Background
Idelalisib is a PI3-kinase inhibitor specific for the delta isoform, approved (in combination with rituximab or ofatumumab) for the treatment of adult patients with Relapsed/Refractory Chronic Lymphocytic Leukemia (R/R CLL). Although the efficacy of idelalisib was reported in clinical trials, it is unclear how this translates into Real World.
Methods
A non-interventional retrospective study was conducted in Spain to describe the clinical characteristics and outcomes of patients diagnosed with R/R CLL that started treatment with idelalisib between 1 February 2015 and 31 December 2017.
The Time from start of idelalisib treatment to either the start of a new anti CLL therapy or death (Time to Next Treatment or Death - TNTD) was defined as primary endpoint. Secondary endpoints included overall survival (OS), time to discontinuation (TTD) and safety, especially Adverse Events of Special Interest (AESI):
≥ grade 3 transaminase elevations, diarrhea /colitis, pneumonitis, neutropenia,
Cytomegalovirus, bacterial, fungal and respiratory virus infections (RVI),
Pneumocystis jirovecii pneumonia (PjP).
Results
Investigators from 21 centers included 77 patients in the study. The median age was 72.1 years (48-86) and 67.5% (n=52) were male. The main baseline characteristics were: Binet stage B-C in 32.5% (n=25), 17p deletion or TP53 mutation in 35.1% (n=27) and the median number previous lines of therapy was 4 (1-16).
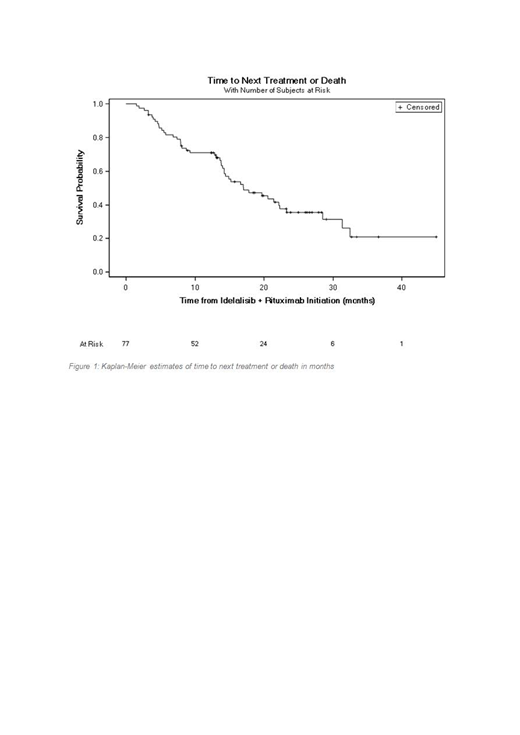
With a median follow up time of 14.4 months (1.2-44.4), 27 patients (35.1%) had started a new treatment and 19 (24.7%) had died.
The median TNTD was 17.1 months (95% CI 14.0-22.3) in the overall population and 13.8 months (95% CI 7.9-15.2) in patients that discontinued idelalisib.
The median OS has not been reached, with a cumulative probability of surviving at 1 and 2 years of 0.84 (95% CI 0.73-0.91) and 0.67 (95% CI 0.52-0.78).
During follow-up 54 patients (70.1%) discontinued idelalisib: 39 (72.3%) due to toxicity and 10 (18.5%) due to disease progression. The median TTD was 13.0 months (95% CI 7.7-15.8); 5.3 months (95%CI 3.8-8.0) in patients who discontinued treatment due to serious adverse events (SAE) or AESI and 10.5 months (95% CI 0.5-16.0) in those who did so due to progressive disease.
A total of 355 AEs were reported, of which 29.0% (n=103) were SAE.
181 AESIs were recorded: the most frequent were neutropenia (n=67; 22 grade >3), diarrhea/colitis (n=37; 14 grade >3) and bacterial infections (n=24; 14 grade >3). Other AESI were CMV infections (n=11), grade ≥ 3 pneumonitis (n=7), RVI (n=6), fungal infections (n=5), grade ≥ 3 transaminase elevations (n=3) and PjP (n=1). The median time from Idelalisib start to first AESI was 9.5 months (95%CI 5.5-15.3).
During follow-up, 19 deaths were registered, 10 of which were caused by idelalisib-related SAEs (4 respiratory infections, 2 pneumonitis, 1 diarrhea/colitis). The time from SAE related to idelalisib that led to death, to death was 0.8 months (95% CI 0.7-1.7).
Conclusions
This real world study shows results of effectiveness of idelalisib consistent with the efficacy findings of the 312-0116 clinical trial. Toxicity was the most common reason for idelalisib discontinuation. Findings of adverse events were consistent with the known safety profile of idelalisib and no new drug-related adverse events were identified.
This is a Gilead Sciences sponsored study.
Perez Encinas:GILEAD SCIENCES: Research Funding; CELGENE: Consultancy; JANSSEN: Consultancy. Lopez Jimenez:GILEAD SCIENCES: Honoraria, Other: Education funding. Ortiz:GILEAD SCIENCES: Research Funding. Cordoba:Pfizer: Consultancy; Celgene: Consultancy, Honoraria, Speakers Bureau; FUNDACION JIMENEZ DIAZ UNIVERSITY HOSPITAL: Employment; Roche: Honoraria, Speakers Bureau; Kyowa-Kirin: Consultancy, Honoraria, Speakers Bureau; Gilead: Consultancy, Research Funding, Speakers Bureau; Servier: Consultancy, Honoraria, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau. Ramirez Payer:GILEAD SCIENCES: Research Funding. González-Barca:Roche: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Celtrion: Consultancy; AbbVie: Consultancy, Honoraria; Kiowa: Consultancy; Takeda: Honoraria; Celgene: Consultancy. Martín Sánchez:GILEAD SCIENCES: Research Funding. Sanchez:Gilead Sciences: Research Funding. Baltasar Tello:JANSSEN: Consultancy, Honoraria; ABBVIE: Honoraria; ROCHE: Honoraria; GILEAD: Honoraria. Amutio:NOVARTIS: Consultancy; JANSSEN: Consultancy, Honoraria; CELGENE: Consultancy, Honoraria; ROCHE: Honoraria; GILEAD SCIENCES: Consultancy, Honoraria; TAKEDA: Consultancy; GSK: Honoraria; BMS: Honoraria; JAZZ PHARMACEUTICALS: Honoraria; MUNDIPHARMA: Consultancy. Vidal Maceñido:GILEAD SCIENCES: Research Funding. Fernandez:GILEAD SCIENCES: Research Funding. Loscertales:Gilead: Honoraria; AbbVie: Honoraria; AstraZeneca: Honoraria; Janssen: Honoraria; Roche: Honoraria. Rodríguez:GILEAD SCIENCES: Research Funding. Alaez:ROCHE: Consultancy. Ramroth:Gilead Sciences: Employment. Palla:Gilead Sciences: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal