Abstract
Recent studies suggest that multivesicular bodies are an intermediate stage in the formation of -granules. In contrast, the kinetics and mode of appearance of dense granules during megakaryocytic maturation has remained poorly understood. Immunoelectron microscopy was used to monitor the appearance of dense granular markers (granulophysin and serotonin) on cryosections of human megakaryocytes (MKs) cultured from CD34+ precursors. The monitoring was done on days 8 and 13 of culture. The data suggest that dense granules appear in immature MKs early during their maturation, concomitantly with -granule formation. In MKs of intermediary maturation stage, granulophysin was mainly localized within dense granules and multivesicular bodies (MVBs), which were also labeled for serotonin. This study provides evidence that granulophysin is a dense granule marker in human MKs and that MVBs are an intermediary stage of dense granule maturation and probably constitute a sorting compartment between -granules and dense granules.
Dense granules belong to the secretory compartment that is released during platelet activation. They contain a variety of important hemostatically active substances1 including matrix proteins that are released into the extracellular medium upon activation (eg, serotonin, catecholamines, adenosine 5′-diphosphate [ADP], adenosine 5′-triphosphate [ATP], and calcium) and integral components2 of the granule limiting membrane (eg, αIIbβ3; glycoprotein Ib [GPIb]; P-selectin2,3; and granulophysin,2,4,5 a specific dense granule marker). However, the biogenesis of dense granules in the platelet precursor, the megakaryocyte (MK), is poorly understood. Recently Heijnen et al6 have shown that multivesicular bodies (MVBs) represent a developmental stage in α-granule maturation. Some receptors, such as αIIbβ3, GPIb, and P-selectin, have been identified in the limiting membrane in both α-granules and dense granules.2-4,7-9 In addition, these granules share remarkable similarities in their protein trafficking pathways, with both endogenous synthesis and endocytosis from the extracellular medium.10-13 In this study we investigated the formation of dense granules during MK maturation on human culture MKs via immunoelectron microscopy. Our data indicate that MVBs are an intermediary compartment where α-granule and dense granule components are sorted.
Study design
Cells
Human MKs were grown in liquid culture from bone marrow precursors obtained from normal adult graft donors as previously described.8
Antibodies
We used the following rabbit polyclonal antibodies (pAbs) at 10 μg/mL: antiglycocalicin, an antibody to GPIb9,14 (gift from Dr Michael Berndt, Victoria, Australia); antiserotonin (Sigma, St Louis, MO) and anti–von Willebrand factor (Dako, Glostrup, Denmark). Antigranulophysin,2 a monoclonal antibody (mAb) (gift from Dr Israels, University of Manitoba, Winnipeg, Manitoba, Canada) was also used at 10 μg/mL. Goat antirabbit (GAR) and antimouse (GAM) immunoglobulin G (IgG) fractions were coupled to 5-nm or 10-nm gold particles (British Biocell International, Cardiff, England).
Electron microscopy
MKs were prepared for immunoelectron microscopy as previously described.3 Briefly, they were fixed in 1% glutaraldehyde, embedded in sucrose, and frozen in liquid nitrogen.15Simple and double immunogold labeling was performed on ultrathin cryosections.3 In parallel, control sections were incubated with gold conjugated antibodies after omitting the primary antibodies, which displayed no labeling. Samples were observed on a Philips CM10 electron microscope (Philips, Eindhoven, The Netherlands).
Results and discussion
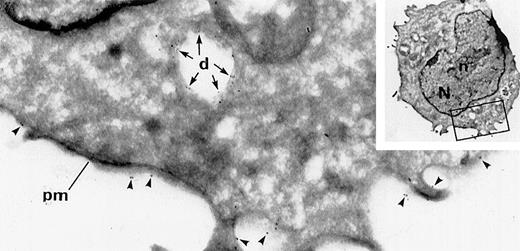
We documented the time of appearance of dense granules during MK maturation using immunolabeling experiments on frozen thin sections of human MKs at day 8 of culture. Double immunolabeling for GpIb (a specific MK marker) and granulophysin (a specific dense granule marker) facilitated the detection of dense granules within immature MKs. Some poorly differentiated blastic cells deprived of specific organelles were identified as immature MKs because they displayed the classical membrane distribution for GPIb. However, consistent labeling for granulophysin was observed within electron luscent granules and vacuolar-like structures similar to the classical dense granular structure (Figure1). One of the problems during preparation of cryosections is that the content of dense granules often becomes extracted, and thus the dense granules appear as vacuolar organelles.2,3 Because of difficulty visualizing dense granules, the time and mode of their appearance during MK maturation has remained poorly understood. Previous investigations had shown the presence of dense granules in MKs by using mepacrine labeling and uranaffin cytochemical reactions, but only mature MKs had been investigated.16 Our present experiments have focused upon the appearance of dense granules in the early stage of MK maturation.
Detection of dense granules in immature MKs at day 8 of culture by double labeling for GPIb and granulophysin.
Arrowheads indicate GPIb (15-nm gold particles), and arrows indicate granulophysin (10-nm gold particles). This immature MK, seen on a frozen thin section, displays a nucleus (N) with abundant euchromatin and a prominent nucleolus (n) and appears morphologically undifferentiated. It can only be identified by GPIb-specific expression on its plasma membrane (pm). Granulophysin labels the membrane of vacuolar structures, which can be identified as dense granules (d) because of the presence of this specific marker (original magnification ×41 800; inset, ×3920).
Detection of dense granules in immature MKs at day 8 of culture by double labeling for GPIb and granulophysin.
Arrowheads indicate GPIb (15-nm gold particles), and arrows indicate granulophysin (10-nm gold particles). This immature MK, seen on a frozen thin section, displays a nucleus (N) with abundant euchromatin and a prominent nucleolus (n) and appears morphologically undifferentiated. It can only be identified by GPIb-specific expression on its plasma membrane (pm). Granulophysin labels the membrane of vacuolar structures, which can be identified as dense granules (d) because of the presence of this specific marker (original magnification ×41 800; inset, ×3920).
To document the intracellular pathway of dense granule formation, double-labeling experiments were performed in mature MKs at day 13 of culture (Figure 2A) using a mixture of antigranulophysin mAb and antiserotonin pAb. Serotonin and granulophysin were detected in some vacuolar structures, with or without a dense content, thereby confirming the identification of these organelles as dense granules (Figure 2B). Granulophysin appeared to be located along the limiting membrane of these organelles as well as around the dense vesicles which often constituted their dense core. This result is consistent with the previous observation of serotonin uptake in the MK precursors, the megakaryoblasts.1 Both markers were also found in MVBs (Figure 2C), and granulophysin was mainly located on the membrane of their internal vesicles. Simple labeling for granulophysin showed that 17% of labeling was associated with dense granule membranes of mature Mks (Figure 2D). Approximately 80% of the labeling was found in the internal vesicles of MVBs or with the membrane of some intermediary MVBs that had lost part of their vesicle structure, and less than 3% of the labeling was associated with α-granules (Figure 2D, E). In addition, the granulophysin containing α-granules was often immature, with a large size and odd shape, and was localized in the vicinity of MVBs, suggesting that they may originate from MVBs, as described by Heijnen et al.6Mature α-granules with a dense nucleoid were deprived of any labeling.
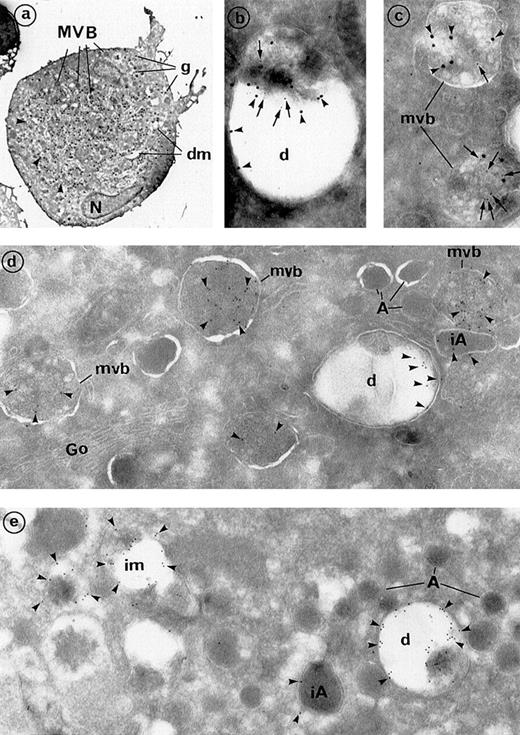
Frozen sections of maturing MKs, immunolabeled for the detection of dense granule components.
(A) Frozen culture MKs can be identified by their large size, indented nucleus (N), numerous secretory granules (g), and prominent demarcation membrane system (dm). Numerous organelles (about 40), which were identified as MVBs by electron microscopy at a higher magnification, are present in this section. Arrowheads indicate dense granules (at least 5), which were morphologically identifiable (original magnification ×2640). (B, C) Double labeling for the 2 dense granule markers is depicted. Arrowheads indicate granulophysin (20-nm gold particles), and arrows indicate serotonin (10-nm gold particles). (B) Serotonin is detected within the (C) MVBs and (D) dense granules. Both structures are also labeled for granulophysin (original magnification ×49 450). (D, E) Simple immunogold labeling for granulophysin. (D) Granulophysin (arrowheads) labels the dense granule limiting membranes (d), which appear like vacuolar structures that represent 17% of total labeling. Internal vesicles of MVBs represented 80% of the total labeling. Minimal labeling (approximately 3%) was associated with immature α-granules of large size and odd shape (iA), whereas mature α-granules (A) were deprived of labeling. Go indicates the Golgi complex (original magnification ×46 300). (E) Granulophysin in mature MKs is mainly found within the dense granules (d). Labeling is also present on some intermediate MVBs (im), which have lost part of their vesicles and have an ultrastructure similar to that of dense granules (original magnification ×46 300).
Frozen sections of maturing MKs, immunolabeled for the detection of dense granule components.
(A) Frozen culture MKs can be identified by their large size, indented nucleus (N), numerous secretory granules (g), and prominent demarcation membrane system (dm). Numerous organelles (about 40), which were identified as MVBs by electron microscopy at a higher magnification, are present in this section. Arrowheads indicate dense granules (at least 5), which were morphologically identifiable (original magnification ×2640). (B, C) Double labeling for the 2 dense granule markers is depicted. Arrowheads indicate granulophysin (20-nm gold particles), and arrows indicate serotonin (10-nm gold particles). (B) Serotonin is detected within the (C) MVBs and (D) dense granules. Both structures are also labeled for granulophysin (original magnification ×49 450). (D, E) Simple immunogold labeling for granulophysin. (D) Granulophysin (arrowheads) labels the dense granule limiting membranes (d), which appear like vacuolar structures that represent 17% of total labeling. Internal vesicles of MVBs represented 80% of the total labeling. Minimal labeling (approximately 3%) was associated with immature α-granules of large size and odd shape (iA), whereas mature α-granules (A) were deprived of labeling. Go indicates the Golgi complex (original magnification ×46 300). (E) Granulophysin in mature MKs is mainly found within the dense granules (d). Labeling is also present on some intermediate MVBs (im), which have lost part of their vesicles and have an ultrastructure similar to that of dense granules (original magnification ×46 300).
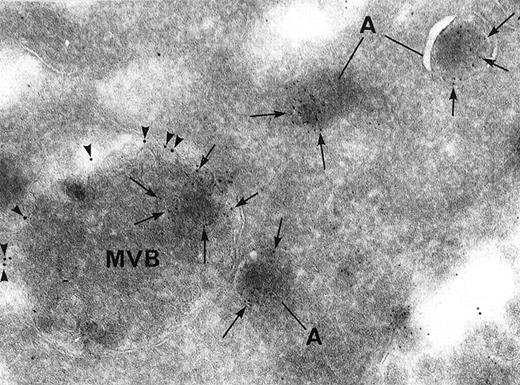
Finally, double labeling experiments showed that both α-granule proteins (von Willebrand factor) and a dense granule marker (granulophysin) could be found in the same MVB. Moreover, immunolabeling for granulophysin was located in internal vesicles distinct from the ones containing von Willebrand factor within the same MVB. The immunolabeling was also often associated with the limiting membrane of MVBs, which suggests that the various granule constituents had started separating (Figure 3). Thus, our observations indicate that dense granule components are segregated from other secretion granules within the MVBs and that these eventually develop into dense granules.
Double immunolabeling for the dense granule marker granulophysin and the -granule marker von Willebrand factor.
Arrowheads indicate granulophysin (10-nm gold particles), and arrows indicate α-granule marker von Willebrand factor (5-nm gold particles). Both proteins can be found within the same MVB, but they are segregated from each other in distinct microvesicles. Granulophysin is also preferentially associated with the limiting membrane of MVBs. Von Willebrand factor is located in α-granules (A) as well as in MVB microvesicles (original magnification ×67 300).
Double immunolabeling for the dense granule marker granulophysin and the -granule marker von Willebrand factor.
Arrowheads indicate granulophysin (10-nm gold particles), and arrows indicate α-granule marker von Willebrand factor (5-nm gold particles). Both proteins can be found within the same MVB, but they are segregated from each other in distinct microvesicles. Granulophysin is also preferentially associated with the limiting membrane of MVBs. Von Willebrand factor is located in α-granules (A) as well as in MVB microvesicles (original magnification ×67 300).
The present study was undertaken because there is cumulating evidence demonstrating similarities between the 2 types of secretory granules, α-granules and dense granules. The plasma membrane receptor αIIbβ3 is a component of the α-granule membrane as well as the dense granule membrane.3,8 P-selectin is also specifically associated with the limiting membrane of both α-granules and dense granules.2,7 The respective composition of both granule types may reflect the dual mechanisms responsible for their protein trafficking: In addition to the classical endogenous MK synthesis,17 α-granules can also acquire their content by incorporation of circulating plasma components.10,11Similarly, dense granules are also able to take up serotonin from the circulating plasma, and this phenomenon applies to platelets and MKs.1,18 This close relationship is further evidenced by the existence of congenital platelet disorders, such as α-δ storage pool disease in humans and gunmetal mutation in mice,19 which involve the association of α-granule abnormalities with dense granule deficiency.20 In addition, it was demonstrated that MVBs are an intermediary stage in the formation of α-granules.6 In this study we show that dense granules follow a similar pathway.
Finally, it has also been demonstrated that through its specific receptors, serotonin accumulation also occurs in platelets. Intracellular trafficking of serotonin receptors may resemble that of granulophysin. First, these receptors would be present on the membrane of the internal vesicles located within MVBs; subsequently, during maturation, they are incorporated into the dense granule limiting membrane. Therefore, in platelets, serotonin accumulation may occur as a result of its specific receptors that are present in the dense granule membrane.
In conclusion, the present study provides evidence that dense granules are formed at the early stage of MK maturation. The study also indicates that MVBs are a sorting compartment for α-granule and dense granule formation, where granulophysin is directed into the dense granule compartment during MK maturation.
Acknowledgments
The authors gratefully acknowledge Dr Najet Debili for megakaryocyte cultures, Mme Josette Guichard for laboratory help, Mr Jean-Marc Massé for photographic work, Dr Paul Harrison and Mr Alain Schmitt for improving the manuscript, and Dr Paul-Henri Roméo for his constant support.
T.Y. is a fellowship recipient from L'Association Nationale pour la Recherche contre le SIDA.
Reprints:Elisabeth Cramer, INSERM U 474, Hôpital Cochin–Port-Royal, 123 Boulevard Port-Royal, 75014 Paris, France; e-mail: elisabeth.cramer@cochin.inserm.fr.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal