To the Editor:
In a recent report, David et al1 addressed the ongoing controversy on expression levels of interleukin-2 receptor (IL-2R) α, β, and γ chains on various mononuclear cells from the peripheral blood. Using freshly isolated peripheral blood mononuclear cells (PBMCs) from (sodium) heparinized blood and fluorescein isothiocyanate (FITC)-labeled commercial monoclonal antibodies, they showed that all three IL-2R chains usually are hardly detectable on either CD4 or CD8 T cells from healthy donors and from hemochromatosis patients. These results are in contrast with the much higher levels of IL-2R subunits on T cells observed by several investigators, including ourselves.2-9 David et al1 tentatively explain the discrepancy by invoking effects of anticoagulant and of storage. Indeed, if Ca2+ chelators were used instead of heparin, the levels of all three IL-2R chains on T cells apparently increased, and overnight storage of heparinized blood also seemed to upregulate IL-2R subunit expression.
These observations are very important, because they not only seem to settle a long-standing controversy on IL-2R expression, but they also imply that the use of Ca2+ chelators as anticoagulant instead of heparin could dramatically influence the sensitivity of the T cells to IL-2. Because IL-2 and other common γ-chain triggering cytokines are central to almost any T-cell function, EDTA or citrate anticoagulants should be avoided if subsequent functional testing is envisioned. A lot of immunological research is based on buffy coats, which routinely are anticoagulated with citrate. In our own studies of T-cell function during human immunodeficiency virus (HIV) infection, we have systematically used EDTA blood as starting material, because it is readily available and because we did not find a functional difference between lymphocytes derived from blood anticoagulated with heparin, citrate, or EDTA in preliminary experiments. In view of the findings of David et al,1 we felt obliged to carefully control the effect of Ca2+ chelators on IL-2R expression and function and we did not observe any significant influence of the anticoagulant.
In three separate experiments, blood from five healthy control subjects (all lab personnel) was drawn at 10 am in three different tubes from Sarstedt containing either sodium heparin (final concentration, 0.3 mg/mL), potassium-EDTA (final concentration, 1.6 mg/mL), or sodium-citrate (final concentration, 10.6 mmol/L). The largest part of each tube was immediately processed for mononuclear cell (PBMC) separation, using Histopaque 1077 (Sigma, Bornem, Belgium), whereas the rest was kept at room temperature. At 2 pm, 50 μL of whole blood and 50 μL of PBMCs (containing 200,000 cells), derived from each of the three anticoagulant tubes, were incubated for 20 minutes at 4°C with 0.1 μg of the nonconjugated reference monoclonals anti-Tac (IL-2Rα–specific; obtained from Dr Thomas Waldman, National Institutes of Health, Bethesda, MD) and with 2R-B (IL-2Rβ–specific; from Dr Takashi Uchiyama, Institute for Virus Research, Kyoto University, Kyoto, Japan). As an isotypic (IgG1) control, we used purified 56D3 directed against an irrelevant parasitic antigen (provided by Dr J. Brandt, Institute of Tropical Medicine, Antwerpen, Belgium). After washing with phosphate-buffered saline (PBS), containing 0.5% bovine serum albumin, 1 μL of FITC-labeled F(ab′)2 goat antimouse IgG (Tago, Burlingame, CA) was added for another 20 minutes. After washing again, the remaining binding sites on the FITC-conjugate were blocked with 5 μL of mouse serum. Next, 5 μL of phycoerythrin (PE)-labeled anti-CD4 and 5 μL of peridinin-chlorophyll A protein (PercP)-labeled anti-CD3 (both from Becton Dickinson, Erembodegem, Belgium) were added for the last 20 minutes. The tubes with whole blood were then subjected to the Becton Dickinson lysing solution. All preparations were washed once and fixed with 1% paraformaldehyde. The samples were analyzed on a FACScan (Becton Dickinson) using the LYSYS I software.
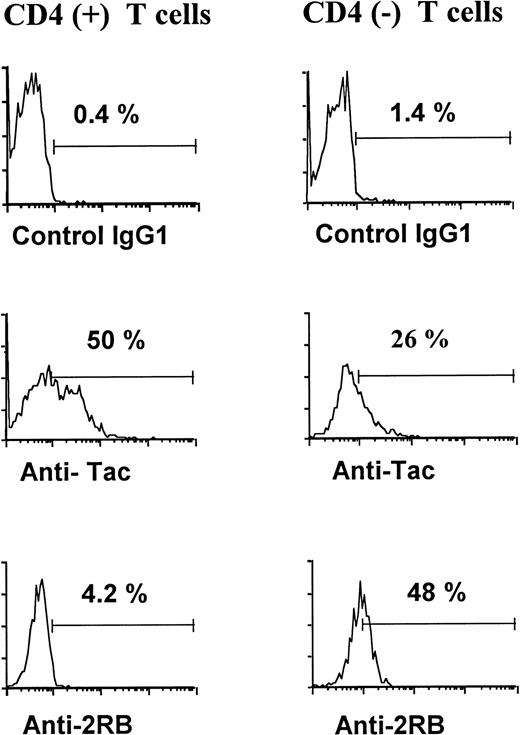
Based on the scatter and the CD3/CD4 expression, the CD4+and CD4− T lymphocytes were gated separately and the distribution of the first fluorescence was represented in a histogram for each subset. An example of this analysis is shown in Fig1. It is evident that, within both the CD4+ and CD4− T-cell populations, the expression profile of IL-2Rα is rather broad and tends to be bimodal (a negative and a positive subpopulation), whereas the curve of IL-2Rβ is unimodal and shows a shift to the right, which is most evident in the CD4− subset. We chose to express the results for both chains as percentage of positive cells, after establishing a narrow threshold at a relative fluorescence intensity of 10, based on the background of the control monoclonal. A summary of the results is shown in Table 1. No significant difference was observed in the level of IL-2R α and β chains on CD4+ or CD4− T cells, according to the anticoagulant used and regardless of whether the cells were stained in the context of whole blood or PBMCs. Comparing the mean fluorescence intensity of all gated cells (instead of the percentage of positive cells) showed similar results and confirmed that the low level of IL-2Rβ expression on CD4+ T cells significantly differed from background (data not shown).
Representative binding profiles of a control MoAb, the anti–IL-2Ra MoAb anti-Tac, and the anti–IL-2Rb MoAb 2RB on CD4+ and CD4− T cells, assessed in three-color flow cytometry.
Representative binding profiles of a control MoAb, the anti–IL-2Ra MoAb anti-Tac, and the anti–IL-2Rb MoAb 2RB on CD4+ and CD4− T cells, assessed in three-color flow cytometry.
Effect of Anticoagulant on T-Cell Expression of IL-2R and β Chains
| . | CD4+ T Cells . | CD4− T Cells . | ||||
|---|---|---|---|---|---|---|
| Control IgG1 . | Anti–IL-2R α . | Anti–IL-2R β . | Control IgG1 . | Anti–IL-2R α . | Anti–IL-2R β . | |
| K-EDTA | ||||||
| Whole blood | 0.3 ± 0.2 | 49.6 ± 8.2 | 2.3 ± 1.3 | 0.7 ± 0.6 | 14.5 ± 5.1 | 28.4 ± 13.4 |
| PBMCs | 0.7 ± 0.3 | 49.2 ± 3.5 | 3.3 ± 0.8 | 1.4 ± 1.2 | 15.5 ± 5.6 | 30.8 ± 10.2 |
| Na-heparin | ||||||
| Whole blood | 3.4 ± 6.6 | 49.7 ± 6.3 | 2.7 ± 1.0 | 2.8 ± 4.9 | 15.9 ± 5.3 | 32.2 ± 14.5 |
| PBMCs | 1.0 ± 0.6 | 48.4 ± 5.7 | 3.3 ± 0.4 | 1.3 ± 0.6 | 14.5 ± 3.6 | 30.7 ± 11.9 |
| Na-citrate | ||||||
| Whole blood | 0.3 ± 0.3 | 49.6 ± 7.9 | 2.6 ± 1.0 | 0.9 ± 0.8 | 16.2 ± 6.1 | 31.1 ± 12.6 |
| PBMCs | 0.9 ± 0.5 | 49.7 ± 6.5 | 2.6 ± 0.7 | 1.4 ± 0.7 | 15.3 ± 4.5 | 26.0 ± 8.6 |
| . | CD4+ T Cells . | CD4− T Cells . | ||||
|---|---|---|---|---|---|---|
| Control IgG1 . | Anti–IL-2R α . | Anti–IL-2R β . | Control IgG1 . | Anti–IL-2R α . | Anti–IL-2R β . | |
| K-EDTA | ||||||
| Whole blood | 0.3 ± 0.2 | 49.6 ± 8.2 | 2.3 ± 1.3 | 0.7 ± 0.6 | 14.5 ± 5.1 | 28.4 ± 13.4 |
| PBMCs | 0.7 ± 0.3 | 49.2 ± 3.5 | 3.3 ± 0.8 | 1.4 ± 1.2 | 15.5 ± 5.6 | 30.8 ± 10.2 |
| Na-heparin | ||||||
| Whole blood | 3.4 ± 6.6 | 49.7 ± 6.3 | 2.7 ± 1.0 | 2.8 ± 4.9 | 15.9 ± 5.3 | 32.2 ± 14.5 |
| PBMCs | 1.0 ± 0.6 | 48.4 ± 5.7 | 3.3 ± 0.4 | 1.3 ± 0.6 | 14.5 ± 3.6 | 30.7 ± 11.9 |
| Na-citrate | ||||||
| Whole blood | 0.3 ± 0.3 | 49.6 ± 7.9 | 2.6 ± 1.0 | 0.9 ± 0.8 | 16.2 ± 6.1 | 31.1 ± 12.6 |
| PBMCs | 0.9 ± 0.5 | 49.7 ± 6.5 | 2.6 ± 0.7 | 1.4 ± 0.7 | 15.3 ± 4.5 | 26.0 ± 8.6 |
Values are the percentage of cells within the indicated subset showing a relative fluorescence intensity of greater than 10 after incubation with the indicated monoclonal (mean ± SD).
We next wanted to know whether the anticoagulant influences the sensitivity to IL-2. To this end, we cultured the three preparations of PBMCs at a final concentration of 106/mL in RPMI, supplemented with antibiotics (GIBCO, Paisley, UK) and 10% bovine calf serum (Hyclone, Logan, UT), in the presence or absence of 100 U/mL IL-2 (Boehringer-Mannheim, Brussels, Belgium) at 37°C in a 5% CO2 atmosphere. After an overnight incubation, the cells were stained with a combination of either FITC-labeled IgG1 or anti-CD69-FITC and anti-CD4-PE plus anti-CD3-PercP (all from Becton Dickinson). As shown in Table 2, the specific expression of CD69 on CD4+ and CD4− T cells is similarly upregulated by IL-2, irrespective of the original anticoagulant. The only (nonsignificant) difference was a lower IL-2–induced CD69 on CD4− T cells from EDTA blood, as compared with heparinized or citrated blood. The latter observation certainly does not plead for a higher sensitivity of T cells from EDTA blood, which is to be expected if the latter anticoagulant was able to upregulate IL-2R expression, as suggested by David et al.1
Effect of Anticoagulant on T-Cell Sensitivity to IL-2
| Culture Condition . | % CD69 Expression After 1 Day of Culture . | |||
|---|---|---|---|---|
| On CD4+ T Cells . | On CD4− T Cells . | |||
| Medium Only . | IL-2 (100 U/mL) . | Medium Only . | IL-2 (100 U/mL) . | |
| PBMC isolated from | ||||
| K-EDTA blood | 3.3 ± 1.5 | 9.5 ± 5.7 | 10.7 ± 2.4 | 28.6 ± 6.4 |
| Na-heparin blood | 2.1 ± 0.6 | 9.8 ± 19.0 | 9.4 ± 3.2 | 40.7 ± 18.6 |
| Na-citrate blood | 3.3 ± 1.6 | 10.0 ± 13.9 | 9.4 ± 1.8 | 37.6 ± 14.7 |
| Culture Condition . | % CD69 Expression After 1 Day of Culture . | |||
|---|---|---|---|---|
| On CD4+ T Cells . | On CD4− T Cells . | |||
| Medium Only . | IL-2 (100 U/mL) . | Medium Only . | IL-2 (100 U/mL) . | |
| PBMC isolated from | ||||
| K-EDTA blood | 3.3 ± 1.5 | 9.5 ± 5.7 | 10.7 ± 2.4 | 28.6 ± 6.4 |
| Na-heparin blood | 2.1 ± 0.6 | 9.8 ± 19.0 | 9.4 ± 3.2 | 40.7 ± 18.6 |
| Na-citrate blood | 3.3 ± 1.6 | 10.0 ± 13.9 | 9.4 ± 1.8 | 37.6 ± 14.7 |
Values are the percentage of cells within the indicated subset showing specific binding to anti-CD69-FITC (mean ± SD).
To make absolutely sure that the storage of 4 hours we used in our standard procedure did not influence the results, we performed two additional experiments in which either whole blood anticoagulated with heparin, EDTA, or citrate or the isolated PBMCs were stained for IL-2Rα or CD69 either immediately or after 4 hours of incubation at room temperature. Again, no differences in CD25 or CD69 expression were observed, regardless of whether completely fresh cells or cells after 4 hours were used, thus excluding spurious activation upon short-term storage of blood or PBMCs.
In summary, our well-controlled flow cytometric measurements, using a sensitive indirect labeling technique and reference anti-α and anti-β monoclonal antibodies, confirm our own and other investigators’ previous findings that both IL-2R α and β chains are expressed to a significant extent on both resting CD4+and CD4− T cells.2-9 We further confirmed that the α chain is predominantly present on CD4 T cells, whereas the β chain is more prominently expressed on the CD4−subset, largely corresponding to the CD8+ T cells. Moreover, our present data clearly show that the level of IL-2R chain expression is not influenced by the use of a Ca2+-chelating anticoagulant and is similar in the context of fresh whole blood and freshly isolated PBMCs. We did not investigate the effect of prolonged storage, because, in our experience, such a treatment results in a variable degree of viability loss, which might induce artifacts. However, short-term storage for 4 hours had no influence on either IL-2Rα or CD69. Our culture experiments with added IL-2 further indicated that the IL-2R function, as measured by upregulation of CD69 on T cells, is also not significantly influenced by the anticoagulant.
In our previous studies, we showed that the CD69 upregulation by IL-2 on CD4 T cells could be blocked by a combination of anti–IL-2R α and β monoclonals, pointing to an implication of the high-affinity receptor. In CD8 T cells, nearly complete blocking could be obtained by anti-β monoclonals alone at high IL-2 concentrations, whereas, at lower concentration, a synergistic effect of anti-α and anti-β monoclonals was obvious.2 Thus, the expression of both α and β chains was shown to be of functional relevance in both T-cell subsets. In a separate report, we recently demonstrated IL-2R γ chain expression on at least 60% of both CD4 and CD8 T cells from healthy donors and HIV-infected subjects.10
An incomplete screening of the literature found that at least two other independent groups of investigators who stained PBMCs isolated from heparinized blood with the same reference anti-Tac monoclonal but a different anti-β (Mik1-β) showed expression levels of IL-2R chains on peripheral T cells very similar to those we observed.5,6However, other investigators, also starting from heparinized blood but using different monoclonals and/or different staining procedures, reported lower or even no measurable expression of the IL-2R subunits, indeed.11-13 The seemingly low expression of IL-2R chains observed by some investigators thus might relate to technical factors, including the affinity of the antibodies used or the sensitivity of the cytofluorometer rather than to the kind of anticoagulant.
In conclusion, we feel that the use of sensitive staining procedures is crucial to correctly assess IL-2R expression on resting T cells. Moreover, we are convinced that the anticoagulant does not influence IL-2R expression or function in peripheral blood T cells.
We have recently published in BLOOD the first extensive survey concerning IL-2Rα, IL-2Rβ, and IL-2Rγ expression by human PBMC subsets.1-1 More than 100 donors were examined through a 4-year period and the results are consistent with our published data.1-1 Throughout the work, care was taken to study healthy donors and resting PBMCs. The reported results were obtained by measuring cell surface and intracellular expression of IL-2Rα, IL-2Rβ, and IL-2Rγ by flow cytometry. The data were verified for the three chains by measuring mRNA expression. For intracellular expression of IL-2Rγ, Western blot analysis was also performed. Table 1-1 summarizes the data.
Expression of IL-2R Chains and Specific mRNA by PBMCs of Healthy Adult Donors: Summary of our Data Presented inBLOOD
| . | CD4 T Lymphocytes . | B Lymphocytes . | CD8 T Lymphocytes . | Monocytes . | NK Cells . |
|---|---|---|---|---|---|
| Cell surface expression (flow cytometry)* | |||||
| α | <5%† | <5% | <5% | <5% | <5% |
| β | <5% | <5% | 14% ± 5.2%‡ | <5% | 78% ± 3.1% |
| γ | <5% | <5% | 8.6% ± 7.5%‡ | 49% ± 6.0%2-153 | <5% |
| Intracellular protein (intracellular flow cytometry) | |||||
| α | <5% | <5% | <5% | ND | <5% |
| β | <5% | <5% | <5% | ND | >40% |
| γ | >60% | >45% | >45% | ND | >60% |
| mRNA (RT-PCR) | |||||
| α | — | — | — | — | — |
| β | — | — | —(+)2-155 | — | + |
| γ | + | + | + | + | + |
| . | CD4 T Lymphocytes . | B Lymphocytes . | CD8 T Lymphocytes . | Monocytes . | NK Cells . |
|---|---|---|---|---|---|
| Cell surface expression (flow cytometry)* | |||||
| α | <5%† | <5% | <5% | <5% | <5% |
| β | <5% | <5% | 14% ± 5.2%‡ | <5% | 78% ± 3.1% |
| γ | <5% | <5% | 8.6% ± 7.5%‡ | 49% ± 6.0%2-153 | <5% |
| Intracellular protein (intracellular flow cytometry) | |||||
| α | <5% | <5% | <5% | ND | <5% |
| β | <5% | <5% | <5% | ND | >40% |
| γ | >60% | >45% | >45% | ND | >60% |
| mRNA (RT-PCR) | |||||
| α | — | — | — | — | — |
| β | — | — | —(+)2-155 | — | + |
| γ | + | + | + | + | + |
Values in bold and underlined are results in disagreement with the letter of Vanham et al, whereas values in italic and underlined are results in partial agreement with the letter of Vanham et al.
Abbreviations: ND, not determined; —, not detected.
Percentage of positive cells.
Expression of less than 5% is considered nonsignificant.
Two kinds of donors can be distinguished. Most of them do not express significantly β or γ IL-2R chains, whereas 30% of them highly expressed these chains. This results in an important standard deviation.
IL-2Rγ chain is expressed for all donors but with a variable intensity.
mRNA detected for few donors in agreement with the ‡ note.
Several reasons may explain why Vanham et al were not able to obtain the same results concerning cell surface expression of IL-2Rα by CD4 and CD8 T lymphocytes.
(1) Their donors were apparently not controlled. Under some circumstances, we also found IL-2Rα expression by CD4 T cells, for instance during and after winter and spring seasonal infections. In our study, donors went through a rigorous medical check-up before blood collection.
(2) Their assay does not identify a clear negative lymphocyte population; therefore, we cannot exclude either that their assay is not reliable or that their cells are activated. Under these conditions, it may not be surprising that they do not find any influence of anticoagulants. In our hands, all the PBMC subsets analyzed from healthy donors were negative for IL-2Rα expression, and we use cells from hemochromatosis patients as a positive control to show that our assay was able to detect IL-2Rα when expressed. These results were misinterpreted by Vanham et al. Furthermore, we show that IL-2Rα could be in vitro induced in IL-2Rα–negative resting CD4 T cells after various stimulations.1-2
(3) The very poor and inconsistent (high standard deviation) induction of CD69 by CD4 T cells after IL-2 stimulation shown by Vanham et al suggests that their cells do not express IL-2R, and not the contrary. Furthermore, we previously demonstrated that IL-2 reactivity does not always correlate with IL-2R expression.1-3
In conclusion, it is commonly accepted that CD25 expression correlates with T-cell activation and that resting T cells do not express CD25.1-4-1-10 Therefore, we believe that Vanham et al should make a more detailed study before claiming high expression of CD25 by resting CD4 T cells. They should be aware that, in the IL-2R system, there are not yet defined reference reagents and techniques and that their methods may have to be improved and controlled if they intend to modify almost consensual data concerning IL-2Rα expression.
REFERENCES
ACKNOWLEDGMENT
Supported by Grants No. 3.0307.95 and 3.0226.96 of the “Fonds voor Wetenschappelijk Onderzoek Vlaanderen” (Fund for Scientific Research of Flanders).

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal