Abstract
INTRODUCTION: Allogeneic hematopoietic stem cell transplantation (allo-HSCT) provides the best and possibly only chance for cure for acute myeloid leukemia (AML). In preparation for allo-HSCT, patients undergo conditioning with chemotherapy and/or irradiation to deplete host hematopoietic stem cells (HSCs), suppress the host immune system, and destroy residual leukemia cells. However, conditioning associated toxicities impose a formidable barrier to patients undergoing this potentially curative therapy. Antibody-drug conjugates (ADCs) have emerged as a promising and potentially safer alternative for allo-HSCT conditioning by allowing targeted depletion of the host HSC niche. Previously, we used saporin-conjugated CD45- or cKit-ADCs as the backbone of a novel conditioning regimen which enabled engraftment between fully mismatched mice. However, saporin ADCs were nonmyeloablative and showed modest antitumor activity. We hypothesized that fully myeloablative ADCs capable of enhanced antileukemia activity could be produced using alternative toxic payloads to saporin. However, direct conjugation of multiple candidate payloads and/or antibodies is costly, time-consuming, and laborious. Here, we describe the production of a panel of streptavidin (SAv)-drug conjugates which allows efficient ADC generation from any biotinylated protein. This flexible platform enabled the rapid production of CD45-ADCs conjugated to multiple toxic payloads, facilitating in vitro screening of their ability to target HSCs and leukemia cells.
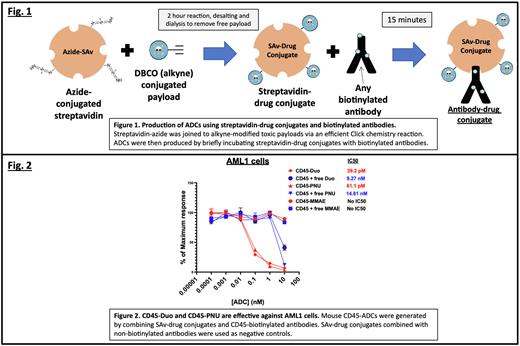
METHODS: Azide-alkyne cycloaddition ("Click" chemistry) was used to covalently link SAv-azide (Protein Mods, Inc) with dibenzocyloocytne (DBCO)-coupled drug payloads (Levena Biopharma) (Fig. 1). We conjugated three toxic payloads to SAv for these studies: monomethyl auristatin E (MMAE), Duocarmycin SA (Duo), and PNU-159682 (PNU). All of these payloads contain cleavable valine-citrulline linkers for payload release following ADC internalization. For the conjugation reactions, SAv-azide was combined with DBCO-coupled payload and incubated 2 hours at 20oC, with 20% DMSO added as cosolvent. Free payload and DMSO were removed using size exclusion chromatography and dialysis. ADCs were freshly prepared before use by mixing SAv-drug conjugates in a 1:1 molar ratio for 15 minutes with biotinylated antibodies. For our initial studies, we evaluated murine anti-CD45.2 (Clone 104) and human anti-CD45 (Clones BC8 and HI30). Non-biotinylated antibodies combined with SAv-drug conjugates were used as negative controls. All ADCs were tested for cytotoxicity in vitro against 1) murine YAC-1 (mCD45.2+) and human Jurkat (mCD45.2-) cell lines using XTT viability assays, 2) primary HSCs from B6 mice using methylcellulose assays, and 3) AML1 primary murine leukemia cells (Dnmt3aR878H/+ and FLT3-ITD; provided by Dr. Tim Ley) using methylcellulose assays. IC50 values for each assay were derived with nonlinear regression of dose response data.
RESULTS: Mouse CD45-Duo and CD45-PNU were highly effective at killing AML1 cells (IC50 values: 39.2pM and 61.1pM, respectively; Fig. 2), as well as B6 HSCs (IC50 values: 0.85nM and 0.13nM, respectively) in vitro. Notably, mouse CD45-MMAE was ineffective against YAC-1 cells, B6 HSCs, and AML1 cells, suggesting that MMAE is not an effective payload for ADC-based conditioning in the mouse. Importantly, nonbiotinylated antibodies mixed with SAv-drug conjugates were largely ineffective, indicating the need for the antibody to bind the SAv-drug conjugate to achieve specific toxicity. Preliminary studies using human CD45-MMAE and CD45-Duo demonstrated that while both payloads could yield ADCs able to kill Jurkat cells, ADCs made using human antibody clone BC8 were considerably more effective than ADCs made with clone HI30.
CONCLUSIONS: Our novel SAv-drug conjugate successfully enabled rapid generation of ADCs with a number of potential toxic payloads. This system identified multiple effective candidate payloads for use in mouse and human ADCs for targeting HSCs and leukemia cells in vitro and in vivo. Our ongoing and future studies with this system are focused in three areas: 1) production and testing of mouse and human cKit ADCs, 2) using mouse ADCs to condition for murine HSCT and target AML1 cells in vivo, and 3) evaluating killing of human cord blood-derived HSCs and patient-derived AML cells with human ADCs.
Disclosures
DiPersio:Amphivena Therapeutics: Research Funding; NeoImmune Tech: Research Funding; Macrogenics: Research Funding; BioLineRx, Ltd.: Research Funding; CAR-T cell Product with Washington University and WUGEN: Patents & Royalties; VLA-4 Inhibitor with Washington University and Magenta Therapeutics: Patents & Royalties; hC Bioscience, Inc.: Membership on an entity's Board of Directors or advisory committees; RiverVest Venture Partners: Consultancy, Membership on an entity's Board of Directors or advisory committees; Incyte: Consultancy, Research Funding; WUGEN: Current equity holder in private company, Research Funding; Magenta Therapeutics: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal