Background: Antibody-drug conjugates (ADCs) are an effective class of cancer therapeutics, but current ADCs face stability challenges, resulting in premature payload release and off-target toxicity. To overcome this, we developed a novel strategy using antibody-polymer-based drug conjugates (pADCs). Our pADCs have a number of advantages compared to currently FDA approved ADCs, including: 1) Increased drug payload without altering binding site numbers, ensuring maximum drug loading without affecting antigen and Fc binding; 2) Potential for synergistic immunotherapy and chemotherapy; 3) Improved pharmacokinetics and reduced immunogenicity with modification using water-soluble N-(2-hydroxypropyl)methacrylamide (HPMA) copolymer, providing significant stealth effect; and 4) Enhanced stability under physiological conditions, reducing premature payload release and off-target toxicity. Panobinostat, a pan-HDAC (histone deacetylase) inhibitor, enhances CD38 expression and improves pADC efficiency against MM cells. Our hypothesis is that our pADC system induces strong cytotoxicity, further enhanced by panobinostat-induced CD38 upregulation, leading to more efficient treatment outcomes.
Methods:mAbs decorated with cyclooctyne (DBCO) moieties in the hinge region were prepared in two steps: mAbs were reduced with tris(2-carboxyethyl)phosphine (TCEP), then thiol groups were capped with DBCO-PEG4-Mal. Azide-functionalized semitelechelic (ST) polymer-drug conjugates (ST-P-drug-N3) were synthesized via RAFT polymerization. Antibody-polymer-drug conjugates were prepared by CLICK chemistry. MM.1S and RPMI 8226 cell lines (ATCC) were used as the MM cell model. The cell internalization was revealed by confocal imaging. Primary MM cells were collected from the aspirate of eight patients with primary plasma cell leukemia and isolated via Ficoll centrifugation. CD38 upregulation was done by incubating with 10 or 25 nM panobinostat for 24 h and the apoptosis detection employed Annexin V and Propidium Iodide staining followed by flow cytometry analysis.
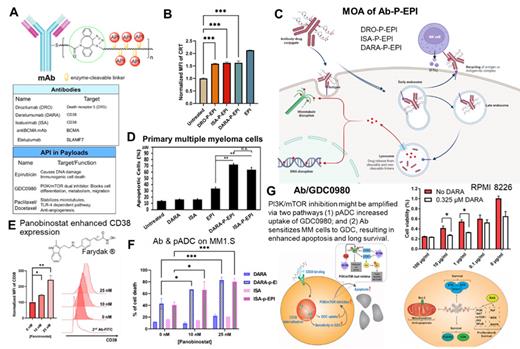
Results: A series of CLICK-able mAbs and semitelechelic polymer-drug conjugates were synthesized. Among them, we firstly prepared Epirubicin (EPI)-based pADCs, such as DARA-P-EPI, ISA-P-EPI and DRO-P-EPI (Fig.1A). Unlike naked mAbs, pADCs can induce EPI concentration-dependent cytotoxicity. Surface calreticulin (CRT) was assessed and demonstrated the induction of EPI-dependent immunogenic cell death on MM.1S cells (Fig.1B). The pADC mechanism of action involves binding to cell surface antigens, internalization via endosomes, and subsequent cleavage of drugs at lysosomes by cathepsin B. This minimizes premature drug release and reduces off-target toxicity (Fig.1C). Ex vivo studies on patient MM cells revealed that pADCs induced significantly higher apoptosis compared to naked mAbs and free EPI, highlighting their efficacy (Fig.1D). CD38-based pADC's efficacy was enhanced by pretreatment with panobinostat (Fig.1F), which increased CD38 expression (Fig.1E). When incubating with NK cells and pADC, the number of target cells decreased significantly indicating the pADC preserve the original mAb ADCC functionality. Notably, co-treatment of RPMI8226 cells with anti-CD38 and GDC0980, a PI3k/mTOR inhibitor, significantly improved cytotoxicity, suggesting DARA's potential for chemo sensitization (Fig.1G).
Conclusion: pADC's innovative design offers a high likelihood of successful clinical development and holds significant promise for MM therapy. Moreover, the unique mechanism provides potential to effectively overcome mAb and multiple drug resistance. Its facile nature allows for easy incorporation of multiple types of payloads into the ADC, making its synthesis highly scalable .
Disclosures
Sborov:Adaptive Biotech: Other: Payment for an educational seminar; Sanofi: Consultancy; Abbvie: Membership on an entity's Board of Directors or advisory committees; Bristol Myer Squibb: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; BinayTara Foundation: Other: Support for attending meetings and/or travel; Karyopharm Therapeutics: Membership on an entity's Board of Directors or advisory committees; Arcellx: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; GSK: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal