Abstract
Mobilized peripheral blood stem cells (PBSC) are used as a source of hematopoietic stem cells for transplantation and gene therapy. It is still unclear, however, whether the PBSC are fully equivalent to normal bone marrow hematopoietic stem cells and whether they are able to provide long-term function of transgene in reconstituted mice. In the present study, mobilized PBSC from male mice were transduced with human adenosine desaminase gene (hADA) and were used for reconstitution of lethally irradiated female mice. At 1, 3, 6, 9, and 12 months after reconstitution, the bone marrow cells were repeatedly collected from each mouse under light anesthesia and the number of colony-forming unit-spleen (CFU-S), spleen repopulating ability (SRA), and the integration of human ADA gene were studied in CFU-S–derived colonies by polymerase chain reaction (PCR) and Southern blot hybridization analyses. After 9 months, the proportion of donor CFU-S detected by PCR with a Y-chromosome–specific probe in mice reconstituted with mobilized PBSC was 75.3% ± 6.0%, which is similar to the concentration of donor CFU-S seen after bone marrow transplantation. Similarly, there was no difference in the concentration of CFU-S in mice reconstituted with transduced mobilized PBSC or bone marrow cells. However, in both cases the CFU-S content in the bone marrow was reduced fivefold to 10-fold compared with the concentration of CFU-S in mice transplanted with nontransduced bone marrow. The SRA of CFU-S in mice reconstituted with peripheral blood and bone marrow cells was the same 1.5 months posttransplantation, but after an additional 4 months, SRA of mice reconstituted with bone marrow cells was fivefold higher as compared with those engrafted by PBSC. The integration of the human ADA gene was observed during 9 months in about 60% of studied CFU-S. The proportion of marked colonies sharply decreased 1 year following reconstitution. One to 9 individually labeled clones could be shown simultaneously by Southern blot hybridization in the same reconstituted mice during the whole period of observation. The time of clone existence was about 3 months. We conclude that long-term marrow repopulating cells mobilized into circulation by treatment with granulocyte colony-stimulating factor (G-CSF ) and stem cell factor (SCF ) are capable of maintaining lifelong polyclonal hematopoiesis in reconstituted mice.
PERIPHERAL BLOOD stem cells (PBSC) are successfully used as a source of stem cells both to provide support for autologous transplantation and as an alternative to bone marrow transplantation.1,2 There are only small numbers of hematopoietic stem cells (HSC) in the peripheral blood.3 However, following chemotherapy and/or growth factor injections, a substantial transient rise in PBSC occurs.4-7 Cytokines mobilize long-term culture-initiating cells (LTC-IC) and young colony-forming unit-spleen (CFU-S) with high proliferative potential into circulation.8-10 Cytokine type and administration protocol determines the proliferative potential of mobilized HSC and their maturation stage. Granulocyte colony-stimulating factor (G-CSF ) in combination with stem cell factor (SCF ) mobilizes more primitive HSC than each factor alone in nonhuman primates, dogs, and mice.11-13 Administration of G-CSF or granulocyte-macrophage colony-stimulating factor (GM-CSF ) to normal donors mobilizes a more mature subpopulation of HSC than their combination.14 In different animal models, it has been shown that a course of 5 to 8 days of cytokine treatment is sufficient for mobilization of hematopoietic precursors.11-13 The quality of mobilized PBSC is dependent on cytokines used and the duration of their administration. It was demonstrated that a 2-week course of SCF with G-CSF administration mobilizes a more primitive population of PHSC with high proliferative potential, including LTC-IC and marrow repopulating cells, as compared with a 5- to 8-day course.10
For characterization of PBSC mobilized by long-term cytokine administration and for their comparison with bone marrow cells, hematopoietic precursors were marked using retroviral gene transfer. The mobilized peripheral blood progenitor cells can be efficiently transduced by recombinant retroviruses.15-18 In addition, the efficiency of gene transfer into PBSC is higher than into bone marrow HSC.19-21
Here, we report for the first time, the developmental fate of individual clones of the CFU-S transduced with human adenosine deaminase (hADA) sequences. For this purpose, we developed a technique for the longitudinal analysis of the individually marked single precursor cell, CFU-S, by sequential study of bone marrow CFU-S from the same reconstituted mouse. It was shown by this method that PBSC are capable of maintaining hematopoiesis for at least 12 months. However, even these “young” PBSC, which were mobilized by an optimized protocol, differ from bone marrow HSC.
MATERIALS AND METHODS
Mice.Twelve- to 25-week-old male and female BDF1 (C57Bl/6xDBA/2)F1 mice were used as donors and recipients, respectively. Recipient mice were exposed to 1,200 cGy 137Cs irradiation (the dose rate 18 cGy/min, IPK irradiator). The dose was divided into two equal exposures, given 3 hours apart.
Cytokine treatment.Donor mice were injected daily, subcutaneously, for 14 days with a combination of recombinant human granulocyte colony-stimulating factor (rhG-CSF ) 250 μg/kg/d and recombinant rat stem cell factor (rrSCF ) 34 μg/kg/d (Amgen, Thousand Oaks, CA). Twenty-four hours after the last injection, peripheral blood was collected. Mononuclear cells were separated by centrifugation over Hystopaque (density 1.083 g/mL) (Sigma, St Louis, MO) for 30 minutes, 450g, and washed three times with αMEM. Femoral bone marrow was flushed out and resuspended in αMEM.
Donor Cell Characteristics
| Donor Cells . | Before Gene Transfer Procedure . | After Gene Transfer Procedure . | No. of CFU-S Injected per Mouse . | ||
|---|---|---|---|---|---|
| . | No. of CFU-S per 105 Cells . | SRA . | No. of CFU-S per 105 Cells . | SRA . | . |
| Peripheral blood | 10.3 ± 1.3 | 124.7 ± 24.5 | 2.1 ± 0.2 | 22.8 ± 5.9 | 140.7 ± 13.4 |
| Bone marrow | 3.0 ± 0.8 | 29.8 ± 8.5 | 1.4 ± 0.2 | 9.1 ± 3.1 | 145.6 ± 20.8 |
| Donor Cells . | Before Gene Transfer Procedure . | After Gene Transfer Procedure . | No. of CFU-S Injected per Mouse . | ||
|---|---|---|---|---|---|
| . | No. of CFU-S per 105 Cells . | SRA . | No. of CFU-S per 105 Cells . | SRA . | . |
| Peripheral blood | 10.3 ± 1.3 | 124.7 ± 24.5 | 2.1 ± 0.2 | 22.8 ± 5.9 | 140.7 ± 13.4 |
| Bone marrow | 3.0 ± 0.8 | 29.8 ± 8.5 | 1.4 ± 0.2 | 9.1 ± 3.1 | 145.6 ± 20.8 |
Survival of Reconstituted Mice
| Donor Cells . | Time After Reconstitution . | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| . | 1.5 mo . | 9 mo . | 12 mo . | . | . | . | |||
| . | Mice Survived/Total No. of Mice . | % of Survival . | Mice Survived/Total No. of Mice . | % of Survival . | Mice Survived/Total No. of Mice . | % of Survival . | . | . | . |
| Peripheral blood | 11/12 | 92 | 7/11 | 64 | 5/11 | 45 | |||
| Bone marrow | 12/12 | 100 | 9/12 | 75 | 6/12 | 50 | |||
| Donor Cells . | Time After Reconstitution . | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| . | 1.5 mo . | 9 mo . | 12 mo . | . | . | . | |||
| . | Mice Survived/Total No. of Mice . | % of Survival . | Mice Survived/Total No. of Mice . | % of Survival . | Mice Survived/Total No. of Mice . | % of Survival . | . | . | . |
| Peripheral blood | 11/12 | 92 | 7/11 | 64 | 5/11 | 45 | |||
| Bone marrow | 12/12 | 100 | 9/12 | 75 | 6/12 | 50 | |||
Retroviral vector and producing cell line.GP + E86 cell line producing a retrovirus containing human ADA-cDNA, expressed internally from the human phosphoglycerate kinase (PGK) promoter, was a generous gift of D.A. Williams (Indianapolis, IN). The details of the vector have been previously reported. 22 This cell line produced virus with a titer 1 × 106 CFU/mL. One day before infection, flasks with confluent producer cells were treated with 5 μg/mL of mitomycin C at 37°C for 2.5 hours, repeatedly washed with Hanks' Balanced Salt Solution (HBSS), detached by trypsin-EDTA, and split 1:2 in αMEM with 20% fetal bovine serum.
Gene transfer.For prestimulation, the marrow cells and peripheral blood mononuclears were cultured for 2 days in 0.1% gelatine-treated T-25 flasks at 37° C in 10-mL αMEM with 20% fetal calf serum; medium was supplemented with rrSCF 50 ng/mL and recombinant human interleukin-6 (50 U/mL) (Amgen). Prestimulated bone marrow cells were transferred on the monolayer of producer cells in the media containing 4 μg/mL polybrene and the same exogenous cytokines. Two days later the hematopoietic cells were washed out from producer cell line and used for reconstitution of irradiated mice. Each of the 12 recipients received 6.7 × 106 peripheral blood mononuclear cells; 12 recipients were injected with 10.4 × 106 bone marrow cells.
Aspiration of bone marrow cells for DNA analysis.Bone marrow samples were obtained under light ether anesthesia from individual reconstituted mice 1.5, 3, 6, 9, and 12 months after transplantation. Bone marrow was aspirated repeatedly from the left and right femurs, in turn, by puncture through the knee joint with a 22-gauge needle. It was usually possible to obtain 5 to 15 × 106 bone marrow cells from the femur of the living mouse. Aliquots of bone marrow from each mouse were injected into six irradiated female recipients for CFU-S analysis and the remaining bone marrow cells were used for DNA isolation.
CFU-S and spleen repopulating ability (SRA) analysis.Irradiated female mice were injected intravenously (IV) with 1 to 4 × 105 bone marrow cells from reconstituted mice. Individual macroscopic spleen colonies were isolated under a dissection microscope 11 days later and used both for DNA analysis and for determination of the SRA (number of CFU-S–8 per 11-day-old spleen colony).23 To determine SRA, half of the colony cells were injected into irradiated secondary recipients. The number of daughter colonies was counted 8 days later and the number of colonies generated per one CFU-S–11 was calculated.24
Polymerase chain reaction (PCR) and Southern blot analysis.DNA from total bone marrow and individual spleen colonies was extracted and PGK-hADA provirus was detected by PCR. Primers in ADA coding sequences (5′GACAAGCCCAAAGTAGAACTGC3′ - sense and 5′TGACCCCGAAGTCTCGCTCC3′- antisense) amplified a 418-bp fragment of proviral genome. Thirty cycles of PCR amplification were performed under the following conditions: denaturation, 30 seconds at 94°C; annealing, 30 seconds at 60°C; extension, 30 seconds at 72°C. DNA from PCR-positive samples was digested with EcoRI, electrophoresed through a 1% agarose gel, transferred to Hybond N filter and hybridized with ADA cDNA probe, prepared from HindIII 1.2-kb PGK-hADA fragment or with PCR amplified 418-bp fragment of ADA gene. Digestion with EcoRI permitted analysis of individual clones of hematopoietic cells, as only one EcoRI restriction site is present within the vector used.
Determination of CFU-S origin in reconstituted mice.Only male mice were used as bone marrow donors, recipients were always female. For identification of CFU-S origin, either PCR analysis or blot hybridization was used. The primers were chosen in the C-terminal domain of the sex-determining region25 26 of the mouse Y-chromosome (5′CTCCTGATGGACAAACTTTACG3′- sense and 5′TGAGTGCTGATGGGTGACGG3′- antisense), amplified a 444-bp fragment of genome. Thirty cycles of PCR amplification were formed under the following conditions: denaturation, 45 seconds at 94°C; annealing, 60 seconds at 60°C; extension, 45 seconds 72°C. Some filters were rehybridized with the Y-chromosome probe after stripping the ADA probe.
Statistics.Statistical analysis was done using Student's t-test.
RESULTS
One day after the course of cytokine injections, peripheral blood and bone marrow cells were harvested from donor mice and transduced by retroviral vector containing human ADA sequence. After gene transfer, peripheral blood and bone marrow cells were used for reconstitution of lethally irradiated recipients. The relative content of CFU-S after the procedure of gene transfer was only 1.5 to 2 per 105, both for peripheral blood mononuclear and bone marrow cells, lower than in the initial innoculum. The number of CFU-S was reduced twice in bone marrow. A more pronounced (fivefold) decrease was observed for peripheral blood mononuclear cells. Moreover, the SRA of freshly harvested donor cells was significantly higher than SRA of cells after gene transfer (Table 1).
CFU-S Content (per 106 cells) in Bone Marrow of Reconstituted Mice
| Donor Cells . | Time After Reconstitution (mo) . | ||||
|---|---|---|---|---|---|
| . | 1.5 . | 3 . | 6 . | 9 . | 12 . |
| Peripheral blood | 17.7 ± 5.0 | 28.6 ± 3.6 | 8.3 ± 1.2 | 12.1 ± 2.5 | 24.3 ± 4.6 |
| Bone marrow | 12.2 ± 1.2 | 10.7 ± 2.6 | 11.5 ± 1.3 | 20.5 ± 5.1 | 12.8 ± 3.3 |
| Donor Cells . | Time After Reconstitution (mo) . | ||||
|---|---|---|---|---|---|
| . | 1.5 . | 3 . | 6 . | 9 . | 12 . |
| Peripheral blood | 17.7 ± 5.0 | 28.6 ± 3.6 | 8.3 ± 1.2 | 12.1 ± 2.5 | 24.3 ± 4.6 |
| Bone marrow | 12.2 ± 1.2 | 10.7 ± 2.6 | 11.5 ± 1.3 | 20.5 ± 5.1 | 12.8 ± 3.3 |
Concentration of CFU-S in the bone marrow of normal BDF1 and mice reconstituted with normal bone marrow cells is about 150 to 200/106.
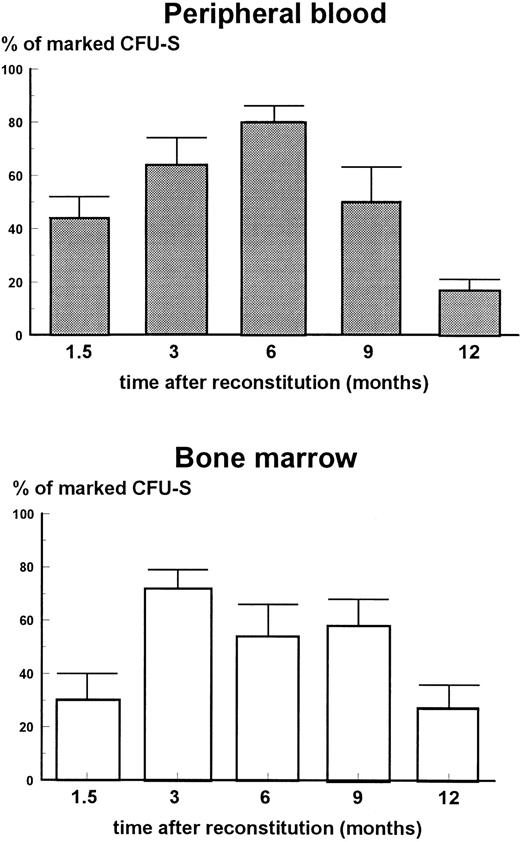
Proportion of marked CFU-S in bone marrow of mice reconstituted with peripheral blood mononuclears and bone marrow cells. Each column with error bar represents the mean ± standard error (SE). A total of 10 to 14 CFU-S were analyzed per group. Y-axis, % of marked CFU-S; X-axis, time after reconstitution).
Proportion of marked CFU-S in bone marrow of mice reconstituted with peripheral blood mononuclears and bone marrow cells. Each column with error bar represents the mean ± standard error (SE). A total of 10 to 14 CFU-S were analyzed per group. Y-axis, % of marked CFU-S; X-axis, time after reconstitution).
Self-Renewal Capacity of CFU-S in Bone Marrow of Reconstituted Mice
| Donor Cells . | Time After Reconstitution . | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| . | 1.5 mo . | 6 mo . | . | . | . | . | ||||
| . | Daughter CFU-S/Colony . | hADA Positive Colonies . | hADA Negative Colonies . | Daughter CFU-S/Colony . | hADA Positive Colonies . | hADA Negative Colonies . | . | . | . | . |
| . | All Colonies . | . | . | All Colonies . | . | . | . | . | . | . |
| Peripheral blood | 22.2 ± 3.8 | 20 | 25.2 ± 7.8 | 4.2 ± 0.9 | 7.4 ± 2.2 | 4.3 ± 3.5 | ||||
| Bone marrow | 16.2 ± 3.6 | 19.6 ± 4.1 | 12.2 ± 4.0 | 18.9 ± 8.1 | 14.2 ± 4.1 | 19.8 ± 15.3 | ||||
| Donor Cells . | Time After Reconstitution . | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| . | 1.5 mo . | 6 mo . | . | . | . | . | ||||
| . | Daughter CFU-S/Colony . | hADA Positive Colonies . | hADA Negative Colonies . | Daughter CFU-S/Colony . | hADA Positive Colonies . | hADA Negative Colonies . | . | . | . | . |
| . | All Colonies . | . | . | All Colonies . | . | . | . | . | . | . |
| Peripheral blood | 22.2 ± 3.8 | 20 | 25.2 ± 7.8 | 4.2 ± 0.9 | 7.4 ± 2.2 | 4.3 ± 3.5 | ||||
| Bone marrow | 16.2 ± 3.6 | 19.6 ± 4.1 | 12.2 ± 4.0 | 18.9 ± 8.1 | 14.2 ± 4.1 | 19.8 ± 15.3 | ||||
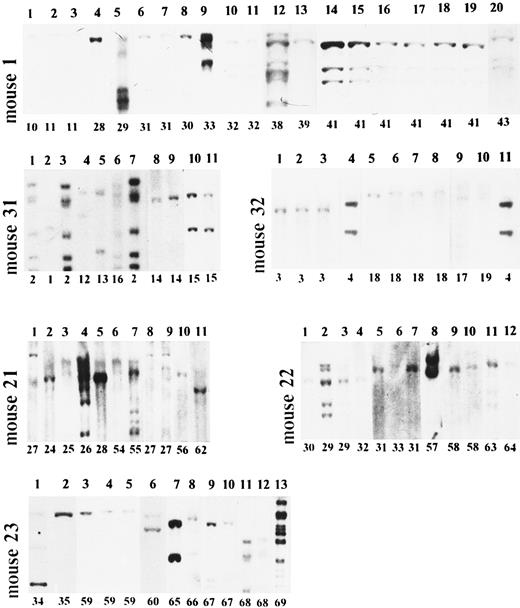
Long-term clonal behavior of engrafted hematopoietic stem cells. Analysis of six long-term reconstituted mice. DNA from CFU-S–derived spleen colonies was cleaved with EcoRI and analyzed by Southern blot. Mice no. 1, 31, and 32 were reconstituted with peripheral blood mononuclears; mice no. 21, 22, and 23 were reconstituted with bone marrow cells. Mouse no. 1, lanes 1 to 3 (clones 10 and 11 on Fig 3) - 6 months; lanes 4 to 11 (clones 28 to 32 on Fig 3) - 9 months; lanes 12 to 20 (clones 38 to 42 on Fig 3) - 12 months after reconstitution. Mouse no. 31, lanes 1 to 3 (clones 1 and 2 on Fig 3) - 3 months; lanes 4 to 11 (clones 2 and 12 to 16 on Fig 3) - 6 months. Mouse no. 32, lanes 1 to 4 (clones 3 and 4 on Fig 3) - 3 months; lanes 5 to 11 (clones 4 and 17 to 19 on Fig 3) - 6 months. Mouse no. 21, lanes 1 to 5 (clones 24 to 28 on Fig 4) - 6 months; lanes 6 to 10 (clones 27 and 54 to 56 on Fig 4) - 9 months; lane 12 (clone 62 on Fig 4 ) - 12 months. Mouse no. 22, lanes 1 to 7 (clones 29 to 33 on Fig 4) - 6 months; lanes 8 to 10 (clones 57 and 58 on Fig 4) - 9 months; lanes 11 to 12 (clones 63 and 64 on Fig 4) - 12 months. Mouse no. 23, lanes 1 and 2 (clones 34 and 35 on Fig 4) - 6 months; lanes 3 to 6 (clones 59 and 60 on Fig 4) - 9 months; lanes 7 to 13 (clones 65 to 69 on Fig 4) - 12 months after reconstitution.
Long-term clonal behavior of engrafted hematopoietic stem cells. Analysis of six long-term reconstituted mice. DNA from CFU-S–derived spleen colonies was cleaved with EcoRI and analyzed by Southern blot. Mice no. 1, 31, and 32 were reconstituted with peripheral blood mononuclears; mice no. 21, 22, and 23 were reconstituted with bone marrow cells. Mouse no. 1, lanes 1 to 3 (clones 10 and 11 on Fig 3) - 6 months; lanes 4 to 11 (clones 28 to 32 on Fig 3) - 9 months; lanes 12 to 20 (clones 38 to 42 on Fig 3) - 12 months after reconstitution. Mouse no. 31, lanes 1 to 3 (clones 1 and 2 on Fig 3) - 3 months; lanes 4 to 11 (clones 2 and 12 to 16 on Fig 3) - 6 months. Mouse no. 32, lanes 1 to 4 (clones 3 and 4 on Fig 3) - 3 months; lanes 5 to 11 (clones 4 and 17 to 19 on Fig 3) - 6 months. Mouse no. 21, lanes 1 to 5 (clones 24 to 28 on Fig 4) - 6 months; lanes 6 to 10 (clones 27 and 54 to 56 on Fig 4) - 9 months; lane 12 (clone 62 on Fig 4 ) - 12 months. Mouse no. 22, lanes 1 to 7 (clones 29 to 33 on Fig 4) - 6 months; lanes 8 to 10 (clones 57 and 58 on Fig 4) - 9 months; lanes 11 to 12 (clones 63 and 64 on Fig 4) - 12 months. Mouse no. 23, lanes 1 and 2 (clones 34 and 35 on Fig 4) - 6 months; lanes 3 to 6 (clones 59 and 60 on Fig 4) - 9 months; lanes 7 to 13 (clones 65 to 69 on Fig 4) - 12 months after reconstitution.
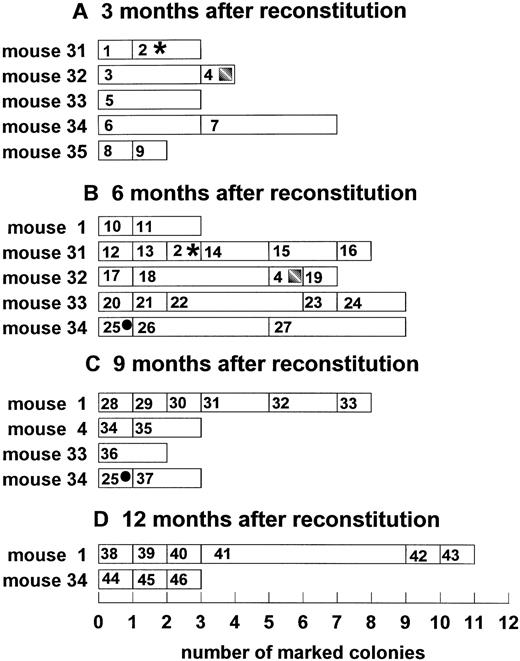
Temporal dynamics of clonal fluctuation in mice reconstituted with peripheral blood mononuclear cells. Each horizontal lane represents the number of spleen colonies with a unique integration site. The size of the rectangle indicates the number of spleen colonies with the same unique marker; rectangles with symbols indicate the persistent clones; numbers in rectangles indicate unique clone identification number. (A) Three months after reconstitution; (B) 6 months after reconstitution; (C) 9 months after reconstitution; (D) 12 months after reconstitution. Y-axis, reconstituted animals; X-axis, number of individual colonies.
Temporal dynamics of clonal fluctuation in mice reconstituted with peripheral blood mononuclear cells. Each horizontal lane represents the number of spleen colonies with a unique integration site. The size of the rectangle indicates the number of spleen colonies with the same unique marker; rectangles with symbols indicate the persistent clones; numbers in rectangles indicate unique clone identification number. (A) Three months after reconstitution; (B) 6 months after reconstitution; (C) 9 months after reconstitution; (D) 12 months after reconstitution. Y-axis, reconstituted animals; X-axis, number of individual colonies.
Temporal dynamics of clonal fluctuation in mice reconstituted with bone marrow cells. Each horizontal lane represents the number of spleen colonies with a unique integration site. The size of the rectangle indicates the number of spleen colonies with the same unique marker; rectangles with symbols indicate the persistent clones; numbers in rectangles indicate the unique clone identification number. (A) Six months after reconstitution; (B) 9 months after reconstitution; (C) 12 months after reconstitution. Y-axis, reconstituted animals; X-axis, number of analyzed individual colonies).
Temporal dynamics of clonal fluctuation in mice reconstituted with bone marrow cells. Each horizontal lane represents the number of spleen colonies with a unique integration site. The size of the rectangle indicates the number of spleen colonies with the same unique marker; rectangles with symbols indicate the persistent clones; numbers in rectangles indicate the unique clone identification number. (A) Six months after reconstitution; (B) 9 months after reconstitution; (C) 12 months after reconstitution. Y-axis, reconstituted animals; X-axis, number of analyzed individual colonies).
The survival of mice reconstituted with peripheral blood cells did not differ significantly from those transplanted with bone marrow cells (Table 2). Donor hematopoiesis was observed in both groups of reconstituted mice. Nine months after reconstitution, the PCR analysis showed that 75.3% ± 6.0% of CFU-S was Y-positive in the group transplanted with peripheral blood mononuclear cells. The same degree of donor CFU-S (62.8% ± 8.5%) was observed in bone marrow-transplanted animals. One year after engraftment the proportion of donor CFU-S decreased to 44.0% ± 9.0% and 42.2% ± 12.4%, respectively.
Bone marrow samples were collected at 1.5, 3, 6, 9, and 12 months after reconstitution from each mouse and the concentration of CFU-S, their self-renewal capacity, and percent of CFU-S marked with hADA gene were determined. It has been shown earlier that 2 to 12 months posttransplantation the concentration of CFU-S in bone marrow of mice reconstituted with 2 to 3 × 106 normal (not transduced) bone marrow cells was 150 to 200 CFU-S per 106 cells, which is 10-fold higher than that observed in the present experiment following the procedure of gene transfer (Table 3). PCR analysis of individual CFU-S–derived colonies showed that 1.5 to 9 months after reconstitution 30% to 80% of CFU-S were infected with hADA-retrovirus; 12 months after engraftment the proportion of transduced CFU-S decreased to 12% to 25% (Fig 1). The proportion of CFU-S transduced with hADA gradually reached a maximum at 3 to 6 months after reconstitution. At this time 75% to 80% of the CFU-S-11 contained hADA sequences. Thereafter, the percentage of transduced CFU-S decreased, particularly in mice reconstituted with peripheral blood cells, to reach 15% to 20% 1 year after reconstitution.
The influence of hADA sequence integration on the quality of CFU-S was studied by comparison of the proliferative potential of marked and unmarked CFU-S (Table 4). There was no difference in SRA of untransduced CFU-S and those that carried the foreign gene. SRA of bone marrow CFU-S in mice reconstituted with peripheral blood cells did not differ from SRA of initially injected CFU-S 1.5 months after transplantation. However, 4.5 months later a fivefold decrease of their SRA was observed. In contrast, SRA of bone marrow CFU-S from mice reconstituted with bone marrow cells was stable for 6 months.
The DNA from hADA positive colonies was also studied by Southern blot hybridization. This technique showed a different pattern of hADA integration in CFU-S–derived colonies (Fig 2). Integration of 1 to 13 hADA copies per genome can be seen. The unique sites of integration allowed the identification of individual clones. For 12 months, numerous individually marked CFU-S–derived clones were observed in the bone marrow of mice reconstituted both with peripheral blood mononuclear and bone marrow cells. The clonal composition of the CFU-S compartment from different reconstituted mice is shown in Figs 3 and 4. One to nine individual clones were functioning simultaneously. Clonal fluctuation was shown during 12 months of observation. Mono-oligoclonal hematopoiesis was shown mainly in mice reconstituted with peripheral blood mononuclear cells. It was observed in all 5 mice studied 3 months after reconstitution, in 1 mouse (no. 1) 6 months and in 3 mice (no. 4, 33, and 34) 9 months after engraftment (Fig 3). In mice reconstituted with bone marrow cells mono-oligoclonal, hematopoiesis was observed only rarely (no. 22 and no. 23 9 months and no. 21 12 months after engraftment) (Fig 4). Most of the animals had polyclonal hematopoiesis during the entire period of observation. The number of clones in an individual mouse changed with time and different clones, as a rule, were observed at different sampling times. The diversity of clones was significantly higher in mice reconstituted with bone marrow cells as compared with the peripheral blood group. Only 4 clones of 115 studied were carrying the same marker during a 3-month interval (clones 2, 4, and 25 in peripheral blood group, Fig 3, and clone 27 in bone marrow group, Fig 4). Persisting clones were detected in three of six mice reconstituted with peripheral blood cells and in only one mouse engrafted with bone marrow cells. Therefore, the persistence of clones seems to have been longer in the peripheral blood group.
DISCUSSION
This study represents the detailed longitudinal analysis and comparison of hematopoiesis in mice reconstituted with transduced peripheral blood mononuclear and transduced bone marrow cells. Long-term survival was essentially the same in both groups of reconstituted mice. The proportion of donor hematopoietic precursor cells did not differ during 9 months and decreased sharply 1 year after reconstitution, particularly in the peripheral blood group.
The data suggest that PBSC are a good alternative to bone marrow cells for transplantation. However, this conclusion comes with reservation. Self-renewal ability of CFU-S from mice reconstituted with bone marrow cells was stable for 6 months after transplantation, while in PBSC reconstituted mice, SRA decreased fivefold by the end of this period. The significance of this effect is not clear, as both types of cells reconstituted hematopoiesis equally well and no selective exhaustion of donor peripheral blood-derived CFU-S occurred during 12 months.
The concentration of CFU-S, as well as their SRA in bone marrow of mice reconstituted with transduced PBSC or bone marrow cells, decreased sharply as compared with mice engrafted with nontransduced bone marrow cells.27 It is unlikely that this defect of hematopoiesis is induced by intensive treatment of donors with cytokines because similar reduction in CFU-S concentration and their SRA was observed in mice reconstituted with transduced bone marrow cells from nontreated mice (manuscript submitted). The decrease of CFU-S proliferative potential, as measured by SRA assay, cannot be explained by proviral integration. The model of sequential analysis of individual CFU-S allowed the direct experimental study of this problem. There was no difference in the SRA of nontransduced CFU-S and those carrying foreign sequences (Table 4). More importantly, no difference was observed between CFU-S with three to 13 copies of hADA and CFU-S with only one to two copies. The proportion of CFU-S with provirus integration was relatively stable during 9 months (Fig 1), which suggests that transduced primitive HSCs have no proliferative disadvantage as compared with nontransduced HSCs. However, 1 year after reconstitution, the proportion of marked CFU-S among donor-derived CFU-S decreased significantly, particularly in the peripheral blood group. It is not clear whether such selective exhaustion of transduced HSCs is the result of integration of foreign DNA sequences. An alternative explanation could be that transduction is only possible into cell cycle active HSCs28 and such forced mobilization could have changed the potential of HSCs irreversibly.
It was shown that the developmental behavior of genetically marked stem cells is primarily a function of time, and that the first 4 months postengraftment are characterized by frequent clonal fluctuation. Gradually, however, a stable hematopoietic system emerges, dominated by a small number of clones.29-32 Here we report the first continuous analysis of hematopoietic clonal kinetics in the compartment of CFU-S. It was shown that hematopoiesis on the level of CFU-S is polyclonal during at least 12 months after reconstitution. CFU-S both in vivo33,34 and in vitro35 have only short-term repopulating ability. Hence, CFU-S analyzed 3 to 12 months after reconstitution represent progeny of primitive long-term repopulating cells, rather than CFU-S transduced in the initial cell inoculum. One to nine individual clones of simultaneously functioning hematopoietic progenitors have been observed in the bone marrow of mice reconstituted both with PBSC and bone marrow cells at least during 12 months (Figs 3 and 4). Taking into account that only a small number of CFU-S–derived colonies were analyzed for each sampling time and that the majority of them had unique site of integration, the number of simultaneously functioning clones may have reached several dozen. The number of functioning clones in mice reconstituted with bone marrow was higher as compared with PBSC, on the average, about 1.5-fold to 2-fold (Figs 3 and 4). It was also shown early by multinomial analysis of marked clones36 that after normal bone marrow transplantation, the number of simultaneously functioning clones is higher than was shown here after PBSC engraftment. This suggests that after a 2-week course of cytokine administration, the content of clonogeneic long-term repopulating cells in peripheral blood is somewhat lower than in bone marrow.
The life of a hematopoietic cell clone is about 3 months. Here we observed only four clones (of 115 studied) persisting for 3 months and none for 6 months (Figs 3 and 4). The clones, which disappeared, never returned. These findings, as well as data published elsewere,36 support the model of hematopoietic clonal succession, with limited proliferative potential of primitive HSC. However, the reservations are needed since persisting clones may be missed among numerous simultaneously existing clones.
In summary, we defined, by the novel technique, similarities and differences in the temporal behavior of individual stem cell clones derived from peripheral blood and bone marrow long-term repopulating cells. The general thesis of CFU-S clonal fluctuation without evolving into monoclonal stability needs reevaluation of the mechanisms of hematopoietic regulation. Further studies are in progress to understand the clonal succession model of stem cell use.
ACKNOWLEDGMENT
The authors gratefully acknowledge the help of Dr Dimitry Kuprash in choosing primers for PCR analysis of hADA and sex-determining region of Y-chromosome. We express our appreciation to Drs Regina Turetskaya, Sergey Nedospasov, and Sergey Sokol for helpful suggestions in molecular biology techniques. We also thank Dr D. Williams for providing us with the ADA-producing cell line.
Supported by Grants No. M5X300 and M5X00 from the International Science Foundation and Russian Fund for Fundamental Research (Moscow).
Address reprint requests to Axel Zander, MD, Universitat Hamburg, Universitats-Krankenhaus Eppendorf, II Medizinische Klinik, Abt. Onkologie/Hematologie, Martinistrasse 52, 20246 Hamburg 20, Germany.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal