Abstract
Background CD19 targeted CAR T cell therapy (CAR-T) can lead to B cell aplasia, and CAR-T patients often have few protective antibodies (Abs) against polysaccharide antigens such as Streptococcus pneumoniae (Walti et al, JCI Insight, 2021). We previously reported that vaccinating CAR-T patients with PCV13, pneumococcal 13-valent conjugate vaccine, did not induce humoral protection when given at 3- or 6-months post therapy (Lee et al, EBMT, 2021). Subsequently, some patients developed humoral protection following vaccination at day+360 or maintained seroprotective IgG Abs present at baseline. Here we present interim results of a Simons two-stage design phase II trial (NCT04745559) testing the hypothesis that PCV13 given before and after CAR-T will induce humoral protection against pneumococcus among CAR-T patients.
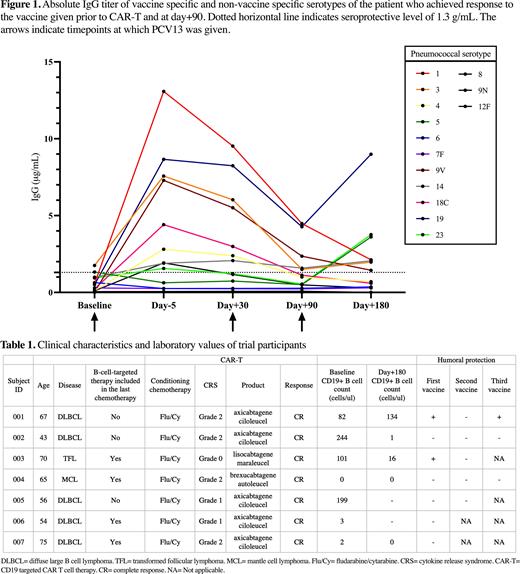
Method Eligible patients were ≥18 years old with large B cell lymphoma (LBCL) and under consideration for CAR-T. All patients received PCV13 7 days (range, 4-21) before apheresis and again at day+30 (range, 21-37) and day+90 (range, 75-115) post infusion. Pre- and post-serum pneumococcal IgG Abs were drawn to assess humoral response. Pneumococcal serotypes tested include those specific (1, 3, 4, 5, 6B, 7F, 9V, 14, 18C, 19F, and 23F) and non-specific (8, 9N, 12F) to PCV13 vaccine. An established serotype specific IgG concentration ≥1.3µg/mL was considered protective, and a patient with ≥6/11 tested vaccine specific serotypes meeting this threshold was deemed to have long-term humoral protection against pneumococcus. Descriptive analysis was conducted for patient characteristics and paired nonparametric t-test was performed to compare absolute IgG Abs between pre-and post-vaccination timepoints.
Results As of 7/01/2022, 7 enrolled patients were evaluable for immune response to vaccine. Median age was 65 (range, 43-75). Four patients received prior autologous stem cell transplant and had history of PCV vaccination. At baseline, only 1 out of 7 patients met the criteria of humoral protection. Three patients had CD19+ B cell count ≤5 cells/ul, and 1 patient had IgG level <400mg/dl.
Seven patients received the first PCV13 before apheresis. Out of 6 patients, who did not have humoral protection at baseline, 2 patients achieved humoral protection prior to the start of conditioning chemotherapy. The patient with humoral protection at baseline had vaccine specific IgG Abs remain similar between these timepoints (P=0.99).
One patient withdrew from the study before day+30, after developing ICANS grade 3. Six patients proceeded to receive the second dose of PCV13, 5 with paired Abs at day+30 and day+90. Two patients who achieved humoral protection with the first vaccine maintained humoral protection at day+30, and one of them subsequently lost protection at day+90. Three patients who did not have humoral protection since baseline did not mount humoral protection at day+90, after the second vaccine was administered at day+30.
Before day+90, 1 patient contracted COVID infection and did not receive the third vaccine. Five patients proceeded to receive the third PCV13 at day+90, and of these patients, 3 patients had paired Abs at day+90 and day+180. One patient who achieved humoral protection with the first vaccine and lost the immunity at day+90 successfully achieved humoral protection again with the third vaccine (Figure 1).
Patients’ clinical characteristics and laboratory values are presented in Table 1. As compared to other subjects, Subject 001 who responded to the first and third vaccine had CD19+ B cells prior to CAR-T and recovery of CD19+ B cells at day+180.
There were no adverse events related to PCV13.
Conclusion Interim results from this prospective clinical trial demonstrate that few LBCL patients had humoral protection against pneumococcus at baseline. Some patients responded to PCV13 given prior to CAR-T, and the immunity was maintained post CAR-T. PCV13 did not induce humoral protection when given at day+30, but one patient who responded to the first vaccine successfully achieved humoral protection again with the vaccine given at day+90. This patient's characteristics were notable for CD19+ B cells present prior to CAR-T and recovered at day+180. PCV13 vaccine was well tolerated in all patients enrolled in this trial. Additional biomarker analysis is ongoing including evaluation for cellular immune response against the vaccine adjuvant.
Disclosures
Lazaryan:Humanigen: Consultancy; Sanofi: Consultancy; AvroBio: Consultancy; Teladoc: Current equity holder in publicly-traded company; AmWel: Current equity holder in publicly-traded company. Gaballa:ADC Therapeutics: Speakers Bureau; Abbvie: Consultancy; TG Therapeutics: Consultancy, Speakers Bureau; Epizyme: Consultancy, Other: research support; Beigene: Consultancy. Shah:Amgen: Consultancy; Pfizer: Consultancy; Novartis: Consultancy; BMS/Celgene/Juno: Consultancy; Kite/Gilead: Consultancy, Other: grants and investigator-initiated trials; Precision Biosciences: Consultancy; Jazz: Consultancy, Other: grants and investigator-initiated trials; Acrotech: Consultancy; Beigene: Consultancy; Pharmacyclics: Consultancy; Adaptive: Consultancy; Century Therapeutics: Consultancy; Autolus: Consultancy; Servier: Other: grants and investigator-initiated trials; PeproMene Bio: Other: Steering committee. Pinilla Ibarz:Pharmacyclics: Consultancy; AbbVie: Consultancy; AstraZeneca: Consultancy; SecuraBio: Research Funding; Janssen Pharmaceuticals: Consultancy. Chavez:Merck: Research Funding; Janssen: Research Funding; ADC Therapeutics: Research Funding; Abbvie: Consultancy; Beigene: Honoraria; Astrazeneca: Research Funding, Speakers Bureau; GenMab: Consultancy; TG Therapeutics: Honoraria; Epizyme: Honoraria, Speakers Bureau; Adicet: Consultancy; Kite Pharma: Consultancy; MorphoSys/Incyte: Speakers Bureau. Jain:MyeloidTx: Consultancy; Novartis: Consultancy; Incyte: Research Funding; BMS: Consultancy; Kite Pharma: Consultancy, Research Funding. Locke:EcoR1: Consultancy; Emerging Therapy Solutions Gerson Lehrman Group: Consultancy; GammaDelta Therapeutics: Consultancy; Iovance: Consultancy; Janssen: Consultancy; Cowen: Consultancy; Calibr: Consultancy; Cellular Biomedicine Group: Consultancy; Bristol Myers Squibb/Celgene: Consultancy; Bluebird Bio: Consultancy, Research Funding; Kite, a Gilead Company: Consultancy, Research Funding; Legend Biotech: Consultancy; Novartis: Consultancy, Research Funding; Umoja: Consultancy; Amgen: Consultancy; Allogene: Consultancy, Research Funding; Wugen: Consultancy; Other: Patents & Royalties: patents, royalties, other intellectual property from several patents held by the institution in my name (unlicensed) in the field of cellular immunotherapy.; Celgene: Consultancy; A2: Consultancy; BMS: Research Funding; Daiichi Sankyo: Consultancy; Sana: Consultancy; Takeda: Consultancy; CERo Therapeutics: Research Funding; ), National Cancer Institute: Research Funding; Leukemia and Lymphoma Society: Research Funding; Aptitude Health: Other: Education or editorial activity; ASH: Other: Education or editorial activity; BioPharm Communications: Other: Education or editorial activity; CAREducation: Other: Education or editorial activity; Clinical Care Options Oncology: Other: Education or editorial activity; Imedex: Other: Education or editorial activity; Society for Immunotherapy of Cancer: Other: Education or editorial activity.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal