Introduction:
Enasidenib is an FDA approved selective-inhibitor of mutant IDH2 ( mIDH2) enzyme commonly found in acute myeloid leukemia (AML). Previous studies found enasidenib unexpectedly improved red blood cell and platelet transfusion needs in mIDH2 AML patients without a reduction in AML disease burden (Pollyea DA, et al. Leukemia 2019; 33(11):2575-2584; DiNardo CD, et al. Lancet Oncol 2021; 22(11):1597-1608). We subsequently demonstrated enasidenib robustly enhanced human erythropoiesis from umbilical cord-blood and bone marrow-derived CD34+ hematopoietic progenitor cells independent of IDH2 status and the oncometabolite R-2-hydroxyglutarate (R-2-HG), thus prompting us to investigate enasidenib as an erythropoiesis-stimulating agent (Dutta R, et al. J Clin Invest 2020; 130(4):1843-1849).
Aim:
To evaluate the safety, tolerability, and efficacy of daily enasidenib in patients with lower-risk MDS or nonproliferative CMML without IDH2 mutations.
Methods:
In this ongoing two-part phase 1b/2 single-center, investigator-initiated, open-label trial, patients with a diagnosis of IDH2 wildtype lower risk MDS and nonproliferative CMML self-administered continuous daily dosing of enasidenib. Two dose levels were planned at 100mg and 200mg daily for Cohort A and B, respectively based on a standard 3 x 3 dose escalation schema. All patients were evaluated for adverse events (AEs) to determine dose-limiting toxicities (DLTs) and establish the recommended phase 2 dose (RP2D). Patients were screened in a consecutive manner to reduce selection bias by the investigators and all consents were translated into participant's native language to ensure full understanding and voluntary participation. Eligible patients are 18 years or older, ECOG ≤3, negative for IDH2 mutation, have adequate organ function, and symptomatic anemia (hemoglobin <10.5g/dL). Other causes such as iron/nutritional deficiencies and bleeding were ruled out during screening. Prior treatment with ESA, G-CSF, and luspatercept was allowed with a 30-day washout period. Each cycle is 28 days. Patients were evaluated by the PI on Day 1 (±3 days), and laboratory evaluation was conducted every 15 ±3 days.
Results:
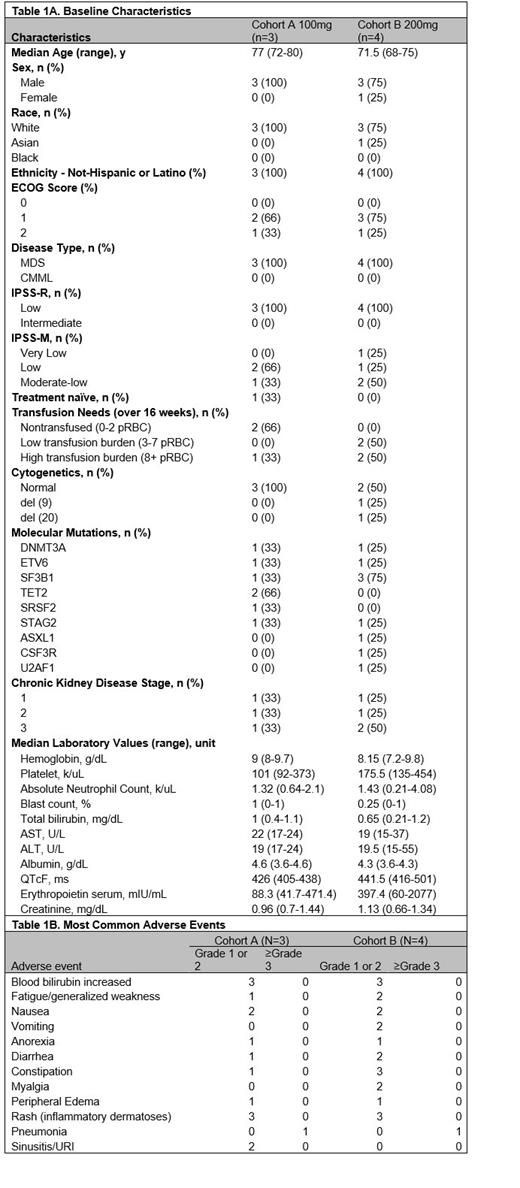
Seven patients were enrolled between July 6, 2022 and December 21, 2022 with three patients in Cohort A receiving 100mg and 4 patients in Cohort B receiving 200mg (Table 1A). No DLTs were seen during the first 6 cycles of treatment. One treatment was discontinued due to the need for treatment of vasculitides related to a new diagnosis of VEXAS made during cycle 2 of the trial (unrelated to study drug). Another patient discontinued treatment after 1 cycle to pursue allogeneic stem cell transplant. All patients experienced at least one treatment-related adverse event (TRAE) which were limited to grade 1 or 2. TRAEs noted were consistent with those previously reported with enasidenib use (Stein EM, et al. Lancet Haematol 2020; 7(4):e309-e319), most commonly hyperbilirubinemia observed in 6 patients (85.7%), nausea in 4 patients (57.1%), and rash (inflammatory dermatoses, previously well-described) in 5 patients (71.4%) (Table 1B) (Parisi R, et al. Leuk Res 2022; 123:106970). No grade 3 or higher TRAE were observed. Two patients (28.7%) experienced a Grade 3 adverse event of pneumonia. Four patients developed an infection (57.1%) with only one requiring a hospitalization. One grade 4 event occurred (duodenal hemorrhage) in a patient with history of duodenal ulcers and concurrent aspirin use. As above, a patient in Cohort A was diagnosed with VEXAS syndrome after enrollment and experienced a complicated course of vasculitis and opportunistic infections in the setting of high dose steroid use. Interestingly, one newly diagnosed patient in the 200mg Cohort B without prior growth factor or ESA is enjoying a sustained trilineage improvement with increase of absolute neutrophil count, hemoglobin, and platelets from baseline (10 cycles at the time of data cutoff of June, 16, 2023).
Conclusions:
Enasidenib at 200mg dose was safe and well tolerated in patients with lower-risk MDS. Thus far, one patient has seen trilineage improvement in blood counts and continues on study. The Phase 2 portion of the study is ongoing to further characterize safety and evaluate for efficacy to improve anemia and decrease transfusion needs.
Disclosures
Mannis:Genentech: Consultancy; BMS/Celgene: Consultancy; Astellas: Consultancy; Macrogenics: Honoraria; Agios: Consultancy; Abbvie: Consultancy; Stemline: Consultancy. Majeti:858 Therapeutics: Membership on an entity's Board of Directors or advisory committees; MyeloGene: Current equity holder in private company; Pheast Therapeutics: Current equity holder in private company; Orbital Therapeutics: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; kodikaz Therapeutic Solutions: Membership on an entity's Board of Directors or advisory committees. Zhang:Bristol Myers Squibb: Research Funding; Servier: Consultancy; Rigel: Consultancy; Abbvie: Consultancy; Stanford University: Current Employment.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal