Introduction: Blinatumomab is a bispecific T-cell engager (BiTE®) targeted immuno-oncology therapy with dual specificity for cluster of differentiation (CD) 19 and CD3 that redirects the patient's CD3-positive cytotoxic T cells to lyse CD19-positive malignant cells. Global studies have evaluated blinatumomab in patients with advanced Philadelphia chromosome-negative relapsed/refractory acute lymphoblastic leukemia (Ph- R/R ALL). In the global phase 3 TOWER study, blinatumomab monotherapy vs standard-of-care chemotherapy resulted in a significantly higher rate of complete remission (CR)/CR with partial hematologic recovery of peripheral blood counts (CRh)/CR with incomplete hematologic recovery of peripheral blood counts (44% vs 25%; P < 0.001) and longer median overall survival (7.7 vs 4.0 months; P = 0.01) (Kantarjian H, et al. NEJM. 2017;376:836-47). There are limited data on the efficacy and safety of blinatumomab in Asian patients, whose immunologic genetic background may differ from other patient populations. Therefore, we conducted a patient-level pooled analysis of the efficacy and safety of blinatumomab in 45 Asian adult patients with Ph- R/R ALL-19 from the blinatumomab arm of TOWER (NCT02013167) and 26 from a phase 1b/2 study in Japanese adults (NCT02412306).
Methods: Patients in both studies were ≥ 18 years old and had Ph- R/R ALL, > 5% blasts, Eastern Cooperative Oncology Group performance status 0-2, and no central nervous system pathology. Patients received a maximum of 2 cycles of induction blinatumomab for 4 weeks by continuous intravenous infusion (cycle 1/week 1: 9 μg/day; cycle 1/weeks 2-4: 28 μg/day; subsequent cycles: 28 μg/day) followed by 2 weeks of no blinatumomab (each 6-week cycle). Responders (≤ 5% bone marrow blasts within 2 induction cycles) received blinatumomab 28 μg/day up to a maximum of 5 induction/consolidation cycles. In TOWER, patients who continued morphologic remission received up to 12 months of maintenance therapy. Patients could undergo stem cell transplantation at any time following the first treatment cycle.
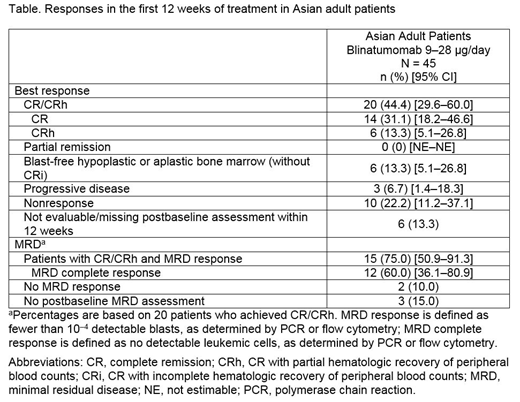
Results: Of the 45 Asian patients enrolled (26 female; median [range] age, 43 [18-75] years; prior hematopoietic stem cell transplantation, 20 [44.4%]; ≥ 1 prior salvage therapy, 30 [66.7%]), 44 received at least 1 cycle of blinatumomab 9-28 μg/day. Responses in the first 12 weeks of treatment (CR/CRh and minimal residual disease response) are shown in the Table. The Kaplan-Meier (KM) median overall survival time was 11.9 (95% CI: 9.9-17.1) months, and the KM median relapse-free survival time was 8.9 (95% CI: 3.8-10.7) months; median overall survival in the blinatumomab arm of TOWER was 7.7 months. Forty-one (93.2%) patients had grade ≥ 3 treatment-emergent adverse events (TEAEs), and 5 (11.4%) had fatal AEs. Grade ≥ 3 events TEAEs of interest included neurologic events (4.5%), cytokine release syndrome (2.3%), cytopenias (6.8%), and infections (20.5%).
Conclusions: The safety and efficacy of blinatumomab in Asian patients were comparable with previous global studies with similar disease response rates and a favorable safety profile with no new safety signals.
Kobayashi:Astellas Amgen BioPharma: Research Funding, Speakers Bureau; Pfizer: Research Funding, Speakers Bureau; SymBio: Consultancy. Iida:Chugai Pharmaceutical Co., Ltd.: Research Funding. Minami:Astellas: Research Funding; Bayer: Honoraria, Other: Clinical trial, Research Funding; Taiho: Honoraria, Other: Clinical trial, Research Funding; Taisho-Toyama: Research Funding; Takeda: Honoraria, Research Funding; CSL Behring: Research Funding; Genomic Health: Honoraria; Daiichi Sankyo: Other: Clinical trial, Research Funding; Sumitomo Dainippon Pharma: Honoraria, Research Funding; Eizai: Honoraria, Research Funding; Janssen: Honoraria; Kowa: Honoraria; Kyowa-Kirin: Honoraria, Research Funding; Nihon Shinyaku: Research Funding; Eli Lilly: Honoraria, Research Funding; Merck Serono: Honoraria, Research Funding; MSD: Honoraria, Other: Clinical trial, Research Funding; Nippon Chemiphar: Honoraria, Research Funding; Ono Yakuhin: Honoraria, Other: Clinical trial, Research Funding; BMS: Honoraria, Other: Clinical trial, Research Funding; Celgene: Honoraria; AstraZeneca: Other: Clinical trial; Boehringer: Honoraria, Research Funding; Otsuka: Honoraria; Pfizer: Honoraria, Other: Clinical trial, Research Funding; Sanofi: Honoraria, Research Funding; Shire Japan: Honoraria; Abbvie: Honoraria; Nihon Medi-Physics: Honoraria; Asahi-Kaseo Pharma: Research Funding; Amgen Inc: Other: Clinical trial; Nihon Kayaku: Research Funding; Shionogi: Research Funding; Novartis: Honoraria, Other: Clinical trial; Chugai: Honoraria, Other: Clinical trial, Research Funding; Yakult: Research Funding; Teijin Pharma: Research Funding. Maeda:Mundipharma Co Ltd.: Honoraria; Kyowa Kirin Co. Ltd.: Honoraria; Astellas Pharma Inc.: Research Funding; Bristol-Myers Squibb: Honoraria, Research Funding. Yoon:Novartis: Consultancy, Honoraria; Yuhan Pharma: Research Funding; MSD: Consultancy; Kyowa Hako Kirin: Research Funding; Genentech, Inc.: Research Funding; Janssen: Consultancy; Amgen: Consultancy, Honoraria. Tran:Amgen: Employment, Equity Ownership. Morris:Amgen: Employment, Equity Ownership. Franklin:Amgen: Employment, Equity Ownership. Chong:Amgen Asia Holding Limited: Employment, Equity Ownership. Kiyoi:Astellas Pharma Inc.: Honoraria, Research Funding; Zenyaku Kogyo Co., Ltd.: Research Funding; Chugai Pharmaceutical Co., Ltd.: Research Funding; Pfizer Japan Inc.: Honoraria; FUJIFILM Corporation: Research Funding; Nippon Shinyaku Co., Ltd.: Research Funding; Otsuka Pharmaceutical Co.,Ltd.: Research Funding; Eisai Co., Ltd.: Research Funding; Kyowa Hakko Kirin Co., Ltd.: Research Funding; Bristol-Myers Squibb: Research Funding; Takeda Pharmaceutical Co., Ltd.: Research Funding; Sumitomo Dainippon Pharma Co., Ltd.: Research Funding; Daiichi Sankyo Co., Ltd: Research Funding; Perseus Proteomics Inc.: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal