Introduction: Patients with newly diagnosed multiple myeloma (NDMM) who are ineligible for autologous stem cell transplant (ASCT) tend to have comorbidities and/or advanced age that make this subset of patients difficult to manage with current drug regimens.
Methods: A comprehensive literature search of PubMed, Embase, Clinicaltrials.gov and Web of Science was performed from inception and completed on 07/17/2019. Studies focusing on efficacy and tolerability of 3-drug regimens in patients with NDMM were included for the review.
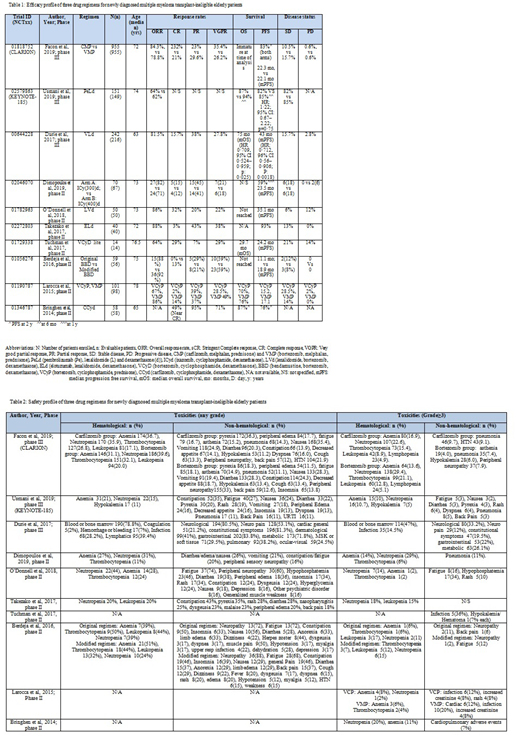
Results: Out of 3579 studies, a total of 10 (08 phase II and 03 phase III) clinical trials in last ten years (2010-2019) using 3-drug regimens in NDMM elderly pts (893M/807F) ineligible for ASCT (determined by investigators) were selected. A total of 1703/1740 NDMM pts were evaluated.
Proteasome inhibitors (PIs) such as carfilzomib (C), bortezomib (V) and ixazomib (I) showed promising results in elderly transplant-ineligible NDMM pts. CLARION trial (phase III, n=955) compared two PIs (C and V) with melphalan (M) and prednisone. There was no statistically significant difference in progression-free survival (PFS) between two groups (median: 22.3 vs 22.1 months; HR: 0.91; 95% CI, 0.75-1.10, p = 0.159) as well as overall survival (OS) (HR: 1.08; 95% CI: 0.82-1.43). Difference in the least square means of the HR-QoL (Health related- quality of life) was 4.99 (p<.0001) favoring C-group. M may not be an ideal drug to combine with carfilzomib in this setting given more AEs.(Facon et al 2019). V as 3-drug regimen in combination with lenalidomide (L) in 242 pts achieved statistically significant prolonged PFS (median 43 mo) and OS (median 75 mo) with great efficacy and acceptable risk-benefit profile. (Durie et al 2017; phase III). Multinational phase II trial (n=70) by Dimopoulos et al (2019) evaluated I, with different fixed doses of cyclophosphamide (Cy). Median duration was 19 cycles, indicating the long-term tolerability of regimen. With favorable toxicity profile and maintained QoL scores, trial concluded that this therapy is tolerable in elderly transplant-ineligible NDMM pts. Tuchman et al (2017) in phase II trial (n=14) investigated (V-Cy-d) and achieved ORR of 64%, with ≥VGPR of 57%. Low dose V showed great efficacy with M yielding ORR of 86% and VGPR or better of 49% in phase II trial (n=101) that also evaluated Cy as 3-drug combination but results were more productive with M with longer PFS and OS which reduced when impact of frailty was examined on outcomes. Since toxicity was higher with M, trial suggested that 2-drug combination should be preferred in elderly frail patients. (Larocca et al 2015). Efficacy was quite promising when Bringhen et al (2014) trialed C with Cy-d; 87% OS and 76% PFS at 1 y in phase II trial (n=58) with much favorable safety profile.
Monoclonal antibodies (mAb) such as elotuzumab (E) and pembrolizumab (Pe) are also tested in elderly. First study conducted on NDMM pts using humanized mAb; E, in phase II trial (n=40) by Takezako et al (2017) attained primary endpoint of the study (ORR) of (88%) and VGPR or better of 45% in Japanese pts with tolerable toxicities in elderly. No subjects on this study experienced severe peripheral neuropathy. KEYNOTE-185; a phase III multinational trial by Usmani et al (2019) evaluated Pe with Ld in 151 pts. FDA halted this study due to unfavorable benefit-risk profile; 19 deaths, 6 due to disease progression (PD), and 13 due to treatment-related AEs. Median PFS and median OS were not reached in either group.
Immunomodulators such as L achieved one of the longest PFS reported in a trial of transplant ineligible patients (35 mo) by using LVd regimen in phase II multicenter trial (n=50). (O'Donnell et al 2018)
Alkylating agents like bendamustine (ben) and M have been tested in different novel regimens. Decreasing intensity and increasing duration of ben resulted in better outcomes in phase II trial (n=59) by Berdeja et al (2016) and can be given as first line treatment. Ben yielded great results with low dose dexa as compared to high dose achieving 92% ORR. Original regimen was effective but relatively more toxic. Incidence of herpes and neuropathy decreased dramatically with the treatment modifications.
Conclusion: Three-drug regimens having PIs, mABs, immunomodulators and alkylating agents have shown desirable results in NDMM transplant (ASCT)-ineligible elderly patients and are likely the emerging standard of care for NDMM.
Anwer:In-Cyte: Speakers Bureau; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal