-AGCGTGGTCGCGGCCGAGGT-3′ for the AD clones. The forward primer 5′-CGCTCTAGAACTAGTGGATC-3′ and reverse primer 5′-TCGAGGTCGACGGTATC-3′ were used for the AX plasmids. PCR was performed using the Advantage cDNA PCR Kit (Clontech). PCR products were analyzed on 1.2% E-Gel (Invitrogen, Carlsbad, CA) by taking 5 μL from each sample before purification. Only samples demonstrating a single band were purified for spotting (QIAquick PCR Purification Kit; Qiagen). Purified PCR products were dried and stored at −20°C and resuspended in 20 μL of 3 × SSC buffer overnight before printing of the microarray.
Preparation of the E-chips
Glass slides were precleaned by submerging in cleaning solution (350 mL containing 250 mL 95% ethanol, 50 g sodium hydroxide, and 100 mL double distilled water) for 2 hours with shaking. After cleaning, the slides were rinsed 5 times in double distilled water with vigorous plunging for 5 minutes to remove all residual sodium hydroxide. The rinsed slides were then submerged for 1 hour in coating solution (280 mL double distilled water containing 35 mL poly-l-lysine solution [Sigma, St. Louis, MO] and 35 mL tissue culture grade phosphate-buffered saline). After a brief rinse with double distilled water for 15 seconds, they were centrifuged (80g) for 3 minutes. Lysine-coated slides were sealed in a clean slide box without cork liner and allow to age at room temperature for 2 weeks before printing. Printing was accomplished using an OmniGrid Microarrayer robot (GeneMachines, San Carlos, CA). The spots contained approximately 200 pL cDNA and had an average center-to-center distance of 350 μm. Finally, 11 × 11 spot arrays were printed in quadruplicate on each glass slide. Some cDNA PCR products were spotted more than once in each quadrant for quality control.
After printing, all slides were numbered using a diamond-tipped scriber (VWR, South Plainfield, NJ) and then UV cross-linked with the DNA side up in a Stratalinker (Stratagene) with 60 000 μJ. The UV cross-linked arrays were submerged in blocking solution (15 mL 1 M boric acid, pH 8.0 [Sigma], added to a solution containing 6 g succinic anhydride [Sigma] in 335 mL 1-methyl-2-pyrrolidinone [Sigma]) with vigorous plunging to avoid artifacts. The chips were then placed on an orbital shaker for 15 minutes, followed by rinsing with double distilled water. A final denaturing step consisted of submerging the chips in boiling water for 2 minutes, then briefly in 95% ethanol. The chips were then centrifuged (80g for 2 minutes) and stored under vacuum before use.
Preparation of the cDNA probes
Total RNA (50 μg) was used as template to generate first-strand cDNA probes labeled with Cy3 or Cy5 dyes using reverse transcription. Each reverse transcription reaction mix contained 8 μL of 5 × first-strand buffer, 2 μL of 2 μg/μL oligo-dT 12-18mer (Life Technologies, Rockville, MD), 2 μL of 20 × low-dT/NTP mix, 4 μL of 1 mM Cy3 or Cy5 dUTP (Amersham Pharmacia Biotech, Piscataway, NJ), 4 μL of 0.1 M dithiothreitol, 1 μL of 40 U/μL RNAsin (Promega, Madison, WI), 50 μg total RNA in 17 μL, and 2 μL of 200 U/μL SuperScript II reverse transcriptase (Life Technologies). The mixture was incubated at 65°C for 5 minutes to denature the total RNA, then allowed to cool at room temperature for 2.5 minutes. After denaturation, 2 μL SuperScript II was added to the mixture and allowed to incubate at 42°C for 25 minutes, then another 2 μL Superscript II was added to the mixture for an additional 35 minutes. Adding 5 μL of 500 mM EDTA stopped the reaction. Total RNA residual was hydrolyzed by adding 10 μL of 1 M sodium hydroxide and heated to 65°C for 1 hour, followed by addition of 25 μL of 1 M Tris-HCl, pH 7.5. The probe was purified by using Microcon YM-30 columns (Millipore, Bedford, MA). Briefly, 500 μL Tris-EDTA (TE), pH 7.4, was added to the Microcon YM column and centrifuged in an Eppendorff Centrifuge 5414D (Brinkmann Instruments, Westbury, NY) at 13 000 rpm for 8 minutes or until all buffer had passed through the column. To each probe sample, 400 μL TE, pH 7.4, was added and then transferred to a prewashed column. Each column was then centrifuged at 13 000 rpm for about 8 minutes or until the probe was in the 20- to 40-μL range. The appropriate Cy3 probes (usually bone marrow derived) were then combined with Cy5 probes (K562 cell derived) in one column and washed with TE buffer twice more (final volume less than or equal to 8 μL). Labeled probes were recovered by centrifuging the inverted columns for 1 minute at 13 000 rpm. Samples volumes were increased to 11 μL with TE buffer. A final hybridization volume of 17.6 μL was achieved by adding 1 μL of 10 μg/μL human COT-1 DNA (Boehringer Mannheim, Indianapolis, IN), 1 μL of 10 μg/μL polyA (Amersham Pharmacia Biotech), 1 μL of 4 μg/μL yeast tRNA (Sigma), 3.1 μL of 20 × SSC, and 0.5 μL of 10% sodium dodecyl sulfate (SDS). The probe was then denatured at 100°C for 2 minutes and immediately centrifuged at 13 000 rpm for 10 minutes at room temperature to pellet any particulate matter. The supernatant was transferred to a fresh tube.
The probes were then placed directly on the E-chip and quickly overlaid with a 22 × 22-mm cover slip. The chips were then placed in hybridization chambers (Telechem International, Sunnyvale, CA) containing 40 μL double distilled water on both sides of the chamber to maintain the humidity throughout hybridization. The arrays were incubated in a 65°C water bath for 16 hours. Hybridized microarrays were washed once in successive buffers of 2 × SSC with 0.1% SDS, 1 × SSC, 0.2 × SSC, and 0.05 × SSC with gentle plunging for 1 minute in each wash buffer. The arrays were centrifuged (100g for 4 minutes) before scanning.
Data collection
The arrays were scanned using an Axon Genepix 4000 scanner (Axon Instruments, Foster City, CA) using Genepix 1.0 software. A quick preview of the microarray was done with low-resolution scan at 40 μ with photo multiplier tube (PMT) set at 800 V for Cy3 (532 nm) and Cy5 (635 nm). A high-resolution scan (10 μ per pixel) was then performed to compile a raw data set for each microarray. All data were saved and analyzed using Arraysuite software (Scanalytics, Fairfax, VA) to determine the signal intensities for 532-nm and 635-nm channels and generated scanned images. Contrast was adjusted automatically by the software, and an 11 × 11 grid was overlaid on each quadrant of the microarray by adjusting the size of the grid manually to fit all spots. Dearray function was chosen next with 635-nm scan to be first and 532-nm scan to be second. A Gipo file was created to identify each spot with the encoded gene. Image files as well as those containing intensity data for each spot in both channels were used for visual correlation and statistical analyses. No E-chip spot images shown in this manuscript were corrected or further pseudocolored before reproduction. All statistical analyses were performed using SAS and SPLUS statistical packages (SAS Institute, Cary, NC).
Results
Clone selection and hybridization controls
The genes selected for study were identified in proliferating erythroid cells.12 13 Those transcripts have been identified and catalogued (http://hembase.niddk.nih.gov/). A small group of 106 cDNAs was spotted in quadruplicate arrays on each chip (Table 1). The encoded genes included leukemia-associated transcripts that we identified in normal erythroid cells as well as genes such as β-globin with defined erythroid specificity. Approximately one third of the transcripts were “novel” (defined by their lack of homology with other genes deposited in the public domain at the time of printing).
Spotted genes and intensities
| Gene . | NBM Intensity . | K562 Intensity . | NBM/K562 Ratio . |
|---|---|---|---|
| 1 γ-globin | 3 667 | 38 316 | 0.10 (P) |
| 2 TOPII | 828 | 5 917 | 0.14 (P) |
| 3 Ad08e03 | 583 | 3 947 | 0.15 (P) |
| 4 Translin | 549 | 3 429 | 0.16 (P) |
| 5 Transglutaminase | 613 | 3 486 | 0.18 (P) |
| 6 NPM-1 | 4 813 | 25 864 | 0.19 (P) |
| 7 p23 | 3 552 | 16 027 | 0.22 (P) |
| 8 ε-globin | 985 | 4 485 | 0.22 (P) |
| 9 nm23-H2 | 3 711 | 16 038 | 0.23 (P) |
| 10 TOM20 | 2 545 | 11 126 | 0.23 (P) |
| 11 pim-1 | 1 840 | 7 740 | 0.24 (P) |
| 12 Madp2 | 615 | 2 444 | 0.25 (P) |
| 13 Stathmin | 3 008 | 11 790 | 0.26 (P) |
| 14 Dek | 1 857 | 6 786 | 0.27 (P) |
| 15 TFG | 765 | 2 872 | 0.27 (P) |
| 16 Triosephosphate isomerase | 3 749 | 13 056 | 0.29 (P) |
| 17 hmlh1 | 827 | 2 734 | 0.30 (P) |
| 18 NADH B-12 | 1 456 | 4 601 | 0.32 (P) |
| 19 Ad13f05 | 11 148 | 33 994 | 0.33 (P) |
| 20 IMP | 1 497 | 4 579 | 0.33 (P) |
| 21 LCAT | 5 513 | 16 188 | 0.34 (P) |
| 22 XP-C protein | 1 205 | 3 461 | 0.35 (P) |
| 23 HMG SSRP1 | 2 649 | 7 299 | 0.36 (P) |
| 24 Ferritin L chain | 8 973 | 24 365 | 0.37 (P) |
| 25 Odc1 | 2 082 | 5 619 | 0.37 (P) |
| 26 APRT | 1 754 | 4 545 | 0.39 (P) |
| 27 Transferrin receptor | 5 400 | 13 562 | 0.40 (P) |
| 28 c-myb | 1 223 | 2 954 | 0.41 (P) |
| 29 NF45 | 2 955 | 7 079 | 0.42 (P) |
| 30 hnRNP D | 7 718 | 16 976 | 0.45 (P) |
| 31 EF-1α | 20 826 | 44 785 | 0.46 (P) |
| 32 dbpB-like protein | 1 363 | 2 902 | 0.47 (P) |
| 33 Ad14g01 | 10 244 | 21 415 | 0.48 (P) |
| 34 Mus ubc4 | 942 | 1 973 | 0.48 (P) |
| 35 HMG-14 | 1 939 | 3 924 | 0.49 (P) |
| 36 α-NAC | 8 908 | 17 607 | 0.51 (P) |
| 37 Ad03c11 | 1 682 | 3 220 | 0.52 (P) |
| 38 Apo1 | 2 993 | 5 465 | 0.55 (P) |
| 39 hEGR1 | 1 209 | 2 142 | 0.56 (P) |
| 40 EGR1 | 1 019 | 1 787 | 0.57 (P) |
| 41 KIAA0201 | 1 684 | 2 863 | 0.59 (P) |
| 42 TOPO II BP | 921 | 1 553 | 0.59 (P) |
| 43 E2 protein | 9 058 | 14 982 | 0.60 (P) |
| 44 SON DNA binding protein | 927 | 1 547 | 0.60 (P) |
| 45 FUS | 3 613 | 5 712 | 0.63 (P) |
| 46 Phospholipase A2 | 4 028 | 6 211 | 0.65 (P) |
| 47 MAN2A2 | 1 615 | 2 433 | 0.66 (P) |
| 48 H3.3 gene exon 4 | 11 524 | 16 678 | 0.69 (P) |
| 49 BLM | 13 455 | 17 599 | 0.76 (P) |
| 50 HMG-17 | 11 355 | 14 720 | 0.77 (P) |
| 51 DIP1 | 8 658 | 10 932 | 0.79 (P) |
| 52 HMG-1 | 10 144 | 12 019 | 0.84 (P) |
| 53 PR264 | 5 400 | 6 400 | 0.84 (P) |
| 54 Ad00178 | 8 092 | 9 147 | 0.88 (U) |
| 55 Mitochondrial DNA, COIII | 15 972 | 17 952 | 0.89 (U) |
| 56 Ferrochelatase | 5 148 | 4 395 | 1.2 (U) |
| 57 Ad08f03 | 1 898 | 1 545 | 1.2 (U) |
| 58 Glycophorin A | 5 882 | 4 547 | 1.3 (U) |
| 59 Ankyrin | 3 573 | 2 847 | 1.3 (U) |
| 60 TCTP | 30 174 | 18 882 | 1.6 (D) |
| 61 Actin | 21 637 | 12 029 | 1.8 (D) |
| 62 ERF-1 | 4 282 | 1 843 | 2.3 (D) |
| 63 Ad02h07 | 3 734 | 707 | 5.3 (D) |
| 64 Ad08c04 | 2 801 | 391 | 7.2 (D) |
| 65 α-globin | 32 620 | 4 241 | 7.7 (D) |
| 66 CD36 | 4 864 | 506 | 9.6 (D) |
| 67 CAI | 27 090 | 933 | 29 (D) |
| 68 δ-globin | 25 847 | 745 | 35 (D) |
| 69 CAII | 7 405 | 150 | 49 (D) |
| 70 c-fos | 10 812 | 203 | 53 (D) |
| 71 β-globin | 40 383 | 333 | 120 (D) |
| 72 ABL | 255 | 1 110 | 0.23 (L) |
| 73 PKCι | 516 | 1 346 | 0.38 (L) |
| 74 Ad00044 | 285 | 540 | 0.53 (L) |
| 75 Ferritin H chain | 674 | 1 125 | 0.60 (L) |
| 76 Ad03e02 | 603 | 985 | 0.61 (L) |
| 77 Ad07f01 | 257 | 424 | 0.61 (L) |
| 78 TAL1 | 911 | 1 470 | 0.62 (L) |
| 79 p21 | 822 | 1 310 | 0.63 (L) |
| 80 M96 | 309 | 486 | 0.64 (L) |
| 81 Ad12d02 | 871 | 1 241 | 0.70 (L) |
| 82 KIAA0152 | 513 | 703 | 0.73 (L) |
| 83 KIAA0806 | 587 | 788 | 0.74 (L) |
| 84 Porcine inhibin subunit | 555 | 720 | 0.77 (L) |
| 85 TOPI | 818 | 1 051 | 0.78 (L) |
| 86 Ad08d07 | 234 | 265 | 0.88 (L) |
| 87 XP22 | 201 | 222 | 0.90 (L) |
| 88 Ad13d04 | 315 | 345 | 0.91 (L) |
| 89 PSM | 241 | 262 | 0.92 (L) |
| 90 Ad09b01 | 724 | 765 | 0.95 (L) |
| 91 Ad13f12 | 493 | 515 | 0.96 (L) |
| 92 Ad14h09 | 238 | 248 | 0.96 (L) |
| 93 HRX | 494 | 492 | 1.0 (L) |
| 94 MTG-8 | 179 | 149 | 1.2 (L) |
| 95 Ad10e12 | 235 | 180 | 1.3 (L) |
| 96 Ad08d12 | 206 | 164 | 1.3 (L) |
| 97 Ad02e07 | 188 | 132 | 1.4 (L) |
| 98 Ad14f03 | 1 347 | 870 | 1.5 (L) |
| 99 CBF-A2 | 172 | 114 | 1.5 (L) |
| 100 Ad08g11 | 529 | 339 | 1.6 (L) |
| 101 Ad03c09 | 152 | 94 | 1.6 (L) |
| 102 Ad06b01 | 568 | 267 | 2.1 (L) |
| 103 AF9 | 445 | 56 | 7.9 (L) |
| 104 Blank | 991 | 714 | 1.4 (L) |
| 105 Vector PCR | 844 | 527 | 1.6 (L) |
| 106 Whole vector | 382 | 156 | 2.4 (L) |
| Gene . | NBM Intensity . | K562 Intensity . | NBM/K562 Ratio . |
|---|---|---|---|
| 1 γ-globin | 3 667 | 38 316 | 0.10 (P) |
| 2 TOPII | 828 | 5 917 | 0.14 (P) |
| 3 Ad08e03 | 583 | 3 947 | 0.15 (P) |
| 4 Translin | 549 | 3 429 | 0.16 (P) |
| 5 Transglutaminase | 613 | 3 486 | 0.18 (P) |
| 6 NPM-1 | 4 813 | 25 864 | 0.19 (P) |
| 7 p23 | 3 552 | 16 027 | 0.22 (P) |
| 8 ε-globin | 985 | 4 485 | 0.22 (P) |
| 9 nm23-H2 | 3 711 | 16 038 | 0.23 (P) |
| 10 TOM20 | 2 545 | 11 126 | 0.23 (P) |
| 11 pim-1 | 1 840 | 7 740 | 0.24 (P) |
| 12 Madp2 | 615 | 2 444 | 0.25 (P) |
| 13 Stathmin | 3 008 | 11 790 | 0.26 (P) |
| 14 Dek | 1 857 | 6 786 | 0.27 (P) |
| 15 TFG | 765 | 2 872 | 0.27 (P) |
| 16 Triosephosphate isomerase | 3 749 | 13 056 | 0.29 (P) |
| 17 hmlh1 | 827 | 2 734 | 0.30 (P) |
| 18 NADH B-12 | 1 456 | 4 601 | 0.32 (P) |
| 19 Ad13f05 | 11 148 | 33 994 | 0.33 (P) |
| 20 IMP | 1 497 | 4 579 | 0.33 (P) |
| 21 LCAT | 5 513 | 16 188 | 0.34 (P) |
| 22 XP-C protein | 1 205 | 3 461 | 0.35 (P) |
| 23 HMG SSRP1 | 2 649 | 7 299 | 0.36 (P) |
| 24 Ferritin L chain | 8 973 | 24 365 | 0.37 (P) |
| 25 Odc1 | 2 082 | 5 619 | 0.37 (P) |
| 26 APRT | 1 754 | 4 545 | 0.39 (P) |
| 27 Transferrin receptor | 5 400 | 13 562 | 0.40 (P) |
| 28 c-myb | 1 223 | 2 954 | 0.41 (P) |
| 29 NF45 | 2 955 | 7 079 | 0.42 (P) |
| 30 hnRNP D | 7 718 | 16 976 | 0.45 (P) |
| 31 EF-1α | 20 826 | 44 785 | 0.46 (P) |
| 32 dbpB-like protein | 1 363 | 2 902 | 0.47 (P) |
| 33 Ad14g01 | 10 244 | 21 415 | 0.48 (P) |
| 34 Mus ubc4 | 942 | 1 973 | 0.48 (P) |
| 35 HMG-14 | 1 939 | 3 924 | 0.49 (P) |
| 36 α-NAC | 8 908 | 17 607 | 0.51 (P) |
| 37 Ad03c11 | 1 682 | 3 220 | 0.52 (P) |
| 38 Apo1 | 2 993 | 5 465 | 0.55 (P) |
| 39 hEGR1 | 1 209 | 2 142 | 0.56 (P) |
| 40 EGR1 | 1 019 | 1 787 | 0.57 (P) |
| 41 KIAA0201 | 1 684 | 2 863 | 0.59 (P) |
| 42 TOPO II BP | 921 | 1 553 | 0.59 (P) |
| 43 E2 protein | 9 058 | 14 982 | 0.60 (P) |
| 44 SON DNA binding protein | 927 | 1 547 | 0.60 (P) |
| 45 FUS | 3 613 | 5 712 | 0.63 (P) |
| 46 Phospholipase A2 | 4 028 | 6 211 | 0.65 (P) |
| 47 MAN2A2 | 1 615 | 2 433 | 0.66 (P) |
| 48 H3.3 gene exon 4 | 11 524 | 16 678 | 0.69 (P) |
| 49 BLM | 13 455 | 17 599 | 0.76 (P) |
| 50 HMG-17 | 11 355 | 14 720 | 0.77 (P) |
| 51 DIP1 | 8 658 | 10 932 | 0.79 (P) |
| 52 HMG-1 | 10 144 | 12 019 | 0.84 (P) |
| 53 PR264 | 5 400 | 6 400 | 0.84 (P) |
| 54 Ad00178 | 8 092 | 9 147 | 0.88 (U) |
| 55 Mitochondrial DNA, COIII | 15 972 | 17 952 | 0.89 (U) |
| 56 Ferrochelatase | 5 148 | 4 395 | 1.2 (U) |
| 57 Ad08f03 | 1 898 | 1 545 | 1.2 (U) |
| 58 Glycophorin A | 5 882 | 4 547 | 1.3 (U) |
| 59 Ankyrin | 3 573 | 2 847 | 1.3 (U) |
| 60 TCTP | 30 174 | 18 882 | 1.6 (D) |
| 61 Actin | 21 637 | 12 029 | 1.8 (D) |
| 62 ERF-1 | 4 282 | 1 843 | 2.3 (D) |
| 63 Ad02h07 | 3 734 | 707 | 5.3 (D) |
| 64 Ad08c04 | 2 801 | 391 | 7.2 (D) |
| 65 α-globin | 32 620 | 4 241 | 7.7 (D) |
| 66 CD36 | 4 864 | 506 | 9.6 (D) |
| 67 CAI | 27 090 | 933 | 29 (D) |
| 68 δ-globin | 25 847 | 745 | 35 (D) |
| 69 CAII | 7 405 | 150 | 49 (D) |
| 70 c-fos | 10 812 | 203 | 53 (D) |
| 71 β-globin | 40 383 | 333 | 120 (D) |
| 72 ABL | 255 | 1 110 | 0.23 (L) |
| 73 PKCι | 516 | 1 346 | 0.38 (L) |
| 74 Ad00044 | 285 | 540 | 0.53 (L) |
| 75 Ferritin H chain | 674 | 1 125 | 0.60 (L) |
| 76 Ad03e02 | 603 | 985 | 0.61 (L) |
| 77 Ad07f01 | 257 | 424 | 0.61 (L) |
| 78 TAL1 | 911 | 1 470 | 0.62 (L) |
| 79 p21 | 822 | 1 310 | 0.63 (L) |
| 80 M96 | 309 | 486 | 0.64 (L) |
| 81 Ad12d02 | 871 | 1 241 | 0.70 (L) |
| 82 KIAA0152 | 513 | 703 | 0.73 (L) |
| 83 KIAA0806 | 587 | 788 | 0.74 (L) |
| 84 Porcine inhibin subunit | 555 | 720 | 0.77 (L) |
| 85 TOPI | 818 | 1 051 | 0.78 (L) |
| 86 Ad08d07 | 234 | 265 | 0.88 (L) |
| 87 XP22 | 201 | 222 | 0.90 (L) |
| 88 Ad13d04 | 315 | 345 | 0.91 (L) |
| 89 PSM | 241 | 262 | 0.92 (L) |
| 90 Ad09b01 | 724 | 765 | 0.95 (L) |
| 91 Ad13f12 | 493 | 515 | 0.96 (L) |
| 92 Ad14h09 | 238 | 248 | 0.96 (L) |
| 93 HRX | 494 | 492 | 1.0 (L) |
| 94 MTG-8 | 179 | 149 | 1.2 (L) |
| 95 Ad10e12 | 235 | 180 | 1.3 (L) |
| 96 Ad08d12 | 206 | 164 | 1.3 (L) |
| 97 Ad02e07 | 188 | 132 | 1.4 (L) |
| 98 Ad14f03 | 1 347 | 870 | 1.5 (L) |
| 99 CBF-A2 | 172 | 114 | 1.5 (L) |
| 100 Ad08g11 | 529 | 339 | 1.6 (L) |
| 101 Ad03c09 | 152 | 94 | 1.6 (L) |
| 102 Ad06b01 | 568 | 267 | 2.1 (L) |
| 103 AF9 | 445 | 56 | 7.9 (L) |
| 104 Blank | 991 | 714 | 1.4 (L) |
| 105 Vector PCR | 844 | 527 | 1.6 (L) |
| 106 Whole vector | 382 | 156 | 2.4 (L) |
Mean signals were ranked by calculating the ratio of normal bone marrow (Cy3) mean intensity and K562 erythroleukemia cell (Cy5) mean intensity. All mean values are derived from 40 spots (10 NBM chips × 4 spots per chip). According to the statistical criteria defined in Figure 1, genes 1-53 were grouped as proliferation associated (P), genes 54-59 were grouped as equivalent or undefined (U), genes 60-71 were grouped as differentiation associated (D), and genes 72-106 with intensities in both channels at or below the level of background or negative controls were grouped as low intensity (L). Further details on the clones used for this study are available athttp://hembase.niddk.nih.gov/.
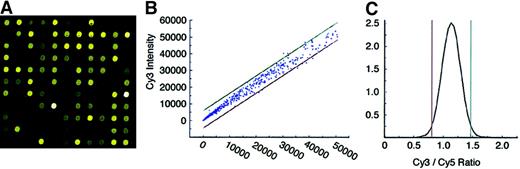
For quality control, cDNAs from a common pool (used throughout the study) of K562 total RNA were labeled with Cy3 and Cy5 tags in separate tubes. The labeled first-strand cDNAs (referred to here as the probes) were then cohybridized on the same chip to determine the level of expression for the corresponding gene in K562 cells as well as the variability of probe hybridization on the E-chips (Figure1). Hybridization signals ranged from 20 to 53 395 fluorescent units (FU) in the Cy3 channel and 20 to 50 560 FU in the Cy5 channel. Although the variation in intensity was slightly greater among the more intensely hybridized spots, a narrow distribution was achieved at all levels (Figure 1B). On the basis of this distribution, we determined that Cy3/Cy5 ratios of less than 0.85 or greater than 1.43 represent a difference of more than 2 SDs in the level of hybridization, as shown in Figure 1C. Those ratios were used in all subsequent experiments to determine the significance of differences between bone marrow–derived and K562 cell–derived probe hybridization.
Quality control of E-chip.
(A) Image of a laser-scanned microarray hybridized with 50 μg Cy3- and Cy5-labeled probe from the same pool of K562 erythroleukemia cell total RNA. (B) Scatter plot of signal intensity data collected from NBM (Cy3, 550-nm) scan and K562 (Cy5, 650-nm) scan with 3 SDs. (C) Density plot of NBM (Cy3) signal intensity over K562 (Cy5) signal intensity. The ratio limits (Cy3/Cy5 ratios of less than 0.85 or greater than 1.43) used to calculate significant changes in expression levels indicate P ≤ .01.
Quality control of E-chip.
(A) Image of a laser-scanned microarray hybridized with 50 μg Cy3- and Cy5-labeled probe from the same pool of K562 erythroleukemia cell total RNA. (B) Scatter plot of signal intensity data collected from NBM (Cy3, 550-nm) scan and K562 (Cy5, 650-nm) scan with 3 SDs. (C) Density plot of NBM (Cy3) signal intensity over K562 (Cy5) signal intensity. The ratio limits (Cy3/Cy5 ratios of less than 0.85 or greater than 1.43) used to calculate significant changes in expression levels indicate P ≤ .01.
E-chip analyses of normal bone marrow
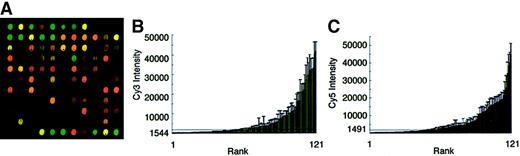
Total RNA was obtained from 10 normal bone marrow (NBM) aspirates from healthy donors and labeled as first-strand cDNA with Cy3. The labeled NBM probe was then cohybridized with Cy5-labeled probe from the common K562 cell pool. One quadrant from a representative E-chip is shown in Figure 2A. A broad range of hybridization intensities was seen. Differential hybridization of the cohybridized probes was noted on many spots, with green spots representing a higher level of gene expression in bone marrow and red spots representing a relatively higher level of expression in K562 cells. Spots fluorescing at equivalent levels in both Cy3 and Cy5 wavelengths appear yellow. Several spots revealed levels of hybridization at or below those of the blank and empty vector controls.
Hybridization intensity ranking.
(A) Image of a laser-scanned microarray hybridized with 50 μg Cy3-labeled NBM total RNA and 50 μg Cy5-labeled K562 erythroleukemia cell (K562) total RNA. (B) Cy3-labeled NBM signal intensity ranking. (C) Cy5-labeled K562 erythroleukemia cell signal intensity ranking. The horizontal bars at (B) 1544 and (C) 1491 fluorescent units denote the level above which signal intensity could be reliably detected above that of the negative controls.
Hybridization intensity ranking.
(A) Image of a laser-scanned microarray hybridized with 50 μg Cy3-labeled NBM total RNA and 50 μg Cy5-labeled K562 erythroleukemia cell (K562) total RNA. (B) Cy3-labeled NBM signal intensity ranking. (C) Cy5-labeled K562 erythroleukemia cell signal intensity ranking. The horizontal bars at (B) 1544 and (C) 1491 fluorescent units denote the level above which signal intensity could be reliably detected above that of the negative controls.
To compare the relative levels of gene activity among bone marrow and K562 cells, we ranked the hybridization intensity for each probe (Figure 2B,C). Each of the bars depicts the mean intensity and SD from a total of 40 spots (10 chips × 4 spots for each array position). Despite the marked differences in gene expression levels that resulted in green versus red fluorescence for individual spots (Figure 2A), the overall distribution of hybridization intensities for NBM and K562 probes was remarkably similar, ranging from 152 FU to 41 760 FU in NBM and 56 FU to 44 785 FU in K562. The lower limit of fluorescence detection was determined from the negative controls (intensity of the blank array position + 2 SDs: Cy3 = 1544 FU; Cy5 = 1491 FU). Hybridization below the lower limits was measured for 57 NBM-probed and 50 K562-probed spots. Low-intensity hybridization for both probes was detected in 35 spotted genes. Notably, the majority of spots encoding “novel” erythroid transcripts fell below the detectable limit. The median intensity for the NBM (Cy3) probes was 1684 ± 667 FU; the median intensity for K562 (Cy5) probes was 2445 ± 800 FU. The higher intensities associated with the K562 probes likely reflect the fact that the spotted genes were selected from a highly proliferative population of erythroid cells. In contrast, the similar levels of variation between the Cy3 and Cy5 signals were unexpected because the Cy3 probes were derived from 10 separate NBM RNA pools and the Cy5 probes were derived from a single K562 RNA pool. Duplicate preparations of RNA from the same marrow donor demonstrated levels of signal intensity variation equivalent to those shown in Figure 2.
To develop a clinically correlative pattern of gene expression for MDS bone marrow, we classified the spotted genes as proliferation associated or differentiation associated (Table 1). Forty-one of 106 spotted cDNAs could not be classified because of low (35 of 106) or equivalent-level (6 of 106) expression in bone marrow and K562 cells. Proliferation-associated genes were those expressed at significantly higher levels (at least 2 SDs) in the erythroleukemia cells than in bone marrow. Differentiation-associated genes were those expressed at significantly higher levels in bone marrow. This definition is based upon 2 assumptions: (1) The K562 erythroleukemia cells are not fully differentiated; and (2) the RNA derived from the undifferentiated cells in unpurified bone marrow is significantly diluted by that of more differentiated populations. Fifty-three of the 71 spots hybridized at detectable levels were scored as proliferation associated. About one third of the proliferation-associated genes identified here have been reported previously as leukemia associated or growth associated. With the exception of the γ- and ε-globin genes, other known erythroid-specific genes did not appear within the proliferation-associated group. Although smaller, the differentiation-associated group clearly represented genes associated with adult erythroid maturation. In addition to the adultglobin genes, carbonic anhydrase I, CD36, andc-fos have all been reported previously in the context of erythroid maturation. ERF-1, also calledbutyrate response factor-1, has not been previously associated with erythroid differentiation. The NBM/K562 ratios foractin and TCTP, although statistically significant, were similar to the low percentage of “false-positive” spots on the control K562/K562 array shown in Figure 1B.
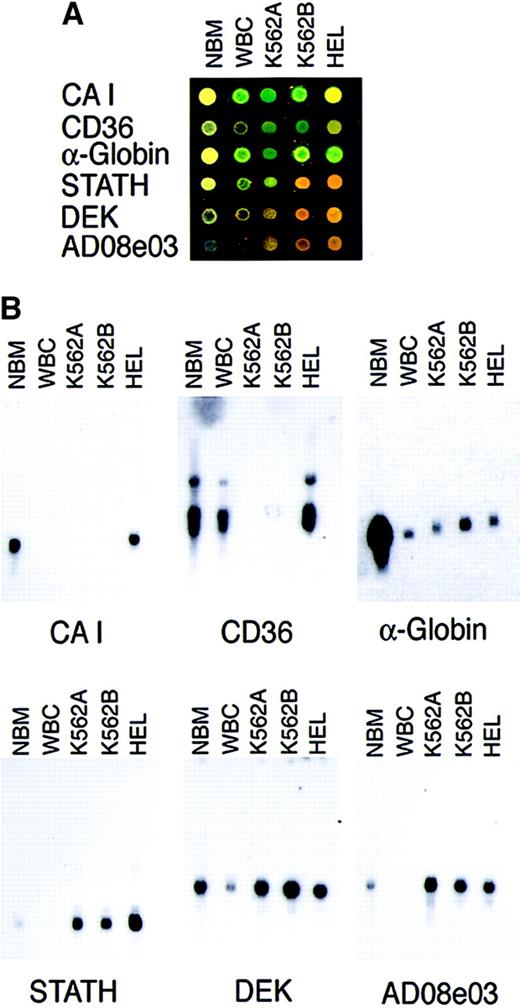
To experimentally confirm the expression patterns detected on the E-chips, we performed Northern blotting assays. We also compared patterns using RNA from K562 versus HEL erythroleukemia cell lines. Using arrays and Northern blots, probes were hybridized to mRNA from bone marrow, peripheral blood mononuclear cells, 2 sources of K562 cells, and the separate erythroleukemia cell line HEL. The results are shown in Figure 3. In each case, the E-chips correctly identified the pattern of gene expression demonstrated by Northern blotting. Notably, the levels of expression among the cell lines were variable, with CAI andCD36 expressed only among HEL cells. This suggests that erythroleukemia cell lines have considerable variation in their transcriptional phenotypes. However, those variations in the signal intensities between cell lines did not result in any spotted gene's being classified as proliferation associated in one cell line and differentiation associated in the other. Because the majority of the spotted genes fell into the same functional category using either HEL or K562 cells, we arbitrarily chose K562 cells as the reference cell line for all subsequent comparisons. A common pool of K562B RNA was used to prepare probe for all of the E-chips reported in this study to avoid confounding data created by passage-dependent changes in the reference RNA.
Cell line comparisons and Northern blot confirmation analyses.
(A) Array-based transcription patterns for bone marrow (Cy3) versus Cy5-labeled bone marrow (NBM), peripheral blood mononuclear cells (WBC), K562 erythroleukemia cells from 2 sources (K562A, K562B), and the HEL erythroleukemia cell line. K562B RNA was the source used for all subsequent E-chip hybridizations in this study. (B) Northern blots probed for representative proliferation- and differentiation-associated genes. Each lane contained 2.5 μg mRNA from carbonic anhydrase I (CAI), CD36, Stathmin (STATH), DEKoncogene (DEK), and hembase clone Ad08c04 (Ad08c04).
Cell line comparisons and Northern blot confirmation analyses.
(A) Array-based transcription patterns for bone marrow (Cy3) versus Cy5-labeled bone marrow (NBM), peripheral blood mononuclear cells (WBC), K562 erythroleukemia cells from 2 sources (K562A, K562B), and the HEL erythroleukemia cell line. K562B RNA was the source used for all subsequent E-chip hybridizations in this study. (B) Northern blots probed for representative proliferation- and differentiation-associated genes. Each lane contained 2.5 μg mRNA from carbonic anhydrase I (CAI), CD36, Stathmin (STATH), DEKoncogene (DEK), and hembase clone Ad08c04 (Ad08c04).
E-chip analysis of myelodysplastic bone marrow
Five different subtypes of MDS were studied (Table2). Bone marrow aspirates were obtained from patients with ages ranging from 33 to 76 years. Bone marrow myeloblasts ranged from less than 1% in MDS3 to 26% in MDS1. The MDS2 patient presented 4 years after chemotherapy with severe anemia and thrombocytopenia with a hypercellular, dysplastic bone marrow. The diagnosis of refractory anemia with excess blasts in transformation (RAEB-T) was based upon 5% blasts in the peripheral blood despite only 4% identified in the marrow. MDS3 was the only marrow in which there were prominent abnormal cytogenetics with trisomy 19, an inversion in chromosome 12, and a deletion in chromosome 21. That patient had 80% ringed sideroblasts in the marrow. In contrast, the MDS4 patient had severe thrombocytopenia and dysplasia with 7% blasts in the bone marrow, but little involvement of the erythroid lineage. MDS4 was the only patient in the study who was not transfusion dependent. MDS5 was selected because of marrow hypocellularity.
Clinical characteristics of the patients
| Characteristic . | MDS1 . | MDS2 . | MDS3 . | MDS4 . | MDS5 . |
|---|---|---|---|---|---|
| FAB type | RAEB-T | RAEB-T* | RARS | RAEB | RA |
| IPSS | High | Int-1 | Int-1 | Int-1 | Low |
| Age, sex | 68, M | 33, F | 72, M | 76, M | 59, M |
| Hb (g/dL) | 7.5 | 9.6 | 10.7 | 13† | 9.5 |
| Cellularity | Hyper | Hyper | Hyper | Hyper | Hypo |
| M:E ratio | 10:1 | 4:1 | 4:1 | 4:1 | 3:1 |
| Cytogenetics | Normal | Normal | Complex | Normal | Normal |
| Characteristic . | MDS1 . | MDS2 . | MDS3 . | MDS4 . | MDS5 . |
|---|---|---|---|---|---|
| FAB type | RAEB-T | RAEB-T* | RARS | RAEB | RA |
| IPSS | High | Int-1 | Int-1 | Int-1 | Low |
| Age, sex | 68, M | 33, F | 72, M | 76, M | 59, M |
| Hb (g/dL) | 7.5 | 9.6 | 10.7 | 13† | 9.5 |
| Cellularity | Hyper | Hyper | Hyper | Hyper | Hypo |
| M:E ratio | 10:1 | 4:1 | 4:1 | 4:1 | 3:1 |
| Cytogenetics | Normal | Normal | Complex | Normal | Normal |
MDS indicates myelodysplastic syndrome; FAB, French-American-British criteria7,26; RAEB-T, refractory anemia with excess blasts in transformation; RARS, refractory anemia with ringed sideroblasts; RAEB, refractory anemia with excess blasts; RA, refractory anemia; IPSS, International Prognostic Scoring System8; Hb, hemoglobin; M:E ratio, ratio of myeloid and erythroid elements within the marrow aspirate.
Patient MDS2 had been previously treated (MOPP × 8 cycles) during a period 4 years before the diagnosis of MDS.
Without transfusion.
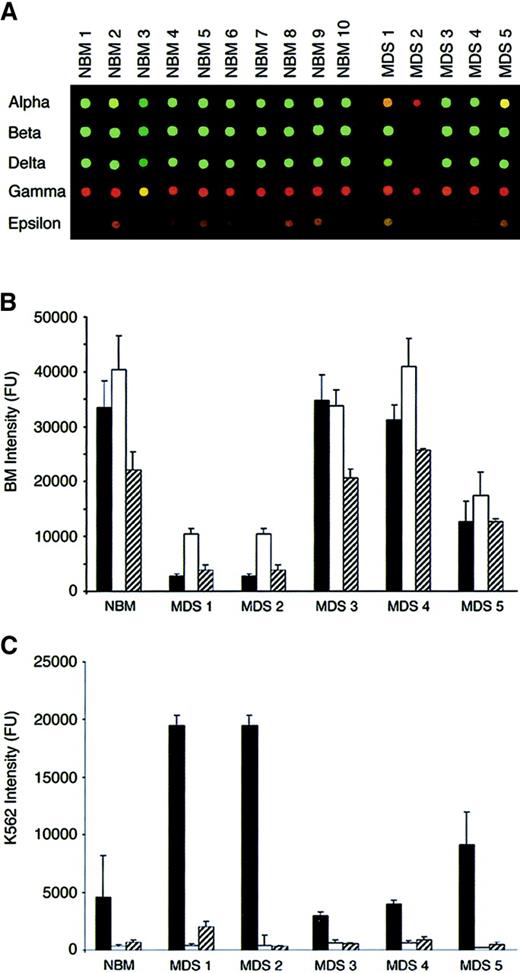
Because the levels of globin gene expression in bone marrow and K562 cells are known,14 those genes were used to examine and validate the array-based expression patterns (Figure4). As expected, the adult chains (α, β, and δ) were expressed at significantly higher levels in normal bone marrow (green spots), and the ε and γ genes were expressed at higher levels in K562 cells (red spots). Interestingly, NBM3 had a higher level of γ-globin gene expression than the other NBM as reflected by the yellow spot. Although abnormalities are predicted visually in 3 of 5 MDS aspirates (MDS1, MDS2, and MDS5), visual inspection alone is suboptimal for interpreting those abnormalities. To determine the basis of the abnormal MDS hybridization patterns, we examined the Cy3 and Cy5 hybridization intensities separately for each spotted gene (Figure 4B,C). The mean Cy3 intensities revealed a balanced pattern of globin gene expression in the bone marrow–derived samples (β ≥ α > δ), with significant diminution of those signals on the MDS1, MDS2, and MDS5 E-chips as compared with NBM. In contrast, the K562 cell–derived pattern of adult globin gene expression demonstrated a distinct pattern of intensities (α > δ > β) because β-globin is not transcribed in those cells. However, the K562 (Cy5) analysis also clearly revealed that significantly increased α-globin signals correlated directly with decreased Cy3 α-globin signals (compare Cy3 and Cy5 intensities for MDS1, MDS2, and MDS5 chips). Because the same pool of reference K562 RNA was used on all the E-chips, this suggests that Cy3- and Cy5-labeled probes competed for hybridization on the α-globin spots. This “competitive” pattern was also noted for other genes expressed at relatively high levels in test (bone marrow) and reference (K562 cell) samples (Tables 3and 4). Thus, probe competition as well as the variability in Cy5 intensities derived from the same pool of K562 RNA (Figure 2C) confounded the statistical interpretation of ratio-derived, interchip comparisons. Therefore, a direct statistical comparison of bone marrow–derived hybridization intensities (Cy3) in the absence of concordant differences in reference intensities (Cy5) was used to determine the significance of interchip differences in probe hybridizations.
Globin gene expression patterns.
(A) α, β, δ, γ, and ε globin spots probed with Cy3-labeled probe from 10 normal bone marrow (NBM) and 5 MDS bone marrow (MDS) samples. In all cases, the Cy5-labeled probe was generated from the same pool of K562 erythroleukemia cell RNA. Graphic displays of the Cy3 (bone marrow–derived) and Cy5 (K562-derived) intensities for the adult globin genes are shown in panels B and C, respectively. The normal bone marrow (NBM)-derived and myelodysplastic bone marrow (MDS)-derived signal intensities are represented as means with SD bars. Key: α (solid bars), β (open bars), δ (hashed bars).
Globin gene expression patterns.
(A) α, β, δ, γ, and ε globin spots probed with Cy3-labeled probe from 10 normal bone marrow (NBM) and 5 MDS bone marrow (MDS) samples. In all cases, the Cy5-labeled probe was generated from the same pool of K562 erythroleukemia cell RNA. Graphic displays of the Cy3 (bone marrow–derived) and Cy5 (K562-derived) intensities for the adult globin genes are shown in panels B and C, respectively. The normal bone marrow (NBM)-derived and myelodysplastic bone marrow (MDS)-derived signal intensities are represented as means with SD bars. Key: α (solid bars), β (open bars), δ (hashed bars).
Abnormal gene expression pattern detected in MDS: proliferation associated
| Description . | MDS1 fold change . | MDS2 fold change . | MDS3 fold change . | MDS4 fold change . | MDS5 fold change . |
|---|---|---|---|---|---|
| c-myb | 1.95 | — | — | 2.26 | — |
| p23 | — | 2.93 | −2.67 | — | — |
| HMG-14 | — | 1.39 | −1.68 | — | — |
| Ad14g01* | — | 2.46 | — | — | — |
| α-NAC | — | 2.75 | — | — | — |
| APRT | — | 2.14 | — | — | — |
| dbpB-like protein | — | 2.16 | — | — | — |
| EF-1α* | — | 2.25 | — | — | — |
| Ferritin L-chain | — | 1.76 | — | — | — |
| hnRNP D | — | 1.67 | — | — | — |
| LCAT | — | 3.21 | — | — | — |
| Mus ubc4 | — | 2.63 | — | — | — |
| NADH B-12 | — | 1.67 | — | — | — |
| nm23-H2 | — | 1.84 | — | — | — |
| Novel ad13f05 | — | 1.91 | — | — | — |
| TOM20 | — | 2.22 | — | — | — |
| Transferrin receptor | −7.21 | — | — | — | — |
| Xp-C protein | — | 1.65 | — | — | — |
| Description . | MDS1 fold change . | MDS2 fold change . | MDS3 fold change . | MDS4 fold change . | MDS5 fold change . |
|---|---|---|---|---|---|
| c-myb | 1.95 | — | — | 2.26 | — |
| p23 | — | 2.93 | −2.67 | — | — |
| HMG-14 | — | 1.39 | −1.68 | — | — |
| Ad14g01* | — | 2.46 | — | — | — |
| α-NAC | — | 2.75 | — | — | — |
| APRT | — | 2.14 | — | — | — |
| dbpB-like protein | — | 2.16 | — | — | — |
| EF-1α* | — | 2.25 | — | — | — |
| Ferritin L-chain | — | 1.76 | — | — | — |
| hnRNP D | — | 1.67 | — | — | — |
| LCAT | — | 3.21 | — | — | — |
| Mus ubc4 | — | 2.63 | — | — | — |
| NADH B-12 | — | 1.67 | — | — | — |
| nm23-H2 | — | 1.84 | — | — | — |
| Novel ad13f05 | — | 1.91 | — | — | — |
| TOM20 | — | 2.22 | — | — | — |
| Transferrin receptor | −7.21 | — | — | — | — |
| Xp-C protein | — | 1.65 | — | — | — |
Abnormal gene expression pattern in proliferation-associated category. Average MDS (Cy3) signal intensities for each gene (spotted 4 times per MDS chip) were compared with the mean NBM (Cy3) signal intensities from 40 spots (10 NBM chips × 4 spots per chip). The fold change in MDS mean Cy3 intensity compared with NBM Cy3 intensity for each chip is shown. Abnormal gene expression is defined as a mean MDS Cy3 intensity at least 2 SDs from the mean of the corresponding mean NBM Cy3 intensity.
Genes that demonstrated a significant shift in the K562 signal intensity that correlated with shifts in MDS-probed intensities (K562 and MDS shifts in opposite directions) like those demonstrated in Figure 4. Other MDS interchip variations in K562 signal intensities were not statistically significant.
Abnormal gene expression pattern detected in MDS: differentiation associated
| Description . | MDS1 fold change . | MDS2 fold change . | MDS3 fold change . | MDS4 fold change . | MDS5 fold change . |
|---|---|---|---|---|---|
| α-globin3-150 | −10.8 | −100 | — | — | −2.57 |
| β-globin | −3.86 | −89.3 | — | — | −2.32 |
| δ-globin3-150 | −5.34 | −25.5 | — | — | −2.11 |
| CAI | −27.01 | −103 | — | — | −5.84 |
| Ad08c04 | −2.00 | −3.99 | — | — | −1.65 |
| CD36 | −7.95 | — | — | — | −4.32 |
| Actin | — | 1.72 | −1.49 | — | — |
| ERF-1 | — | 3.88 | — | — | — |
| TCTP3-150 | — | 1.75 | — | — | — |
| Description . | MDS1 fold change . | MDS2 fold change . | MDS3 fold change . | MDS4 fold change . | MDS5 fold change . |
|---|---|---|---|---|---|
| α-globin3-150 | −10.8 | −100 | — | — | −2.57 |
| β-globin | −3.86 | −89.3 | — | — | −2.32 |
| δ-globin3-150 | −5.34 | −25.5 | — | — | −2.11 |
| CAI | −27.01 | −103 | — | — | −5.84 |
| Ad08c04 | −2.00 | −3.99 | — | — | −1.65 |
| CD36 | −7.95 | — | — | — | −4.32 |
| Actin | — | 1.72 | −1.49 | — | — |
| ERF-1 | — | 3.88 | — | — | — |
| TCTP3-150 | — | 1.75 | — | — | — |
Abnormal gene expression pattern in differentiation-associated category. Average MDS (Cy3) signal intensities for each gene (spotted 4 times per MDS chip) were compared with the mean NBM (Cy3) signal intensities from 40 spots (10 NBM chips × 4 spots per chip). The fold change in MDS mean Cy3 intensity compared with NBM Cy3 intensity for each chip is shown. Abnormal gene expression is defined as a mean MDS Cy3 intensity at least 2 SDs from the mean of the corresponding mean NBM Cy3 intensity.
Genes that demonstrated a significant shift in the K562 signal intensity that correlated with shifts in MDS-probed intensities (K562 and MDS shifts in opposite directions) like those demonstrated in Figure 4. Other MDS interchip variations in K562 signal intensities were not statistically significant.
Only those genes demonstrating statistically significant (P < .01) changes in MDS- versus NBM-derived hybridization among the MDS samples are shown in Tables 3 and 4. The fold changes in the MDS-derived intensities relative to the mean intensity in NBM ranged from a 100-fold reduction for the α-globin gene to a 2.9-fold increase for the p23 gene. Significant reductions (at least 2 SDs) in the same 5 genes (the adult globins, CAI, and a previously uncharacterized transcriptAd08c04) were noted in 3 of 5 MDS marrow aspirates. MDS4 (RAEB without transfusion requirements) demonstrated only one abnormal spot (increased c-myb). Despite the complex cytogenetics in the transfusion-dependent patient with refractory anemia with ringed sideroblasts (RARS; MDS3), the E-chip provided no evidence of reduced differentiation and no evidence of increased proliferation. In contrast, MDS2 (secondary MDS in transformation) had the most abnormal pattern, with a significant increase in 16 proliferation-associated genes. Overall, 18 of 53 proliferation-associated genes and 9 of 12 differentiation-associated genes were abnormally expressed in at least one MDS marrow. No significant change was identified in any of the genes categorized as “undefined” (Table5). When grouped, 21 proliferation-associated and 21 differentiation-associated abnormalities were identified among the 5 MDS aspirates. The levels among 18 of the 21 differentiation-associated abnormalities were reduced in the setting of MDS. Conversely, the levels of 18 of the 21 proliferation-associated abnormalities were increased. All of the increased proliferation-associated abnormalities were found in the RAEB or RAEB-T marrow samples.
Abnormal gene expression pattern detected in MDS: summary
| Category . | Proliferation associated . | Differentiation associated . | No defined association . |
|---|---|---|---|
| Up-regulated | 18 | 3 | 0 |
| Down-regulated | 3 | 18 | 0 |
| Total | 21 | 21 | 0 |
| Category . | Proliferation associated . | Differentiation associated . | No defined association . |
|---|---|---|---|
| Up-regulated | 18 | 3 | 0 |
| Down-regulated | 3 | 18 | 0 |
| Total | 21 | 21 | 0 |
Discussion
In this pilot study, we demonstrate patterns of normal and dysplasia-related transcription among unpurified bone marrow mononuclear cells. The patterns were derived from a subset of genes transcribed in highly proliferative, primary human erythroid cells.12 Those genes were identified as part of a separate effort to catalog the transcriptional phenotype of human erythroid cells. Several thousand transcripts have been sequenced from those cells, but the interpretation of those genetic data in vivo is far from complete. A full understanding of erythropoiesis will require that transcription patterns be integrated, quantified, developmentally staged, and compared. Our comparisons of bone marrow and erythroleukemia gene expression patterns (Figure 2) represent an initial attempt to move in this direction. In addition, to provide a transcriptional assessment aimed at a broader range of hematologic disorders, gene activity must be assigned for each lineage. The list of hematopoietic cells for which a transcriptional profile has been reported currently includes lymphocytes,15CD34+ cells,16,17 and dendritic cells.18 Our goal is the generation of transcription-based profiles equivalent to the morphology-based, complete blood counts used today.
Our ability to generate relevant gene transcription patterns from heterogeneous marrow cell populations suggests that this technology may be usefully applied to complex malignant cell populations. Here we demonstrate that the steady state of transcription normally found in bone marrow may be used to define abnormal transcription patterns in MDS. In this group of disorders, the predicted pattern of increased proliferation and decreased differentiation among a heterogeneous population of hematopoietic cells was identified. Additionally, patient-specific patterns were identified. The patient with RAEB, with minimal evidence of abnormal erythroid gene activity, also had the least evidence of ineffective erythropoiesis (untransfused hemoglobin of 13 g/dL). Predictably, in accordance with its high predisposition to transform to acute leukemia, only secondary MDS had a markedly abnormal pattern of increased proliferation. Among the proliferation-associated genes, c-myb was the only gene with abnormally increased expression detected in more than one patient. That gene has long been implicated in maintaining proliferation and subsequent oncogenesis.19 In contrast, a group of differentiation-associated genes was consistently detected at abnormally low levels in MDS (adult globins, CAI, CD36, and the uncharacterized Ad08c04 transcript). Surprisingly, a significant decrease in the transcription of this group of genes was not detected in the patient with transfusion-dependent RARS. Perhaps the involved genes were not well represented or the mechanisms involved are not well suited for detection at the transcriptional level. Serial analyses and larger groups of patients must be formally examined to determine whether consistent and reproducible gene expression patterns exist for individual patients or MDS subtypes.
Despite the capability of cDNA arrays to identify clinically correlative patterns of gene transcription,20 significant differences exist between array-based patterns and absolute quantitation of transcription. The apparent competition between reference and test probes for coexpressed genes (α-globin gene used as an example in this study; Figure 4) supports the conclusion that array patterns represent biologic correlates rather than quantitative standards upon which to define biology. The existence of probe competition may also have an impact on the interpretation of ratio-based, interchip comparisons. Dual-probe fluorescence-based assays provide relative, not absolute, quantitation of gene activity. Thus, some degree of awareness and caution is required when interpreting quantitative aspects of analyses from a single chip or a series of chips probed with the same reference sample. Costs, standards,21 reproducibility, and the stability of RNA in clinical specimens should also be factored in to determine the most appropriate clinical translation of effort.
We conclude that transcription patterning as demonstrated here may be appropriate for larger studies of MDS aimed at correlating genetic and clinical findings. Assuming this technology is reliable for comparing samples collected over broader periods of time, prospective examination of MDS disease progression and therapeutic response will be particularly important to determine its value in clinical practice. In planning such studies, several factors including the choice of spotted cDNA must be considered. Ironically, the growth of genetic information in recent years already permits the accumulation of more patient-relevant data than are easily interpretable. Our preselection of erythroid-relevant genes resulted in a high percentage that correlated with disease activity. Therefore, we propose that the clinical application of array technology may be practical with a relatively small number of disease-focused genes. This will require the removal of uninformative genes, such as those expressed at very low levels, as well as the inclusion of new genes derived from screening arrays composed of several thousand spots. Known MDS-related genes such as telomerase,22nm23-H1,23 cytokines and their receptors,24 and apoptosis-related transcripts25 should also be tested. Arrays could also be designed to combine disease loci mapped from MDS cytogenetics with those loci normally active in hematopoietic cells as a means of determining cause-and-effect relationships for the disease. Notwithstanding their complexity, high-throughput technologies will greatly facilitate the genetic description of complex diseases such as MDS during the coming era of genomics-based hematology research.
We thank Jennifer Sqalari for technical assistance in preparing the reference RNA. Dr Kevin Shannon was helpful in the selection of MDS bone marrow and many discussions. We also thank Drs Alan and Geraldine Schechter for critical reading of the manuscript.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Jeffery L. Miller, Laboratory of Chemical Biology, Bldg 10, Rm 9B17, National Institutes of Health, Bethesda, MD 20892; e-mail: jm7f@nih.gov.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal