Abstract
Introduction and preclinical rationale: Patients with primary and secondary myelofibrosis (MF) have a reduced life expectancy compared to age-matched controls. The prognosis of individual patients is defined by clinical, cytogenetic, and molecular disease characteristics. Several oral Janus Kinase (JAK)-1/2 inhibitors such as ruxolitinib are approved by the US FDA for the treatment of patients with intermediate or high-risk MF based on improvements in symptom burden and spleen volume reduction compared to physician choice of best available therapy. However, these inhibitors often have a limited duration of response, and survival in patients after ruxolitinib discontinuation is poor (median survival of 13 months). Novel mechanism-based therapies for patients with suboptimal response or progression on ruxolitinib are thus needed.
Preclinical studies have demonstrated that JAK2 V617F mutations promote transition from G1 to S-phase of the cell cycle via increased expression of CDC25A. Notably, CDC25A expression has been shown to be upregulated in primary MPN patient samples. Additional cell cycle regulators such as CDK6 and Cyclin D have also been demonstrated to play a role in MPN pathogenesis and to drive myeloproliferation. Preclinical studies testing the combination of the CDK4/6 inhibitor ribociclib and ruxolitinib demonstrated reduction in spleen and liver size when compared to ruxolitinib alone in murine models, suggesting synergism (Rampal et al., Clinical Cancer Research 2021).
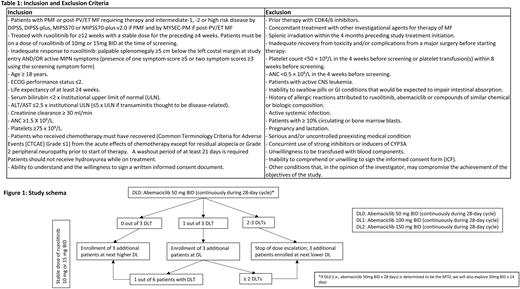
Methods: Based on these promising preclinical data, we hypothesize that combining ruxolitinib with a CDK4/6 inhibitor could have synergistic effects in patients with primary or secondary MF who have an inadequate response to ruxolitinib monotherapy. In this multicenter, phase I dose-escalation trial we evaluate the safety of combination therapy with ruxolitinib and the CDK4/6 inhibitor abemaciclib. Patients with primary or secondary MF with intermediate-1/2 or high-risk disease by DIPSS who require treatment and had an inadequate response to ruxolitinib are eligible for enrollment. An inadequate response will be defined by (I) palpable splenomegaly ≥5 cm below the left costal margin at study entry AND/OR (II) active MPN symptoms [presence of one symptom score ≥5 or two symptom scores ≥3 using the screening MPN-SAF TSS (Emanuel at al. JCO 2012). (Table 1). This study follows a conventional "3+3” dose-escalation design, in which patients on a stable dose of ruxolitinib (10mg or 15mg BID) with sufficient baseline platelet and absolute neutrophil count will be treated with increasing doses of abemaciclib. The pre-study dose of ruxolitinib will be maintained to determine the maximum tolerated dose of the combination of ruxolitinib and abemaciclib (Figure 1). The primary endpoint of this study is to determine the safety and tolerability, and to identify the recommended phase II dose of abemaciclib in MF patients on a stable, standard dose of ruxolitinib. Secondary endpoints include overall response rate (defined as a composite of complete remission [CR], partial remission [PR], and clinical improvement [CI; defined as anemia, spleen or symptom response]), rates of CR, PR, and CI as well as progression-free survival (PFS), overall survival (OS), and duration of response (DOR) as defined in accordance with the modified 2013 consensus definitions proposed by the International Working Group - Myeloproliferative Neoplasms Research and Treatment (IWG-MRT) and the European Leukemia Net (ELN).
As part of this clinical trial several correlative studies are planned to identify biomarkers predictive of response to ruxolitinib and abemaciclib as well as mechanisms of resistance (e.g., MAP kinase pathway activation). These include evaluation of changes in variant allele fraction of driver mutations, gene expression profile, cell cycle, clonal architecture, cytokine profile, and bone marrow fibrosis, aiming to inform patient selection as well as other alternative combination therapies for future clinical trials.
Disclosures
Verstovsek:Celgene: Consultancy, Research Funding; Sierra Oncology: Consultancy, Research Funding; AstraZeneca: Research Funding; Blueprints Medicines Corp.: Research Funding; Protagonist Therapeutics: Research Funding; NS Pharma: Research Funding; Novartis: Consultancy, Research Funding; Roche: Research Funding; Incyte: Consultancy, Research Funding; Gilead: Research Funding; ItalPharma: Research Funding; Promedior: Research Funding; PharmaEssentia: Research Funding; Constellation Pharmaceuticals: Consultancy; Pragmatist: Consultancy; Genentech: Research Funding; CTI BioPharma Corp.: Research Funding. Pemmaraju:stemline: Consultancy; abbvie: Consultancy; immunogen: Consultancy; mustangbio: Research Funding; incyte: Consultancy; novartis: Research Funding; pacylex: Consultancy, Research Funding; samus: Research Funding; daiichi sankyo: Research Funding; cellectis: Research Funding; cellularity: Research Funding. Stein:Auron Therapeutics: Current equity holder in private company; PinotBio, Bristol Myers Squibb, Jazz Pharmaceuticals, Foghorn Therapeutics, Blueprint Medicines, Gilead Sciences, Janssen Pharmaceuticals: Consultancy; Daiichi-Sankyo, Celgene Pharmaceuticals, and Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Astellas Pharmaceutical, Agios Pharmaceuticals, and Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees; PTC Therapeutics and Syros: Membership on an entity's Board of Directors or advisory committees; Syndax: Consultancy, Research Funding; Bayer: Research Funding; Amgen, AbbVie, Seattle Genetics, and Biotheryx: Consultancy. Mauro:AbbVie, Bristol Myers Squibb, Novartis, Pfizer, Takeda: Consultancy, Honoraria, Other: Travel, accommodation, expenses , Research Funding; Sun Pharma/SPARC: Research Funding. Rampal:Galecto: Consultancy, Membership on an entity's Board of Directors or advisory committees; Sunimoto Dainippon: Consultancy; Novartis: Consultancy; Stemline: Consultancy, Research Funding; Zentalis: Consultancy, Research Funding; Stemline: Consultancy, Research Funding; Kartos: Consultancy, Membership on an entity's Board of Directors or advisory committees; PharmaEssentia: Consultancy; Disc Medicines: Consultancy; Sierra Oncology: Consultancy; Gilead: Consultancy; Jazz Pharmaceuticals: Consultancy; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Blueprint: Consultancy; CTI: Consultancy; Promedior: Consultancy; Celgene/BMS: Consultancy; Incyte: Consultancy, Research Funding; Constellation Pharmaceuticals, Inc., a MorphoSys Company: Consultancy, Research Funding. Bose:Constellation Pharmaceuticals, Inc., a MorphoSys Company: Honoraria, Research Funding; Ionis: Research Funding; Kartos: Research Funding; Cogent: Honoraria, Research Funding; AbbVie: Consultancy; Sierra Oncology (now GSK): Consultancy; Pfizer: Research Funding; Astellas: Research Funding; Karyopharm: Consultancy; BMS: Consultancy, Research Funding; Blueprint Medicines Corporation: Honoraria, Research Funding; Disc Medicine: Research Funding; NS Pharma: Research Funding; Telios: Research Funding; Promedior: Research Funding; Pharma Essentia: Honoraria; Novartis: Honoraria; CTI BioPharma: Honoraria, Research Funding; Incyte: Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal