Abstract
Primary and secondary thrombotic microangiopathies (TMA) constitute a group of life-threatening diseases of different aetiologies characterized by similar symptoms. The comprehensive compilation of data related to TMA is challenging due to their rare occurrence. The objective of this study was to provide an overview of the incidence of thrombotic thrombocytopenia (TTP) and non-TTP TMAs in the Province of Quebec (PQ) (8.485 Million inhabitants in 2019) by taking advantage of the centralized ADAMTS-13 activity and antibody testing for PQ since 2013 at the CHU Sainte-Justine (CHUSJ)
All ADAMTS-13 activity and antibody titration were performed locally at CHUSJ. ADAMTS-13 results and patient demographic characteristics from April 2012 to December 2019 were extracted from the Laboratory Information System of CHUSJ and used in an anonymized database. Information on previous TMA episodes was obtained from a standardized clinical assessment form accompanying each plasma sample. Statistical analyses were performed with IBM SPSS version 26.0. The annual incidence rates were calculated based on the number of patients with a first-time recorded diagnosis of TMA using the Quebec mid-year (yr) estimated population of the years at stake. Patients with suspected TMA were further divided into two categories: individuals with TTP, defined by either an ADAMTS-13 activity ≤10% or a positive anti-ADAMTS-13 antibody titration, and patients with a suspected TMA other than TTP (non-TTP TMA) with a result of ADAMTS-13 activity >10%. The study was approved by the Research Ethics Committee of CHUSJ.
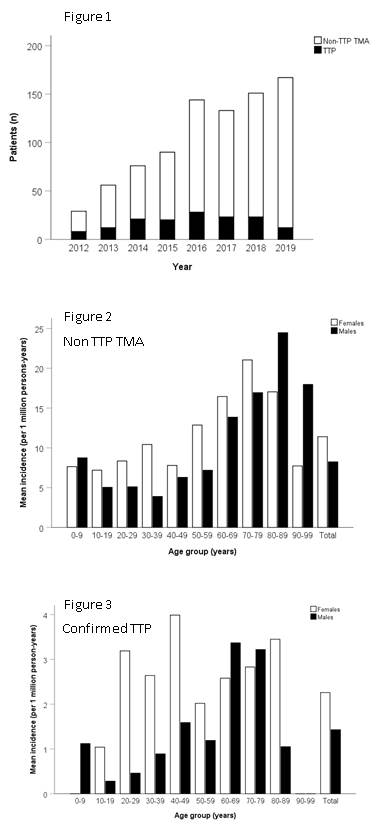
A progressive increase in the annual requests for ADAMTS13 activity was observed over the study period. The number of new patients increased from 2012 to 2016 but plateaued after 2016 at an average number of 170 new patients per year. The number of confirmed TTP cases in Quebec was higher from 2014 to 2018 compared to in 2012, 2013 and 2019 (Figure 1). A total of 2081 requests for ADAMTS13 activity testing were received during the study period, representing 846 subjects with a suspected TMA. 147 subjects (17%) had a confirmed TTP and 699 (83%) had a suspected non-TTP TMA.
TMAs were suspected more often in females (59%), both in confirmed TTP (62%) and non-TTP TMA (58%).
The mean annual incidence rate (MAIR) of TTP was 1.91 case/million/yr (95%CI: 1.46-2.35). This was higher in females (2.36 cases/million/yr; 95%CI: 1.34-3.37) compared to males (1.42 case/million/yr; 95%CI: 0.57-1.44; p=0.001). The MAIR of non-TTP TMA was 9.97 cases/million/yr (95%CI:5.85-14.09), 11.52 for females (95%CI:6.71-16.33) versus 8.41 for males (95% CI: 4.81-12.02; p=0.001).
In non-TPP TMA, the MAIR for males ranged from 3.9 to 7.2 cases per million prior to 60 year of age and increased after up to 24.5 at ages 80-89. For females, a first peak MAIR above 10 cases per million was observed during their thirties and a second peak was observed during their seventies (Figure 2).
In TPP, peaks incidences in females were observed from ages 20 to 49. It dipped from ages 50 to 69 and then increased again. In males, a highest MAIR was observed between ages 40 to 79.
In conclusion, the provincial centralization of ADAMTS-13 testing has enabled us to depict comprehensive picture of TTP and other suspected non-TPP TMA, thereby providing valuable information for caregivers and health authorities into these rare diseases. Sex and age related incidences observed in this study are comparable to those obtained through other registries and aggregated studies. Further analyses on clinical presentation of TMAs and patient follow up are now possible using the identification of this large cohort.
Lapeyraque: Alexion Pharmaceuticals Inc: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Rivard: Bayer Inc: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer Inc: Membership on an entity's Board of Directors or advisory committees, Research Funding; Octapharma Inc: Membership on an entity's Board of Directors or advisory committees; Novo Nordisk Inc: Membership on an entity's Board of Directors or advisory committees; CSL Behring Inc.: Membership on an entity's Board of Directors or advisory committees, Research Funding. Bonnefoy: Sanofi Genzyme Inc: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; CSL Behring Inc: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Alexion Pharmaceuticals Inc: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal