Introduction:
Smoking is a potential risk factor for the development of non-Hodgkin lymphoma (NHL), and prior studies have reported inferior survival in tobacco users with certain subtypes of the disease (Taborelli et al, BMC Cancer, 2017; Ollberding et al, Br J Haematol, 2013). For instance, tobacco smokers with NHL had an inferior overall survival (OS) compared to non-smokers in a series of 471 patients who were managed up front with either chemotherapy (68%), radiation (27%), or observation, and this appeared to be most pronounced in patients with follicular lymphoma and in those with a 20+ pack year smoking history (Geyer et al, Cancer, 2010). The impact of tobacco use on survival specifically in patients with mantle cell lymphoma (MCL) has not been well studied. We conducted a multicenter study in MCL and evaluated the prognostic impact of tobacco use.
Methods:
We included patients with MCL from 12 sites who were ≥18 years old and for whom smoking status was known at the time of diagnosis. Cases were evaluated for reported smoking status at the time of diagnosis (active smoker, prior smoker, or never smoker) and standard baseline clinical prognostic data were obtained for each patient. Descriptive statistics were generated for these characteristics and were then compared across smoking status using chi-squared tests, Fisher's exact tests, or ANOVA, where appropriate. Overall survival (OS) and progression free survival (PFS) were estimated using the Kaplan-Meier method, and were compared using log-rank tests.
Results:
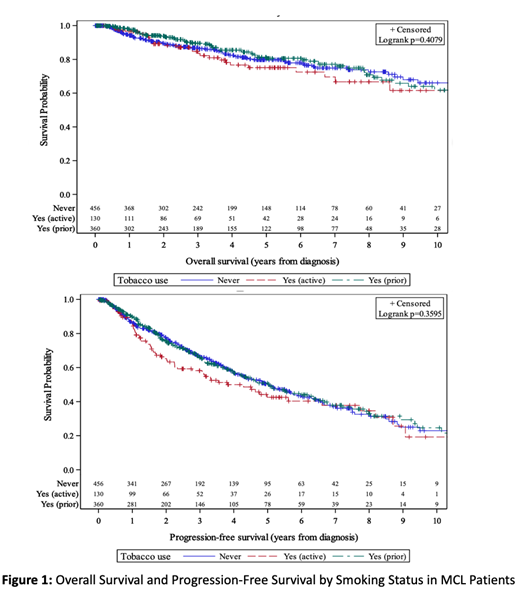
Of 946 included patients, 456 (48.2%) reported never using tobacco, 360 (38.7%) reported prior tobacco use, and 130 (13.7%) reported active tobacco use at the time of diagnosis. Median age was 59 in the active smoker group, 65 in prior smokers, and 61 in never smokers (p < 0.001). Any major medical comorbidity (defined as the presence of CAD, CHF, diabetes, CKD, ESRD, COPD, DVT, prior malignancy, or cirrhosis) was present in 59 (45.4%) of the active smokers, 143 (39.7%) of the prior smokers, and 140 (30.7%) of the never smokers (p = 0.002). Intensive induction regimens were used in 58.2% of active smokers, 47.2% of prior smokers, and 58.4% of never smokers (p=0.007). There were no significant differences between groups in regards to sex, race, ECOG performance status, Ann Arbor stage, time to first treatment, and use of auto transplant in first remission. Patients with no prior history of tobacco use were less likely to have a high risk MIPI score at diagnosis (26% high risk) compared to prior smokers (39.5%) and active smokers (32.5%, p=0.019). With a median follow up of 3.5 years after diagnosis, there was no significant difference between the 3 groups with regards to PFS or OS (Figure 1). Five-year OS in the never smoker group was 79.8% (95% CI: 74.8%, 83.9%) vs 75.1% (64.5%, 82.9%) in the active smoker group, and 80.6% (74.6%, 85.3%) in the prior smoker group (log rank p = 0.4079). Five- year progression free survival was 50.4% (44.6%, 56.0%) in the never smoker group, 42.5% (32.2%, 52.5%) in the active smoker group, and 50.2% (43.5%, 56.6%) in the prior smoker group (log rank p= 0.3595).
Conclusions:
Our data suggest that active or prior smoking does not significantly impact OS or PFS in patients with MCL. This study is limited by the fact that amount of current or former tobacco use was not available and it is not known how many current tobacco users ultimately stopped smoking during the course of their treatment. Future studies should incorporate more specific information regarding smoking history including pack-years and time between discontinuation of tobacco use and date of diagnosis. While tobacco use and other modifiable cardiovascular risk factors should be addressed as appropriate for all patients with MCL, current and former tobacco users can still achieve prolonged PFS and OS and may be candidates for intensive treatments after consideration of their other comorbidities and disease-specific risk factors.
Calzada:Seattle Genetics: Research Funding. Kolla:Amgen: Equity Ownership. Bachanova:Gamida Cell: Research Funding; GT Biopharma: Research Funding; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees; Incyte: Research Funding; Celgene: Research Funding; Novartis: Research Funding; Kite: Membership on an entity's Board of Directors or advisory committees. Gerson:Seattle Genetics: Consultancy; Abbvie: Consultancy; Pharmacyclics: Consultancy. Barta:Janssen: Membership on an entity's Board of Directors or advisory committees; Celgene: Research Funding; Mundipharma: Honoraria; Janssen: Membership on an entity's Board of Directors or advisory committees; Celgene: Research Funding; Takeda: Research Funding; Merck: Research Funding; Mundipharma: Honoraria; Bayer: Consultancy, Research Funding; Seattle Genetics: Honoraria, Research Funding. Danilov:Celgene: Consultancy; Abbvie: Consultancy; TG Therapeutics: Consultancy; Bayer Oncology: Consultancy, Research Funding; Gilead Sciences: Consultancy, Research Funding; Janssen: Consultancy; AstraZeneca: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Aptose Biosciences: Research Funding; Bristol-Meyers Squibb: Research Funding; MEI: Research Funding; Pharmacyclics: Consultancy; Verastem Oncology: Consultancy, Other: Travel Reimbursement , Research Funding; Curis: Consultancy; Takeda Oncology: Research Funding; Seattle Genetics: Consultancy. Grover:Seattle Genetics: Consultancy. Karmali:Astrazeneca: Speakers Bureau; Takeda, BMS: Other: Research Funding to Institution; Gilead/Kite; Juno/Celgene: Consultancy, Speakers Bureau. Hill:Seattle Genetics: Consultancy, Honoraria; Takeda: Research Funding; Amgen: Research Funding; TG therapeutics: Research Funding; AstraZeneca: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celegene: Consultancy, Honoraria, Research Funding; Genentech: Consultancy, Research Funding; Kite: Consultancy, Honoraria; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Ghosh:Pharmacyclics: Consultancy, Research Funding, Speakers Bureau; Seattle Genetics: Consultancy, Speakers Bureau; Genentech: Research Funding; Celgene: Consultancy, Research Funding, Speakers Bureau; Forty Seven Inc: Research Funding; Gilead/Kite: Consultancy, Speakers Bureau; Spectrum: Consultancy, Speakers Bureau; AbbVie: Consultancy, Speakers Bureau; T G Therapeutics: Consultancy, Research Funding; Astra Zeneca: Speakers Bureau. Park:BMS: Consultancy, Research Funding; Rafael Pharma: Membership on an entity's Board of Directors or advisory committees; G1 Therapeutics: Consultancy; Teva: Consultancy, Research Funding; Gilead: Speakers Bureau; Seattle Genetics: Research Funding, Speakers Bureau. Epperla:Pharmacyclics: Honoraria; Verastem Oncology: Speakers Bureau. Hamadani:Pharmacyclics: Consultancy; ADC Therapeutics: Consultancy, Research Funding; Merck: Research Funding; Celgene: Consultancy; Janssen: Consultancy; Medimmune: Consultancy, Research Funding; Sanofi Genzyme: Research Funding, Speakers Bureau; Otsuka: Research Funding; Takeda: Research Funding. Kahl:TG Therapeutics: Consultancy; BeiGene: Consultancy; Seattle Genetics: Consultancy; ADC Therapeutics: Consultancy, Research Funding. Martin:Janssen: Consultancy; Sandoz: Consultancy; I-MAB: Consultancy; Teneobio: Consultancy; Celgene: Consultancy; Karyopharm: Consultancy. Flowers:Karyopharm: Consultancy; Denovo Biopharma: Consultancy; Burroughs Wellcome Fund: Research Funding; AbbVie: Consultancy, Research Funding; Gilead: Consultancy, Research Funding; Spectrum: Consultancy; AstraZeneca: Consultancy; Pharmacyclics/Janssen: Consultancy, Research Funding; Bayer: Consultancy; Acerta: Research Funding; Genentech, Inc./F. Hoffmann-La Roche Ltd: Consultancy, Research Funding; Optimum Rx: Consultancy; Millenium/Takeda: Research Funding; Eastern Cooperative Oncology Group: Research Funding; National Cancer Institute: Research Funding; V Foundation: Research Funding; BeiGene: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; TG Therapeutics: Research Funding. Cohen:Genentech, Inc.: Consultancy, Research Funding; Janssen Pharmaceuticals: Consultancy; Takeda Pharmaceuticals North America, Inc.: Research Funding; Gilead/Kite: Consultancy; LAM Therapeutics: Research Funding; UNUM: Research Funding; Hutchison: Research Funding; Astra Zeneca: Research Funding; Lymphoma Research Foundation: Research Funding; ASH: Research Funding; Seattle Genetics, Inc.: Consultancy, Research Funding; Bristol-Meyers Squibb Company: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal