Abstract
Cutaneous B-cell infiltrates showing a prominent follicular growth pattern with germinal centers are thought by some authors to represent either marginal zone lymphomas with reactive germinal centers or pseudolymphomas. To establish whether a true primary cutaneous follicular lymphoma exists, we studied biopsies from 15 patients with skin lesions characterized histopathologically by the presence of B-cell infiltrates with follicular pattern. Staging investigations, including bone marrow biopsy, were negative in all patients. All were negative for bcl-2 protein expression and did not present the t(14;18). In all biopsy specimens neoplastic follicles showed 1 or more morphologic or immunophenotypic criteria of malignancy (presence of a reduced mantle zone, absence of tingible body macrophages, reduced proliferation rate). In 9 specimens a monoclonal rearrangement of JH genes could be detected by polymerase chain reaction analysis. After laser beam microdissection, a band of the same length could be observed in 6 probes from different follicles from the same specimen, indicating the presence of the same monoclonal population of follicle center cells. Follow-up examinations in all patients revealed no evidence of extracutaneous spread (mean follow-up, 48.7 months). Our study demonstrates that primary cutaneous follicular lymphoma represents a distinct entity of the cutaneous B-cell lymphomas.
In the classification for cutaneous lymphomas recently published by the European Organization for Research and Treatment of Cancer (EORTC)-Cutaneous Lymphoma Project Group, follicle center cell lymphoma (FCL) is considered the most common variant of the primary cutaneous B-cell lymphomas (PCBCL).1 This type of PCBCL usually is evident histopathologically with a diffuse growth pattern.2-7 The presence of a follicular pattern has been reported only in a few patients.8 On the other hand, the existence of a true cutaneous follicular lymphoma has been questioned by some authors, who suggest that cases of cutaneous lymphoid infiltrates showing a follicular pattern with prominent germinal centers represent in fact either marginal zone lymphoma–immunocytoma with reactive germinal centers or B-cell pseudolymphoma.9-11 The main reason the existence of a primary cutaneous follicular lymphoma is denied by these authors resides in the negativity for bcl-2 protein expression and the absence of the 14;18 interchromosomal translocation in these cases.12-14 In contrast, both these features are observed in most follicular lymphomas of the lymph nodes.15-18
In this article we present 15 cases of cutaneous follicle center lymphoma with prominent follicular growth pattern (follicle center lymphoma, follicular). To establish the true nature of these lesions, we analyzed clinicopathologic and molecular features of the infiltrates by various techniques, including clonality analysis after laser beam computerized microdissection.
Patients and methods
Patients
Fifteen patients with primary cutaneous B-cell lymphoma characterized by prominent follicular growth pattern were included in this study. Primary skin involvement was defined as the presence of cutaneous lymphoma without nodal or visceral involvement for a period of at least 6 months after complete staging procedures (including, for all patients, complete blood examination, computerized tomography of chest and abdomen, sonography of the superficial lymph nodes, and bone marrow biopsy) were performed. Patients with extracutaneous lymphomas with secondary skin manifestations were excluded from this study. Three patients with cutaneous marginal zone lymphoma (MZL) showing reactive germinal centers and 7 patients with cutaneous B-cell pseudolymphoma (lymphadenosis benigna cutis, lymphocytoma cutis), characterized by the presence of a benign B-cell infiltrate with reactive germinal centers, comprised the control group.
Histology
Biopsy specimens were fixed in 10% buffered formalin and subsequently embedded in paraffin. Sections were stained with hematoxylin-and-eosin and Giemsa stains for routine histopathologic evaluation. Cases were classified according to diagnostic criteria published in the Revised European-American Lymphoma (REAL) and EORTC classifications.1 19 All sections were reviewed by at least 2 independent dermatopathologists (L.C., H.K.).
Immunohistology
Detailed immunophenotyping was performed for both groups on routinely fixed, paraffin-embedded tissue sections according to a previously described 3-step immunoperoxidase technique20using a broad panel of monoclonal antibodies. Microwave enhancement was used for most of the antibodies. Second and third antibodies were obtained from Dakopatts (Glostrup, Denmark). Normal skin structures and tonsil tissues served as internal and external positive controls, respectively. Negative controls were performed by omitting the primary antibody or replacing it with normal human serum.
Laser beam microdissection
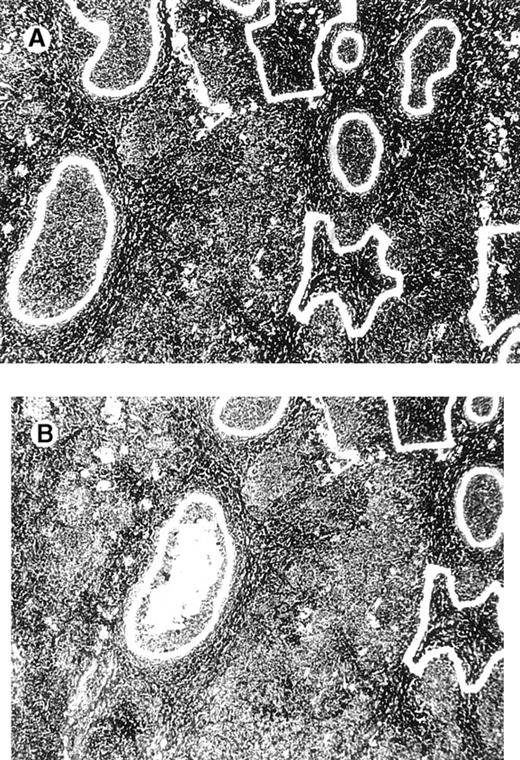
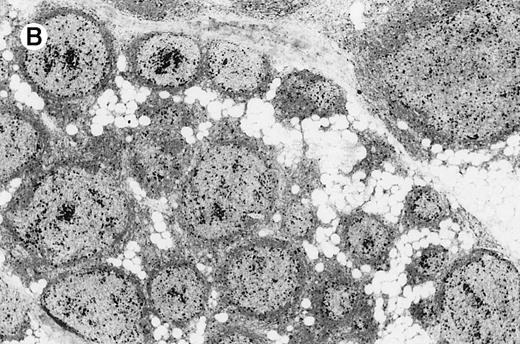
Laser beam microdissection was carried out in 12 patients with cutaneous follicular lymphoma, 3 patients with MZL, and 4 patients with B-cell pseudolymphoma. In all specimens analyzed, samples containing 60 to 100 cells each from at least 5 different follicles (or from all follicles if fewer than 5) and 5 different interfollicular areas from the same specimen were obtained for polymerase chain reaction (PCR) analysis. Microdissection was performed as follows: 4-μm sections cut from formalin-fixed, paraffin-embedded tissues were mounted on glass slides with a thickness of 0.17 mm (very thin glass slides are needed to prevent laser energy from being dispersed before reaching the section of tissue), deparaffinized, and stained with methyl green. An ultraviolet laser microscope system was then used to isolate particular populations of cells (UV-laser microbeam; PALM, Bernried, Germany). In short, a pulsed UV laser of high beam quality (nitrogen laser, wavelength 337 nm, maximum frequency 20 pulses/s, pulse duration 3 nsec) was combined with an inverse microscope and focused through an objective of high numerical aperture into the tissue plane. Beam spot diameter measured approximately 0.3 to 0.5 μm. Because of the extremely high energy density within the focal point (laser energy at object plane approximately 5 μJ), all biologic material is completely destroyed.21 Using the UV laser beam at a high repetition rate (approximately 20 pulses/s), a circle was cut around the target cells. This resulted in complete separation of the target population from neighboring tissues (Figure1). For this procedure the laser beam was fixed while the section was carefully moved to perform the cutting desired. Microscopic movements of tissue sections were made possible by the use of a motorized, computer-driven stage of the microscope. With the aid of a motorized micromanipulator and a thin injection needle (single-use injection needle; diameter, 0.8 mm; Henke–Sass–Wolf GmbH, Tuttlingen, Germany), the isolated cells were scraped from the coverslip and placed in a proteinase K solution. The rate of successful collection of cells for DNA analysis with this method was more than 90%. The computer-controlled stage and the micromanipulator had the same high resolution of 40 nm per microstep. Travel speed varied from less than 1 μm to several mm per second.
Computerized microdissection.
(A) Preparation of follicle center cells and interfollicular cells by laser beam for computerized microdissection. (B) Detail of the specimen after computerized microdissection of 1 follicle.
Computerized microdissection.
(A) Preparation of follicle center cells and interfollicular cells by laser beam for computerized microdissection. (B) Detail of the specimen after computerized microdissection of 1 follicle.
Molecular biology
Molecular analysis of the JH gene rearrangement was performed using a standard technique. Briefly, DNA was prepared from formalin-fixed, paraffin-embedded tissue specimens as described previously.22 The JH gene was analyzed using the semi-nested PCR technique described by Wan et al,23with minor modifications. For the first PCR, 20 μg template DNA was used in a 50-μL PCR reaction containing dNTP (0.2 mmol/L), primers Fr3A and LJH (0.1 mmol/L each), MgCl2 (1 mmol/L), KCl (50 mmol/L), and Tris HCl, pH 8.3 (10 mmol/L). Thirty cycles of the reaction were carried out (denaturation, 94°C, 60 seconds; annealing, 57°C, 60 seconds; extension, 72°C, 60 seconds) followed by a 7-minute final extension step at 72°C. A second PCR was performed using 1 μL from the first PCR as template in 50 μL of the same buffer, but primer VLJH was used instead of LJH. Twenty-five cycles were carried out at the temperature setting of the first PCR.
For determination of the bcl-2/immunoglobulin heavy chain gene rearrangement, 2-step PCR was performed as described by Gribben et al24 using primers for the JH consensus region and the major breakpoint region with minor modifications. Only 0.2 and 0.5 μg of template DNA were used, and only a 2-μL aliquot was used for the second round of PCR. The MgCl2 concentration was 1.5 mmol/L; 30 cycles (denaturation, 94°C, 60 seconds; annealing, 61°C, 60 seconds; extension, 72°C, 90 seconds) were carried out for each amplification. After gel electrophoresis, PCR products were further analyzed by Southern blotting using an internal alkaline phosphatase-labeled oligonucleotide and detection by the Lumiphos system (Sigma Genosys, Cambridge, UK).
DNA sequencing
For JH gene sequencing, amplified DNA fragments were separated on a 2.5% Metaphor agarose gel (FMC) and were purified using the QIAquick Gel Extraction Kit (Qiagen, Hilden, Germany). DNA preparation (60 ng) was submitted to a cycle sequencing reaction (ABI PRISM BigDye Terminator Cycle Sequencing Ready Reaction Kit; Perkin–Elmer–Cetus, Norwalk, CT) and was analyzed with the ABI PRISM 310 Genetic Analyzer (Perkin–Elmer–Cetus).
Results
Clinical features
Fifteen patients with follicular lymphoma (4 men, 11 women; mean age, 53.1 years; range, 24 to 87 years) had solitary (n = 4), clustered (n = 5), or multiple (n = 6) reddish plaques or tumors located on the scalp (n = 3), face (n = 4), trunk (n = 4), upper extremities (n = 1), or multiple sites (n = 3) (Figure2, Table 1).
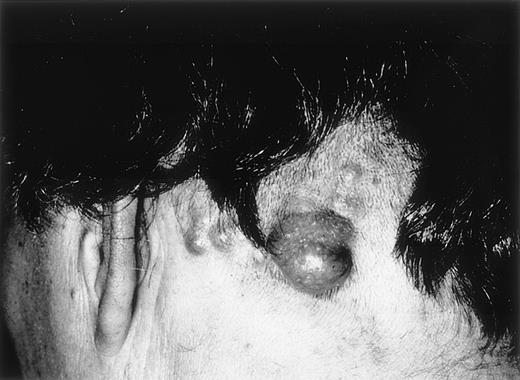
Cutaneous follicular lymphoma.
Clustered erythematous papules and tumors on the scalp, (patient 14).
Cutaneous follicular lymphoma.
Clustered erythematous papules and tumors on the scalp, (patient 14).
Clinical data and results of immunohistologic and molecular studies
| Patient . | Sex/age (y) . | Site (solitary/ clustered/multiple lesions) . | κ/λ . | JH-PCR (entire specimen) . | MD-PCR Follicles . | MD-PCR IF . | Therapy (response) . | Follow-up (mo) . |
|---|---|---|---|---|---|---|---|---|
| 1 | M/35 | Cheek, arm (MU) | − | M | M | P′ | SE (CR); recurrence | A&D (31) |
| 2 | F/53 | Forehead (CL) | − | P | P | P | Rx (CR) | A&W (39) |
| 3 | F/52 | Forehead (SO) | − | P | P | P | SE (CR); recurrence | A&D (21) |
| 4 | M/60 | Forehead (CL) | − | P | P | P | SE (CR); recurrence: Rx (CR) | A&W (38) |
| 5 | M/62 | Scalp (SO) | − | M | M | P′ | Rx (CR); recurrence | A&D (13) |
| 6 | F/48 | Scalp, arms (MU) | +κ | P | P | P | Rx (CR); recurrence: Rx (CR) | A&W (95) |
| 7 | F/42 | Shoulders (MU) | − | M | ND | ND | SE (CR) | A&W (14) |
| 8 | F/56 | Breast (SO) | − | P | P | P | SE + Rx (CR) | A&W (63) |
| 9 | F/75 | Abdomen (CL) | − | M | M | P′ | Rx (CR) | A&W (11) |
| 10 | F/64 | Forehead (CL) | − | M | ND | ND | SE + Rx (CR) | A&W (65) |
| 11 | F/87 | Scalp (MU) | − | M | M | P′ | Rx (PR) | A&D (13) |
| 12 | F/52 | Arms (MU) | − | M | ND | ND | SE (CR); recurrence: SE + Rx (CR) | A&W (113) |
| 13 | M/24 | Scalp, cheek (MU) | − | M | M | P | Rx (CR); recurrence: Rx (CR) | A&W (79) |
| 14 | M/46 | Scalp (CL) | − | P | P | P | Rx (CR) | A&W (81) |
| 15 | F/41 | Breast (SO) | − | M | M | P | SE + Rx (CR); recurrence | A&D (54) |
| Patient . | Sex/age (y) . | Site (solitary/ clustered/multiple lesions) . | κ/λ . | JH-PCR (entire specimen) . | MD-PCR Follicles . | MD-PCR IF . | Therapy (response) . | Follow-up (mo) . |
|---|---|---|---|---|---|---|---|---|
| 1 | M/35 | Cheek, arm (MU) | − | M | M | P′ | SE (CR); recurrence | A&D (31) |
| 2 | F/53 | Forehead (CL) | − | P | P | P | Rx (CR) | A&W (39) |
| 3 | F/52 | Forehead (SO) | − | P | P | P | SE (CR); recurrence | A&D (21) |
| 4 | M/60 | Forehead (CL) | − | P | P | P | SE (CR); recurrence: Rx (CR) | A&W (38) |
| 5 | M/62 | Scalp (SO) | − | M | M | P′ | Rx (CR); recurrence | A&D (13) |
| 6 | F/48 | Scalp, arms (MU) | +κ | P | P | P | Rx (CR); recurrence: Rx (CR) | A&W (95) |
| 7 | F/42 | Shoulders (MU) | − | M | ND | ND | SE (CR) | A&W (14) |
| 8 | F/56 | Breast (SO) | − | P | P | P | SE + Rx (CR) | A&W (63) |
| 9 | F/75 | Abdomen (CL) | − | M | M | P′ | Rx (CR) | A&W (11) |
| 10 | F/64 | Forehead (CL) | − | M | ND | ND | SE + Rx (CR) | A&W (65) |
| 11 | F/87 | Scalp (MU) | − | M | M | P′ | Rx (PR) | A&D (13) |
| 12 | F/52 | Arms (MU) | − | M | ND | ND | SE (CR); recurrence: SE + Rx (CR) | A&W (113) |
| 13 | M/24 | Scalp, cheek (MU) | − | M | M | P | Rx (CR); recurrence: Rx (CR) | A&W (79) |
| 14 | M/46 | Scalp (CL) | − | P | P | P | Rx (CR) | A&W (81) |
| 15 | F/41 | Breast (SO) | − | M | M | P | SE + Rx (CR); recurrence | A&D (54) |
CL, clustered lesions; MU, multiple lesions; SO, solitary lesions; κλ, immunohistochemical analysis of immunoglobulin light chain expression; MD-PCR follicles, microdissection-based PCR analysis of JH genes in follicles; MD-PCR IF, microdissection-based PCR analysis of JH genes in interfollicular areas; M, monoclonal pattern; P, polyclonal pattern; P′, polyclonal pattern with 1 or more interfollicular lanes showing a band at the same length as that observed within follicles; ND, not done; Rx, radiotherapy; SE, surgical excision; CR, complete response; PR, partial response; A&D, alive with skin disease; A&W, alive and well.
Three patients with MZL (3 men; mean age, 39.0 years; range, 27 to 57 years) had solitary erythematous tumors on the back (n = 2) or upper extremities (n = 1).
Seven patients with cutaneous B-cell pseudolymphoma (2 males, 5 females; mean age, 36.3 years; range, 9 to 60 years) had solitary, erythematous skin papules or small nodules located on the face (n = 1), ear (n = 3), trunk (n = 2), or upper extremities (n = 1).
Histology
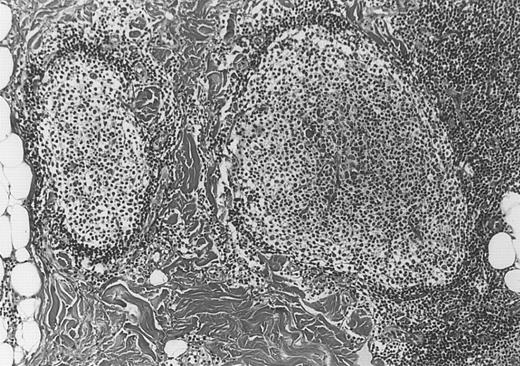
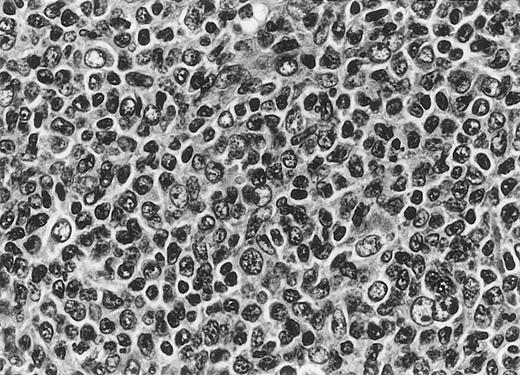
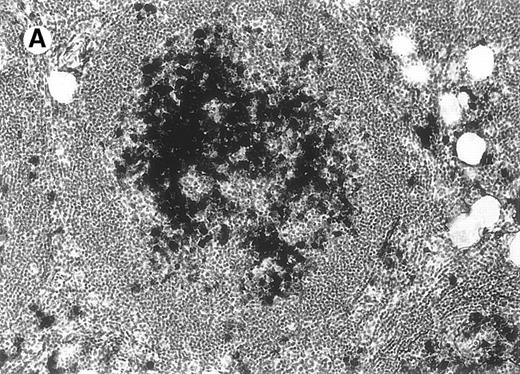
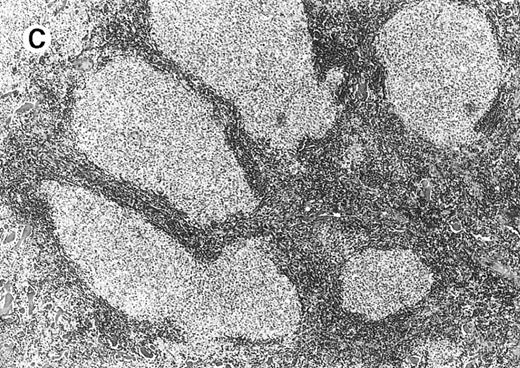
All cases of follicular lymphoma were classified as grade 2 according to the REAL classification. Histology showed bottom-heavy lymphoid infiltrates with prominent follicular pattern (Figures3 and 4). A well-formed mantle zone around most follicles was present only in 2 patients. In the other specimens the mantle zone was either reduced (11 patients) or nearly absent (2 patients) (Figure5). A clear-cut marginal zone was never observed. Tingible body macrophages within follicles were present only in 1 patient and were absent in the other 14 patients. Cytomorphology of follicles in all patients showed variable proportions of centrocytes (cleaved follicle center cells) and centroblasts (Figure6). The interfollicular areas revealed the presence of small lymphocytes and histiocytes admixed with a few larger cells and occasionally other inflammatory cells such as eosinophils and plasma cells.
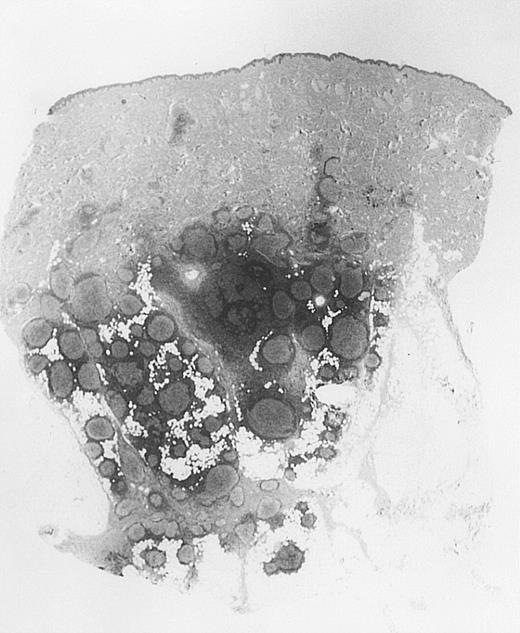
Bottom-heavy lymphoid infiltrate with prominent follicular pattern.
Located in the deep dermis and subcutaneous tissue (patient 9).
Bottom-heavy lymphoid infiltrate with prominent follicular pattern.
Located in the deep dermis and subcutaneous tissue (patient 9).
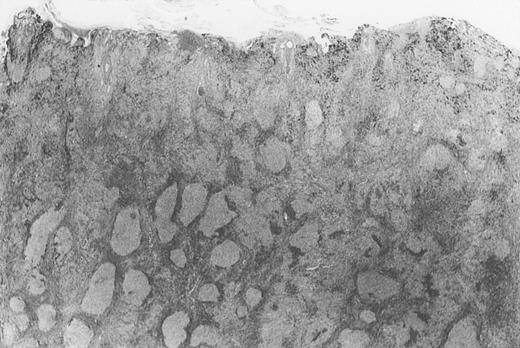
Dense infiltrate with prominent follicular pattern.
Appears within the entire dermis (patient 5).
Dense infiltrate with prominent follicular pattern.
Appears within the entire dermis (patient 5).
Neoplastic follicles.
The follicles lack a normal mantle zone and reveal nearly complete absence of tingible body macrophages (patient 9).
Neoplastic follicles.
The follicles lack a normal mantle zone and reveal nearly complete absence of tingible body macrophages (patient 9).
Cytomorphology.
Centroblasts and centrocytes evident within a follicle (patient 3).
Cytomorphology.
Centroblasts and centrocytes evident within a follicle (patient 3).
Histologic examination of patients with MZL revealed nodular infiltrates within the entire dermis and superficial subcutaneous fat tissue, characterized by the presence of follicles containing reactive germinal centers surrounded by a dense, pale staining perifollicular and interfollicular population of marginal zone cells admixed with plasma cells. Reactive germinal centers were small and showed tingible body macrophages and a normal mantle zone.
Histologic examination of patients with B-cell pseudolymphoma showed dermal nodules with a top-heavy arrangement of the infiltrates. Reactive germinal centers were characterized by regular morphology, prominent mantle zone, and tingible body macrophages. Small lymphocytes, histiocytes, eosinophils, and plasma cells were present in the interfollicular areas.
Immunohistology
In all patients with FCL, there was a predominant B-cell population (CD20+, CD79a+). Neoplastic follicles were outlined by CD21+ follicular dendritic cells. B-lymphocytes within follicles revealed CD20+, CD10+, CD5−, and CD43− phenotypes (Figure7A). Scattered CD10+ large B-lymphocytes also could be observed as solitary units or arranged in small clusters in the interfollicular areas in 9 patients. Staining with MIB-1 monoclonal antibody showed a decreased pattern of proliferation activity of the follicles (defined as the presence of less than 50% of positive cells) in 8 patients and a normal pattern in the other 7 (Figure 7B). Follicle center cells showed a monoclonal expression of κ light chain in 1 patient and were negative for both immunoglobulin light chains in the other 14 patients. Plasma cells, when present, expressed light chains at a normal ratio. Staining for the bcl-2 protein was negative in all follicles but positive in mantle cells and in small T and B-lymphocytes within interfollicular areas (Figure 7C). Variable numbers of reactive T lymphocytes were present within the follicles and in the interfollicular areas.
Immunohistology of neoplastic follicles.
(A) CD10 positivity of follicle center cells; note positivity of scattered cells within interfollicular areas. (B) Decreased reactivity of the follicles for the proliferation-associated antibody MIB-1. (C) Bcl-2 negativity of follicle center cells.
Immunohistology of neoplastic follicles.
(A) CD10 positivity of follicle center cells; note positivity of scattered cells within interfollicular areas. (B) Decreased reactivity of the follicles for the proliferation-associated antibody MIB-1. (C) Bcl-2 negativity of follicle center cells.
Neoplastic marginal zone cells in MZL were characterized by a CD20+, CD79a+, CD5−, CD43−, and Bcl-2+ immunophenotype. Marginal zone cells and plasma cells showed monoclonal restriction of immunoglobulin light chain expression in 2 patients (λ positive). One patient was negative for both κ and λ light chains. Reactive germinal centers in these patients were outlined by a network of CD21+ follicular dendritic cells. Germinal center cells were CD10+ and Bcl-2−. Clusters of CD10+ interfollicular B-lymphocytes were not observed.
Patients with pseudolymphoma showed a mixed infiltrate containing B and T lymphocytes. Reactive germinal centers showed a pattern similar to that observed in germinal centers of MZL. In these patients, clusters of CD10+ interfollicular B-lymphocytes were not observed.
Molecular biology
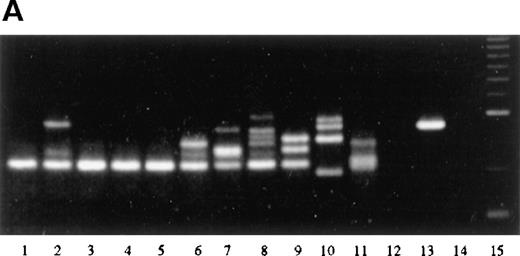
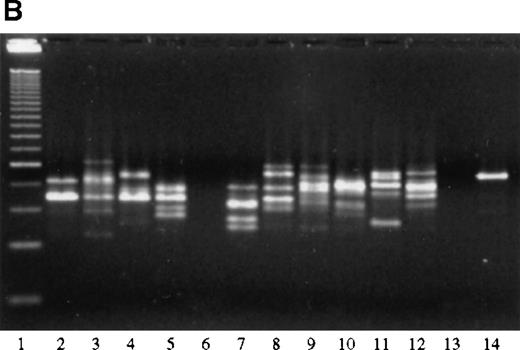
Analysis of the JH gene rearrangement after laser beam microdissection performed in 12 patients with FCL revealed in 6 of them the presence of a single band of the same length within samples from different follicles, indicating the existence of a monoclonal population of cells within different follicles from the same specimen (Figure 8A, Table 1). Interfollicular areas in these patients showed a polyclonal pattern of JH gene rearrangement, characterized by the presence of several bands of different lengths in the different areas analyzed. In 4 of the 6 patients showing the same distinct band within different follicles, a band of similar length could be detected also in 1 or more interfollicular areas from the same specimen, probably indicating the presence of neoplastic follicle center cells within these areas as well.
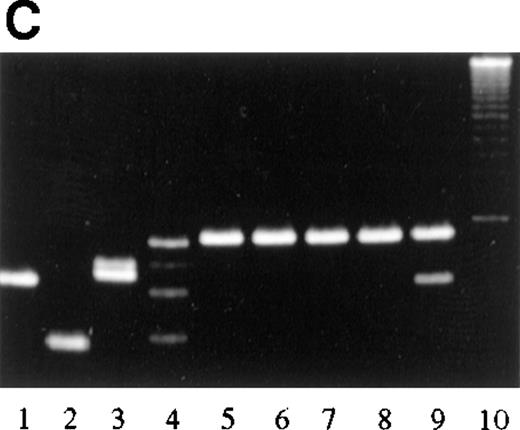
PCR analysis of microdissected specimens.
(A) Example of a monoclonal pattern of JH gene rearrangement of cells within the follicles after laser-based microdissection in 1 patient with FCL (patient 9). Lanes: 1 to 5, 5 different follicles; 6 to 11, 6 different interfollicular areas; 12, negative control (no DNA, external PCR run); 13, positive control (cutaneous B-cell lymphoma); 14, negative control (no DNA, internal PCR run); 15, molecular ladder. (B) Example of a polyclonal pattern of JH gene rearrangement of follicle center cells and interfollicular cells after laser-based microdissection in 1 patient with FCL (patient 6). Lanes: 1, molecular ladder; 2 to 7, 6 different follicles; 8 to 12, 5 different interfollicular areas; 13, negative control (no DNA, external PCR run); 14, positive control (cutaneous B-cell lymphoma). (C) Marginal zone lymphoma reveals monoclonality of interfollicular cells and polyclonal pattern of reactive germinal centers. Lanes: 1 to 4, reactive germinal centers; 5 to 9, interfollicular areas; 10, molecular ladder. One of the bands in lane 4 may represent follicular colonization by neoplastic marginal zone cells.
PCR analysis of microdissected specimens.
(A) Example of a monoclonal pattern of JH gene rearrangement of cells within the follicles after laser-based microdissection in 1 patient with FCL (patient 9). Lanes: 1 to 5, 5 different follicles; 6 to 11, 6 different interfollicular areas; 12, negative control (no DNA, external PCR run); 13, positive control (cutaneous B-cell lymphoma); 14, negative control (no DNA, internal PCR run); 15, molecular ladder. (B) Example of a polyclonal pattern of JH gene rearrangement of follicle center cells and interfollicular cells after laser-based microdissection in 1 patient with FCL (patient 6). Lanes: 1, molecular ladder; 2 to 7, 6 different follicles; 8 to 12, 5 different interfollicular areas; 13, negative control (no DNA, external PCR run); 14, positive control (cutaneous B-cell lymphoma). (C) Marginal zone lymphoma reveals monoclonality of interfollicular cells and polyclonal pattern of reactive germinal centers. Lanes: 1 to 4, reactive germinal centers; 5 to 9, interfollicular areas; 10, molecular ladder. One of the bands in lane 4 may represent follicular colonization by neoplastic marginal zone cells.
In the other 6 specimens analyzed after laser beam microdissection, no obvious monoclonal pattern of JH gene rearrangement could be observed either in the samples containing follicle center cells or in those including cells from interfollicular areas (Figure8B).
In 3 specimens in which laser beam microdissection could not be performed, analysis of the JH gene rearrangement was carried out using the entire section of tissue. After gel electrophoresis, a distinct band could be observed in all 3 specimens, indicating the presence of a monoclonal population of B lymphocytes.
Bands of the same length were detected within samples from different interfollicular areas of patients with MZL (Figure 8C), indicating the presence of the same monoclonal population of B lymphocytes in different interfollicular areas from the same specimen. Germinal centers revealed bands of different lengths in different areas analyzed, indicating that the population of germinal center cells was not derived from the same clone.
In all 4 pseudolymphoma specimens analyzed after laser beam microdissection, there was a polyclonal pattern of JH gene rearrangement in germinal center cells and in B lymphocytes from interfollicular areas.
Results of PCR and Southern blot analyses of the Bcl-2/IgH gene rearrangement in all patients with lymphoma and in 6 with pseudolymphoma were negative.
DNA sequencing
Analysis of JH gene sequences was performed in 3 of the 6 patients with FCL showing a monoclonal population of follicle center cells by laser-based PCR. The same DNA sequences could be observed in samples from different follicles in all 3, confirming the monoclonality of follicle center cells. Neoplastic germinal center cells within interfollicular areas could be demonstrated by DNA sequencing in 1 of them.
Treatment and follow-up
Data on treatment and follow-up could be obtained for all patients (mean follow-up, 48.7 months; range, 11 to 113 months; median, 39 months) (Table 1). Extracutaneous spread was not observed. Recurrent disease developed in 4 of 5 patients treated by surgery alone; 2 of them are alive with skin disease after 21 and 31 months, respectively. After further therapy, complete remission resulted in 2 patients, and they are alive without disease after 38 and 113 months, respectively. The fifth patient is in complete remission after 14 months. Six patients treated by local radiotherapy achieved a complete response. Three of them are alive with no evidence of disease after 11, 39, and 81 months, respectively. The other 3 had recurrent lesions at the same locations or at different skin sites. After further radiotherapy, 2 of these 3 patients achieved complete remission and are alive without skin lesions after 79 and 95 months, respectively, whereas the third is alive with skin disease 13 months after first diagnosis. One patient achieved a partial response after local radiotherapy and is alive with disease 13 months after first diagnosis. Three patients treated with surgical excision followed by radiotherapy achieved complete remission. Two of them are alive with no evidence of disease after 63 and 65 months, respectively. The third patient had a cutaneous recurrence at the same location and is alive with skin disease 54 months after the first diagnosis.
Discussion
B-cell lymphomas arising primary in the skin have been recognized recently as a distinct type of extranodal lymphoma with a particular clinicopathologic presentation and a favorable prognosis.1,25,26 Some authors maintain that they are neoplasms of germinal center cells, though a follicular pattern has been described only rarely27; others think that for the most part they represent examples of either immunocytoma or marginal zone lymphoma (MALT-type cutaneous lymphoma).9-11 We described herein 15 patients who had PCBCL with a clear-cut follicular growth pattern and demonstrated that these represent true examples of cutaneous follicular lymphoma.
The diagnoses of follicular lymphoma in our patients were based on clinicopathologic and molecular features. Most of our patients had large nodules and tumors on the head, a typical location of PCBCL. Histopathologic aspects suggesting follicular lymphoma included dense infiltrates with a bottom-heavy arrangement and a follicular growth pattern and atypical features of the follicles, including reduced or absent mantle zone (86.7% of patients), absent tingible body macrophages (93.3% of patients), and reduced proliferation rate (53.3% of patients).28,29 Immunohistology revealed a CD20+, CD10+, CD5−, CD43− phenotype of follicular B-lymphocytes, confirming their follicle center origin. The presence of interfollicular clusters of CD10+ cells in 9 patients also argued in favor of a diagnosis of follicular lymphoma. An immunoglobulin light chain restriction could be detected in only 1 patient. A monoclonal rearrangement of the JH gene could be observed in 9 of 15 patients (60.0%), a figure similar to that observed in nodal follicular lymphomas.30
The neoplastic nature of follicle center cells could be confirmed in half of the patients evaluated by PCR after laser beam microdissection of the specimens. Laser beam microdissection is a new technique that allows the isolation of single cells or small groups of cells for molecular analysis.31,32 The advantage of this method resides in the possibility of obtaining a pure population of cells uncontaminated by the DNA of neighboring cells, thus allowing a precise correlation between morphologic and molecular features. Using this technique, we could identify the same monoclonal band in cells from different follicles in the same specimen for 6 patients, confirming that follicle center cells indeed represent the neoplastic population. Sequencing of DNA in 3 of these 6 patients proved beyond any doubt the monoclonal origin of follicle center cells. It is of interest that in 4 of these 6 patients, a band similar to that observed in probes containing follicle center cells was present in 1 or more probes containing interfollicular lymphocytes. A similar finding was described recently33 in follicular lymphomas of the lymph nodes, possibly indicating the presence of neoplastic follicle center cells that move actively between follicles and interfollicular areas. In fact, analysis of JH gene sequences confirmed the presence of the neoplastic clone in interfollicular areas in 1 of 3 patients tested. In most of our patients, the presence of CD10+interfollicular cells in small clusters, the positivity of the follicles for CD10, and the absence of a clear-cut marginal zone excluded neoplastic populations of marginal zone cells showing follicular colonization, as described in some patients with nodal follicular lymphoma.34 This was confirmed, using the same technique, by the analysis of 3 patients who had cutaneous MZL with reactive germinal centers; a different pattern was clearly shown in this type of cutaneous PCBCL, characterized by the monoclonality of cells within interfollicular areas and the polyclonality of germinal centers.
In our study, after laser beam microdissection, 6 patients had no obvious monoclonal pattern of B-lymphocytes in follicles or interfollicular areas. However, the follicles showed morphologic and immunohistochemical features of malignancy, as discussed above. In 1 patient there was monoclonal expression of immunoglobulin light chain κ within germinal centers. The PCR assay result might have been negative because of the lack of sensitivity of the primers or the presence of a monoclonal rearrangement not detectable by our set of primers. This finding is similar to what can be observed in nodal follicular lymphoma, in which monoclonality can be detected by molecular analysis in approximately two thirds of patients.30 The polyclonal pattern of interfollicular cells in patients without evidence of monoclonality of the follicles, however, excludes that these cases are, in fact, examples of immunocytoma or marginal zone lymphoma with reactive germinal centers.
In the lymph nodes, follicular lymphoma often exhibits the 14;18 interchromosomal translocation involving the bcl-2oncogene.15-18,35-37 We and others previously demonstrated that t(14;18) is extremely rare in PCBCL.12-14 Analysis of t(14;18) and of bcl-2 protein expression in our patients revealed negative results, confirming the differences between nodal and cutaneous follicular lymphoma. It seems likely that lymphomas showing similar morphologic features but arising in different organs are characterized by different pathogenetic mechanisms. At present, no specific genetic alteration has been found in patients with PCBCL.
The prognosis and long-term follow-up of cutaneous follicular lymphoma seem to be similar to those described for other low-grade malignant PCBCL. Although in our patients cutaneous recurrences after surgical excision or local radiotherapy were common (53.3% of patients), extracutaneous spread was not observed after a mean follow-up of more than 4 years. Our data are in keeping with those reported in 1986 by Garcia et al.8 In that report, 3 of 9 patients were alive with skin disease, and 6 were alive with no evidence of disease, after a mean follow-up of 23.6 months (range, 2 to 60 months). Extracutaneous spread was not observed.
In conclusion, we have described a peculiar variant of PCBCL characterized by the proliferation of neoplastic follicle center cells with a prominent follicular pattern. These lymphomas represent a distinct morphologic subtype of PCBCL.
Acknowledgment
We thank Uli Schmidbauer for preparing the excellent histologic sections and the immunohistochemical stainings.
Reprints:Lorenzo Cerroni, Department of Dermatology, University of Graz, Auenbruggerplatz 8, A-8036 Graz, Austria; e-mail:lorenzo.cerroni@kfunigraz.ac.at.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal