B-cell receptor (BCR) signaling kinases are important targets in therapy of CLL. Resistance of lymphoid niche-resident CLL cells to BCR-signaling inhibition is fostered by the tumor microenvironment. We found that stromal B-cell activation factor (BAFF)-mediated activation of spleen tyrosine kinase (SYK) triggered BCR signaling, thereby contributing to apoptosis resistance in CLL cells (Paiva et al, 2017). The SYK inhibitor entospletinib (ENTO) abrogated BAFF-mediated BCR signaling accompanied by a decrease in pSTAT3 and MCL1 in vitro. We designed a Phase I/II investigator-sponsored trial of ENTO in combination with Obinutuzumab (Obin) in patients (pts) with relapsed/refractory CLL and non-Hodgkin lymphoma (NHL). Eligible pts were aged ≥18 years, had CLL/NHL (Phase I) or CLL (Phase II), relapsed and/or refractory to ≥1 prior therapies (no prior SYK inhibitor), ECOG performance status ≤2 and preserved organ function. The Phase I part of the study followed a standard 3+3 design with two dose levels (DL1: ENTO 200 mg PO BID; DL2 - ENTO 400 mg PO BID). ENTO was given for 7 days (run-in). Subsequently, Obin was given IV concurrently with ENTO on days 1, 2, 8, 15 of Cycle 1 and Day 1 of Cycles 2-6 in standard doses. ENTO was given until disease progression. Primary study objectives were toxicity (Phase I) and efficacy (objective response rate (ORR); Phase II). Correlative analysis of samples was performed at baseline, after run-in phase (7 days of entospletinib single agent) and after 6 cycles of combination therapy. Reverse protein phase array was performed at the RPPA core facility at MD Anderson Cancer Center. T cell populations and cytokine production were analyzed by flow cytometry.
At DL1 of Phase I, six pts were enrolled (4 - CLL; 2 - follicular lymphoma). One pt experienced a dose-limiting toxicity (DLT: grade 3 asymptomatic LFT abnormalities which failed to resolve within 72 hours) attributed to ENTO. Other grade 3-4 toxicities included 2 grade 3 infusion reactions and one transient grade 4 neutropenia (attributed to Obin). Two pts remain on therapy after a median follow-up of 15 months. Three pts were enrolled at DL2 without DLTs.
In Phase 2, 18 pts with CLL received ENTO 400 mg PO BID (in combination with Obin). One pt was deemed ineligible due to Richter's transformation at study entry. Of the 17 evaluable pts, 71% were men. Median age was 66 years (range 47-76), and 76% were aged >65 years. 94% had ECOG performance status ≤1. Six pts (35%) had a complex karyotype, 6 (35%) had a TP53 aberration, 2 - NOTCH1 and 3 - SF3B1 mutation. Median number of prior therapies was 2 (range, 1-6): 47% had received prior fludarabine, 53% - bendamustine, 35% - ibrutinib (5 pts with intolerance, 1 with progression).
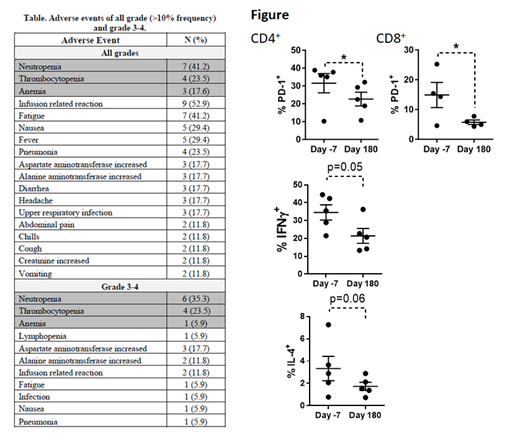
As of July 1, 2019, with median follow-up of 9.5 months (range, 3-17 months), 71% of pts remain on treatment. Median relative dose intensity of ENTO (ratio of actual to planned cumulative dose during drug exposure period) was 97%. ORR was 82%, 11 (65%) pts with partial response, three (17%) pts had CR (two MRD-negative in the bone marrow). All patients had a reduction in lymphadenopathy. Median duration of response and median progression-free survival (PFS) were not reached. All pts are alive. Five (29%) pts discontinued treatment (1 withdrawal of consent; 1 with recurrent LFT abnormalities; 1 for concomitant comorbidity and 2 with progression of disease). The most frequently occurring adverse events of all grades were infusion-related reactions, neutropenia and fatigue (Table). There were no Grade 5 events.
Treatment with ENTO for 7 days during run-in phase led to downmodulation of pSTAT3 and MCL1 in CLL cells (RPPA assay), consistent with our pre-clinical observations.
MCL1 has been previously implicated in survival of T cells at multiple stages of development. At the end of cycle 6, we observed a decrease in CD19+ and concomitant increase in CD3+ cells compared with baseline. There was no change in naïve, T-effector memory and T central memory cells within the CD4+ or CD8+ populations. Treatment with ENTO led to decreased PD-1 expression in CD4+ (22.6±3.8 vs. 31.5±5.4%, p=0.02) and CD8+ cells (p=0.05; Figure). Meanwhile, CTLA-4 expression was unchanged. PMA/ionomycin-stimulated CD4+ T cells demonstrated a decrease in IFNγ and IL-4.
In summary, a combination of ENTO and Obin was effective and well tolerated in patients with R/R CLL, and was accompanied by downmodulation of MCL1 in CLL cells and PD-1 in T cells.
Persky:Debiopharm: Other: Member, Independent Data Monitoring Committee; Bayer: Consultancy; Sandoz: Consultancy; Morphosys: Other: Member, Independent Data Monitoring Committee. Spurgeon:Janssen: Research Funding; Astra Zeneca: Research Funding; Bayer: Other: drug support only, Research Funding; Ariad: Other: drug support only, Research Funding; KITE: Other: drug support only, Research Funding; Acerta: Research Funding; Bristol Myers Squibb: Research Funding; Genentech: Honoraria, Research Funding; Novartis: Other: drug support only, Research Funding. Danilov:Gilead Sciences: Consultancy, Research Funding; Abbvie: Consultancy; Takeda Oncology: Research Funding; Bristol-Meyers Squibb: Research Funding; Verastem Oncology: Consultancy, Other: Travel Reimbursement , Research Funding; Genentech: Consultancy, Research Funding; TG Therapeutics: Consultancy; Bayer Oncology: Consultancy, Research Funding; Celgene: Consultancy; Curis: Consultancy; Seattle Genetics: Consultancy; Aptose Biosciences: Research Funding; AstraZeneca: Consultancy, Research Funding; MEI: Research Funding; Janssen: Consultancy; Pharmacyclics: Consultancy.
Entospletinib in CLL
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal