Introduction
Elotuzumab is monoclonal antibody (mAb) that specifically targets signaling lymphocytic activation molecule family member 7 (SLAMF7) that is present on myeloma cells. It fights myeloma cells by stimulating phagocytic action of NK cells and via ADCC (antibody‐dependent cell‐mediated cytotoxicity) pathway. Daratumumab is an anti-CD38 mAb with dual mechanisms of action i.e. tumoricidal and immunomodulation. The aim of this study is to review the efficacy and toxicity of elotuzumab and daratumumab based 3-drug combinations in patients (pts) with multiple myeloma (MM).
Methods
A systematic search of PubMed, Embase, Clinicaltrials.gov, Cochrane and Web of Science was performed for elotuzumab and daratumumab based regimen in MM patients from inception to June 12, 2019. Out of 604 studies, 08 phase II and III clinical trials based on 3-drug regimen were finalized.
Results
Total 2809 patients (pts) were evaluated out of 2879 enrolled pts. 856 were newly diagnosed (ND) and 1953 were relapsed/refractory (R/R). Elotuzumab (E) based 3-drug regimen were evaluated in 560 whereas daratumumab (D) based 3-drug regimen were analyzed in 889 pts.
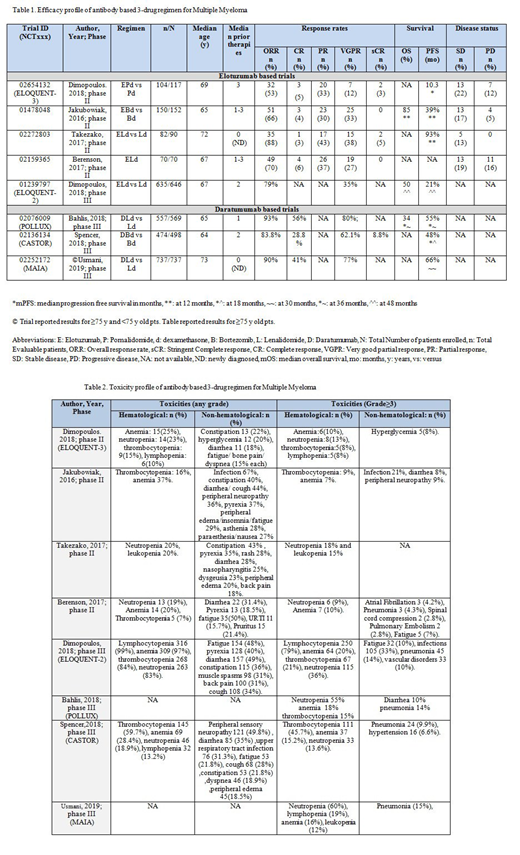
ELOQUENT-3 trial (n=117) in phase II used E + pomalidomide (P) and dexamethasone (d) (EPd) for R/R pts with ≥2 prior therapies. Median (m) progression free survival (PFS) was 10.2 months (mo) in EPd versus (vs) 4.6 mo in Pd arm [Hazard ratio (HR) 0.54 (95% CI: 0.34-0.86; p=0.008)], i.e. 46% lower risk of progression or death in EPd vs Pd arm. ORR (overall response rate) was 53% (complete response [CR] 5% + stringent CR [sCR] 3% + partial response [PR] 33% + very good partial response [VGPR] 12%) in EPd vs 26% in Pd arm (odds ratio [OR]: 3.25 (1.49-7.11). Grade (G) ≥3 adverse events (AEs) were anemia (10%), neutropenia and infections (13% each). (Dimopoulos et al. 2018).
Phase II trial by Jakubowiak et al. (2016) observed 1 year (y) PFS of 39% in E-Bortezomib(B)-d vs 33% in Bd arm in 152 R/R pts (HR: 0.72; p = .09) with 28% decrease in progression or death with EBd vs Bd. ORR was 66% (4% CR+ 33% VGPR + 30% PR) vs 63%. OS (overall survival) at 1 y was 85% (EBd) and 74% (Bd) (HR:0.6). G≥3 AEs were infections (21%), thrombocytopenia and peripheral neuropathy (9% each). In phase II trial, ELd (E-Lenalidomide-d) arm yielded ORR of 88% (CR 3% + sCR 5% + PR 43% + VGPR 38%) vs 74% in Ld arm in 82 ND pts. PFS at 1 yr was 93% vs 91%. G≥3 AEs included neutropenia (18%) and leukopenia (15%). (Takezako et al. 2017). Berenson et al. (2017) in phase II trial (n=70) studied G≥3 infusion reactions (IRs) using ELd in ND and R/R pts. ORR was 70% (CR 6% + VGPR 27% + PR 37%). G3 AEs included anemia in 10% pts (no G3 IRs).
ELOQUENT-2 trial randomized 646 R/R pts in phase III. PFS at 4 y is 21% vs 14% (HR: 0.71, 0.59-0.86; p= .0004), favoring ELd with 29% reduction in myeloma progression or death. With VGPR of 30%, ORR was 79% vs 66 % (ELd vs Ld) with HR: 0.77; 0.62-0.95; p = 0.0176. OS at 4 y was 50% vs 43% (HR: 0.78; 0.63-0.96). G≥3 AEs included lymphocytopenia (79%), neutropenia (36%), anemia (20%) and thrombocytopenia (21%). (Dimopoulos et al, 2018).
POLLUX trial used daratumumab (D)-Ld regimen in 569 R/R patients in phase III. PFS at 3 y was 55% vs 27% (DLd vs Ld) in pts with 1-3 prior therapies. With 56% CR and 80% VGPR; ORR was 93% vs 76%. DLd arm achieved 30% minimal residual disease (MRD)-negative status compared to 5% in Ld arm (p<0.001). OS at 3-yr was 34% vs 42%. G≥3 AEs were neutropenia (55%), anemia (18%), thrombocytopenia (15%) and pneumonia (14%). (Bahlis et al. 2018).
CASTOR trial in phase III (n=498) showed 18-mo PFS of 48% vs 7.9% in DBd vs Bd arm in R/R pts (HR: 0.31 (0.24-0.39); p<0.0001). ORR was 83.8% (CR 28.8%+ sCR 8.8% + VGPR 62.1%) vs 63.2% (p<0.001). DBd-treatment led to 11.6% MRD-negative status vs 2.4% in Bd-treated pts (p=0.000034). Thrombocytopenia (45.7%), anemia (15.2%) and neutropenia (13.6%) were G≥3 AEs. (Spencer et al. 2018).
A Phase III trial (n=737) observed 30 mo PFS of (66 vs 52) % in ≥75 y old pts [HR 0.63 (0.44-0.92)] with DLd vs Ld arms in ND transplant ineligible pts with ORR of (90 vs 81) % (≥CR 41% + ≥VGPR 77%). MRD- negative rate was (19 vs 8) % in DLd vs Ld arms respectively. G≥3 AEs were neutropenia (60%), lymphopenia (19%), anemia (16%), pneumonia (15%) and leukopenia (12%). <75 y old showed ORR of (95 vs 82) % with 30 mo PFS of (75 vs 58) % in both arms. (Usmani et al 2019).
Conclusion: Elotuzumab and daratumumab based 3-drug combinations showing great improvements in ORR, PFS and OS of ND and RR MM patients with favorable toxicity profile.
Anwer:In-Cyte: Speakers Bureau; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal