Background: Definitive diagnosis of intraocular lymphoma (IOL) is often challenging, at least partially due to limited accessibility to the lesion and the rarity of tumor cells, with pathologically false-negative results reportedly occurring in approximately 30% of cases. Delayed diagnosis is often an important clinical issue, and the average time to diagnosis is approximately 1.5 years.Because of delayed diagnosis, IOLs are often found only after central nervous system (CNS) invasion has occurred, possibly leading to worse treatment outcomes. In this study, we tried to overcome diagnostic difficulties by using gene panel diagnostics of IOL. Furthermore, genetic analysis of IOLs may be able to predict the risk of recurrence.in Japan, a second-generation Bruton's tyrosine kinase (BTK) inhibitor, tirabrutinib, has been approved for the treatment of relapsed/refractory CNS lympoma. Herein, we present a case of IOL and CNS lesion with MYD88 mutation treated with tirabrutinib as part of precision medicine.
Methods: We have developed a digital polymerase chain reaction-based system for multiplex gene panel testing of cell-free DNA (cfDNA) obtained from intraocular liquid biopsies (patent number 7149000, 2022/9/30). Our system can detect specific gene mutations in minute cfDNA by amplifying specific gene loci in bubbles, even from an extremely small number of cells. Using this multiplex gene panel testing, we analyzed genetic mutations in nine regions of four genes, MYD88, CD79B, BTG2, and PIM1, using cfDNA obtained from intraocular liquid biopsies. The diagnostic performance of the novel system was evaluated in 86 samlpes of patients with IOL. We also retrospectively analyzed the prognostic potential of the genetic mutations for IOL. Lastly, in the case of CNS lymphoma and IOL, we presented the MYD88 mutation treated with tirabrutinib.
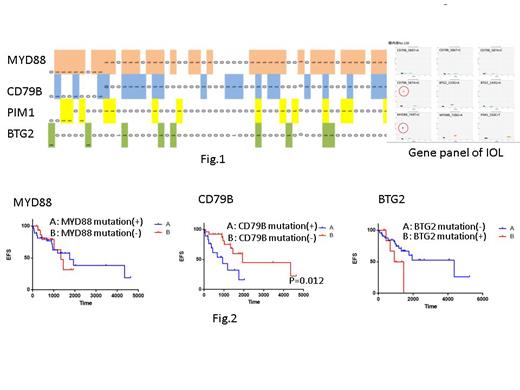
Results: Among the four disease-specific genes, the MYD88 mutation was present in 55 of 86 cases (64%), and frequencies of the CD79B, BTG2, and PIM1 mutations were 47%, 38%, and 29%, respectively. At least one of these mutations was found in 83 of the 86 IOL cases examined (97%), making this assay highly sensitive (Fig. 1). The MYD88 mutation was not associated with recurrence in patients with recurrent and refractory CNS lymphoma and IOL. Conversely,patients with the CD79B mutation had high rates of relapse for CNS lymphoma: the 4-year rate of recurrence-free survival was 35%, compared to 79% in patients without the CD79B mutation (p=0.012). Patients with the BTG2 mutation also had high rates of relapse (Fig. 2). Furthermore, we present a case of CNS lynphoma and IOL. By testing IOL gene panel, only MYD88 mutation was detected in the vitreous sample of IOL patient. We have experienced this case in which the second-generation BTK inhibitor,tirabrutinib, was effective not only for central but also for intraocular lesions.
Conclusion: Our panel enables rapid and accurate diagnosis of IOL with only a small sample of intraocular liquid. We believe that this multiplex gene panel testing may aid in the diagnosis of IOL in difficult-to-diagnose patients, including those with chronic uveitis, by preventing CNS invasion due to diagnostic delay. Moreover, genetic profiling panels of IOL can help to stratify clinical outcomes.CD79B and BTG2 mutation is a potent predictive marker for relapse. In the clinical setting, we experienced a case of IOL with MYD88 mutation in which tirabrutinib was effective. Potentially, tirabrutinib also penetrates directly into the eye in intraocular lesions. Oral drug, tirabrutinib as eye dripping drug, might be effective as a therapeutic option for IOL and should be assessed in future clinical trials of precision medicine for IOL.
Disclosures
Kurokawa:MSD K.K.: Honoraria; Asahi Kasei Pharma Corporation: Honoraria, Research Funding; Astellas Pharma Inc.: Honoraria; AstraZeneca K.K.: Research Funding; AbbVie GK: Honoraria, Research Funding; Amgen Inc.: Honoraria; Eisai Co., Ltd.: Honoraria; Otsuka Pharmaceutical Co., Ltd.: Honoraria, Research Funding; ONO PHARMACEUTICAL CO., LTD.: Honoraria; Kyowa Kirin Co., Ltd.: Honoraria, Research Funding; Sanofi K.K.: Honoraria; Sumitomo Pharma Food & Chemical Co., Ltd.: Honoraria, Research Funding; DAIICHI SANKYO COMPANY, LIMITED: Honoraria, Research Funding; Takeda Pharmaceutical Company Limited.: Honoraria, Research Funding; Nippon Kayaku Co., Ltd.: Honoraria; Nippon Shinyaku Co., Ltd.: Honoraria; Pfizer Japan Inc.: Honoraria; Bristol-Myers Squibb K.K.: Honoraria; MOCHIDA PHARMACEUTICAL CO.,LTD.: Honoraria; Janssen Pharmaceutical K.K.: Honoraria; Shionogi & Co., Ltd.: Research Funding; Teijin: Research Funding; Chugai Pharmaceutical Co., Ltd.: Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal