In the 2022 European Leukemia Net (ELN) risk classification, acute myeloid leukemia (AML) with translocation t(8;16) was newly classified as AML defined by specific cytogenetic abnormalities, associated with unfavorable prognosis. Translocation (8;16)(p11;p13) is a rare abnormality in AML with female preponderance and occurs more frequently in secondary AML (sAML), especially therapy-related, than in de novo disease. Clinically, AML with t(8;16) is associated with high risk of bleeding, occurrence of extramedullary manifestations, myelomonocytic differentiation, and signs of hemophagocytosis in the bone marrow. The translocation leads to the fusion of acetyltransferase KAT6A (also known as MOZ or MYST3) and CREB binding protein ( CREBBP) genes, both of them involved in hematopoiesis.
ELN risk stratification of AML with t(8;16) was based on studies that included relatively low numbers of patients receiving allogeneic stem cell transplantation (alloSCT). In the largest report by Kayser et al. ( Br J Haematol 2021) 21 patients (15 transplanted in first complete remission [CR1], median follow-up 5.5 years) were analyzed. A 5-year overall survival (OS) of 38% was achieved after alloSCT compared to 11% in patients who received chemotherapy only.
The aim of our study was to evaluate the outcome of patients with AML harboring t(8;16) after alloSCT in a larger cohort from the EBMT registry. All types of donor, conditioning regimen and graft were included. Sixty patients were identified, of whom 44 had been transplanted in CR1. Median follow-up from alloSCT was 72.3 months, median age was 46.3 years, and 73.3% were female. Karnofsky performance status was >80% in 59 patients, one had missing data.
At 2 years after alloSCT, OS and leukemia free survival (LFS) of the entire cohort were 42.7% and 42.2%, respectively, (28.6% and 14.3% in patients transplanted in advanced disease). Cumulative incidence of relapse (RI) and non-relapse mortality (NRM) at two years was 36.9% and 15.3%, respectively, in patients transplanted in CR1, and 78.6% and 7.1%, respectively, in patients transplanted in advanced disease.
The 44 patients transplanted in CR1 were analyzed in detail. Basic characteristics were comparable to the entire cohort as described above. Twenty-two of the patients had de novo, 22 had sAML. According to the ELN 2017 classification (not yet including t(8;16) as a factor for poor prognosis), 54.5% had an intermediate and 45.5% an adverse cytogenetic risk. Furthermore, 38.8% had a complex karyotype. Donors were matched related in 34%, matched unrelated in 54%, haploidentical in 5% and cord blood in 7%. Conditioning was myeloablative in 52.3%, whereas 47.7% had received reduced intensity conditioning.
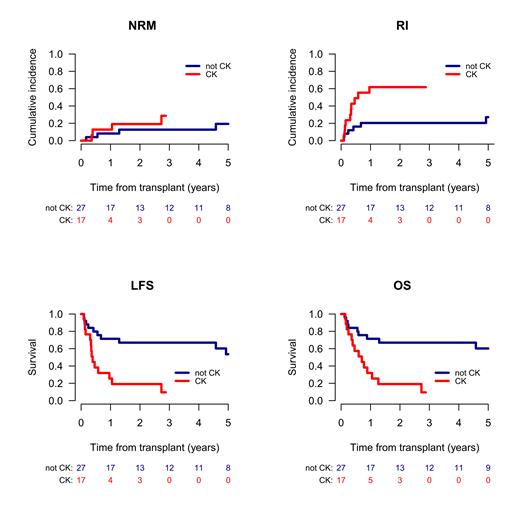
Among patients transplanted in CR1, 2- and 5-year OS from alloSCT was 48% and 39.9%, the respective LFS was 46.8% and 35.5%. On multivariate analysis (MVA), harboring t(8;16) within a complex karyotype was the major risk factor for outcome, being associated with higher RI (hazard ratio [HR] 3.38, p=0.01), lower LFS (HR 4.17, p=0.016), and lower OS (HR 3.08, p=0.017). In contrast, patients with t(8;16) outside of a complex karyotype achieved excellent results (Figure 1). Besides a complex karyotype, MVA revealed sAML as an additional, independent risk factor with a higher RI (HR 3.73, p=0.026). Age was the major factor for NRM, reaching 5% vs. 26.5% in patients below or above the median (p=0.02).
In conclusion, according to the largest series analyzed in this setting so far, overall results after alloSCT in AML with t(8;16) transplanted in CR1 were favorable, especially in patients without multiple additional cytogenetic abnormalities, achieving a low risk of relapse and a 5-year OS >60%. Hence, alloSCT in CR1 appears to abrogate the unfavorable prognostic value of this translocation. Whether or not t(8;16) does have any additional influence on the outcome of patients with a complex karyotype remains to be evaluated.
Figure Legend
Figure 1: Outcome of AML with t(8;16) with and without complex karyotype
Disclosures
Versluis:AbbVie: Honoraria; ExCellThera: Consultancy. Chevallier:Mallinckrodt Pharmaceuticals: Honoraria; Sanofi: Honoraria; Incyte: Honoraria, Research Funding; Takeda: Honoraria; Immedica Pharma: Honoraria; Servier: Honoraria. Forcade:Novartis: Consultancy, Other: Travel support, Speakers Bureau; Alexion: Other: Travel support, Speakers Bureau; Astellas: Speakers Bureau; Gilead Sciences: Other: Travel support, Speakers Bureau; GSK: Speakers Bureau; Sanofi: Speakers Bureau; MSD: Other: Travel support. Baron:ExCellThera Inc: Consultancy; Takeda: Honoraria; Incyte Biosciences: Consultancy. Bug:Gilead: Honoraria, Other: travel grant; BMS: Honoraria; Novartis: Honoraria; Jazz: Honoraria, Other: travel grant; Neovii: Other: trvel grant; Pfizer: Honoraria. Esteve:Abbvie: Consultancy; Jazz Pharmaceuticals: Consultancy, Research Funding; Kronos Bio: Research Funding; Astellas: Consultancy; Gilead: Consultancy; Pfizer: Research Funding. Ciceri:ExCellThera: Other: Scientific Advisory Board . Mohty:JAZZ PHARMACEUTICALS: Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal