Introduction: allogeneic transplantation (allo-HSCT) is a curative treatment for patients with advanced lymphoma. Haploidentical (haplo-SCT) transplantation extended the accessibility to allo-HSCT, overcoming the issue of donor availability. However, alternative donor allo-HSCT is still considered at higher risk of non-relapse mortality due to the HLA disparity and thus an anticipated higher incidence of GVHD. In this context, the use of a non myeloablative conditioning (NMAC) regimen combined with post transplantation cyclophosphamide (PT-Cy) based GVHD prophylaxis may reduce procedure related toxicity. The aim was to evaluate the toxicity and efficacy of haplo-SCT using NMAC with PT-Cy in advanced lymphoma patients.
Methods: We here report the retrospective experience of a bicentric transplantation program. We analyzed a cohort of lymphoma patients undergoing Haplo-SCT and homogeneously receiving NMAC and PT-Cy. Inclusion criteria were: 1) first allo-HSCT for advanced lymphoma between 2009 and 2018; 2) haploidentical donor; 3) NMAC (fludarabine cyclophosphamide and 2 gray TBI GVHD prophylaxis consisted of PT-Cy day+3 and +4 , cyclosporine A and MMF starting from day +5. Multivariate analyses included age, disease type (NHL vs HL), HCT-CI (< vs ≥ 3), graft source (PBSC vs BM), disease status at haplo-SCT (CR vs other).
Results: One hundred forty seven patients (73 NHL; 74 HL) with a median age of 46 years (range: 19-71) were included. PBSC (peripheral blood stem cell) was used as graft source in 96 patients (65%). Patients received a median number of 3 conventional chemotherapy lines before haplo-SCT (1-8). Sixty-five (44%) had relapse after Auto-HCT. At the time of haplo-SCT, 96 patients (66%) were in complete remission.
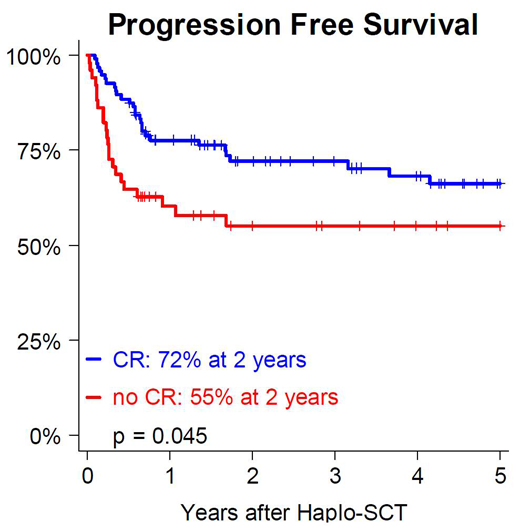
The cumulative incidences of day+100 grade 2-4 and 3-4 acute GVHD were 30% and 3%, respectively. The cumulative incidences of 2-year chronic and moderate or severe chronic GVHD were 13% and 8%, respectively. With a median follow up of 39 months (6-114), 2-year NRM was 14%, with a trend for higher risk in patients with HCT-CI ≥ 3 (HR 0.39, 95CI [0.15-1.04] p = 0.061) while age was not associated with an increased risk of NRM (HR 1.01, 95CI [0.98-1.05], p = 0.450). Two-year cumulative incidence of relapse (CIR) was 21% and 18% in HL and NHL patients, respectively. Disease status at the time of haplo-SCT was strongly associated with relapse (HR 2.99, 95CI [1.41-6.35], p = 0.004) In HL patients, 2-year PFS, OS and GRFS were 65%, 77% and 57%, respectively, while corresponding values in NHL patients were 65%, 69% and 55%, respectively. Two-year PFS and GRFS were significantly higher in patients who underwent haplo-SCT in CR (PFS: CR vs. no CR: 72% vs. 55%, p=0.045; GRFS: CR vs. no CR: 63% vs. 42%, p=0.010). There was a trend for better 2-year OS in CR (OS: CR vs. no CR: 78% vs. 63%, p=0.063.
Conclusion: We confirm the feasibility of haplo-SCT using NMAC and PT-Cy with low incidence of GVHD (notably severe forms) and NRM. In addition, we observed a relatively low incidence of relapse (19%) in this cohort of heavily pretreated patients, underlining a potent graft-versus-lymphoma effect after haplo-SCT, leading to promising survivals, including high rate of GRFS (>50%), suggesting a preserved long term quality of life in survivors. We conclude that NMAC haplo-SCT with PT-Cy should be considered as a valuable curative option for advanced lymphoma patients, with a favorable toxicity profile and promising long term survival.
Stoppa:celgene: Other: travel fees, lecture fees; takeda: Other: travel fees. Carlo-Stella:MSD: Honoraria; BMS: Honoraria; Janssen: Other: Travel, accommodations; Boehringer Ingelheim: Consultancy; Genenta Science sr: Consultancy; Sanofi: Consultancy, Research Funding; ADC Therapeutics: Consultancy, Other: Travel, accommodations, Research Funding; Novartis: Consultancy, Research Funding; Servier: Consultancy, Honoraria, Other: Travel, accommodations; F. Hoffmann-La Roche Ltd: Honoraria, Other: Travel, accommodations, Research Funding; Rhizen Pharmaceuticals: Research Funding; Celgene: Research Funding; Amgen: Honoraria; Takeda: Other: Travel, accommodations; Janssen Oncology: Honoraria; AstraZeneca: Honoraria. Chabannon:EBMT: Other: Working Party Chair, Board member; Fresenius Kabi: Other: research support; Miltenyi Biotech: Other: research support; Terumo BCT: Other: speaker's fees; Celgene: Other: speaker's fees; Novartis: Other: speaker's fees; Gilead: Other: speaker's fees, hospitalities; Sanofi SA: Other: research support, speaker's fees, hospitalities. Santoro:Takeda: Speakers Bureau; BMS: Speakers Bureau; Roche: Speakers Bureau; Abb-Vie: Speakers Bureau; Amgen: Speakers Bureau; Celgene: Speakers Bureau; Servier: Consultancy, Speakers Bureau; Gilead: Consultancy, Speakers Bureau; AstraZeneca: Speakers Bureau; Pfizer: Consultancy, Speakers Bureau; Arqule: Consultancy, Speakers Bureau; Lilly: Speakers Bureau; Sandoz: Speakers Bureau; Eisai: Consultancy, Speakers Bureau; Novartis: Speakers Bureau; Bayer: Consultancy, Speakers Bureau; MSD: Speakers Bureau; BMS: Consultancy. Blaise:Sanofi: Honoraria; Jazz Pharmaceuticals: Honoraria; Molmed: Consultancy, Honoraria; Pierre Fabre medicaments: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal