Introduction: Peripheral T-cell lymphomas (PTCL) are a heterogeneous group of diseases associated with a poor outcome with anthracycline-based chemotherapy, even when intensified induction regimens and consolidative autologous stem cell transplantation (ASCT) are employed. CD30 expression is universal in anaplastic large cell lymphoma (ALCL) and variable CD30 expression has also been demonstrated in other PTCL subtypes. Brentuximab vedotin (BV) is a CD30-directed antibody drug conjugate that prolongs progression-free survival (PFS) and overall survival (OS) when combined with cyclophosphamide, doxorubicin, and prednisone (CHP) as compared to CHOP chemotherapy (Horowitz, 2019). Despite this major advance, there remains room for improvement as the 3y PFS was 57% in patients (pts) who received BV-CHP.
Based on retrospective studies, CHOP plus etoposide (CHOEP) is commonly used as initial therapy for PTCL, and appears to benefit younger pts as compared to CHOP. In a phase 1 study, BV-CHP followed by BV monotherapy without consolidative ASCT led to excellent outcomes (Fanale, 2018). We performed a multicenter phase 2 trial to evaluate the safety and efficacy of adding etoposide to BV-CHP (CHEP-BV) followed by BV consolidation in pts with newly diagnosed CD30-expressing PTCL.
Methods: Adult pts with newly diagnosed CD30-expressing (≥ 1% of tumor cells) PTCL were eligible, including pts with ALK+ ALCL with IPI score ≥ 2, ALK-negative ALCL, PTCL not otherwise specified (NOS), angioimmunoblastic T-cell lymphoma (AITL), adult T-cell leukemia or lymphoma, enteropathy-associated T-cell lymphoma, or hepatosplenic T-cell lymphoma. Pts could receive prephase steroids and/or 1 cycle of standard CHOP-equivalent chemotherapy (SOC) prior to study entry. 6 patients were treated at the expected phase 2 dose of CHEP-BV as part of a safety lead-in: 6 x 21-day cycles of BV 1.8mg/kg on d1, cyclophosphamide 750mg/m2 on d1, doxorubicin 50mg/m2 on d1, prednisone 100mg daily on d1-5, and etoposide 100mg/m2 on d1-3. G-CSF primary prophylaxis was mandatory. After the safety-lead in, a 2-stage design was employed and if 7/16 pts had a complete response (CR) after CHEP-BV, 28 pts total would be enrolled. Responding pts could receive consolidation with 1.8mg/kg BV every 21 days for up to 10 additional cycles either after ASCT or directly after CHEP-BV if no ASCT was performed at investigator discretion. The primary endpoints were safety and the CR rate by PET-CT after CHEP-BV assessed by investigators according to the 2014 Lugano classification. Secondary endpoints were PFS and OS. In this preliminary analysis, we report the overall response rate (ORR) and CR rate after 3 cycles and at completion of CHEP-BV.
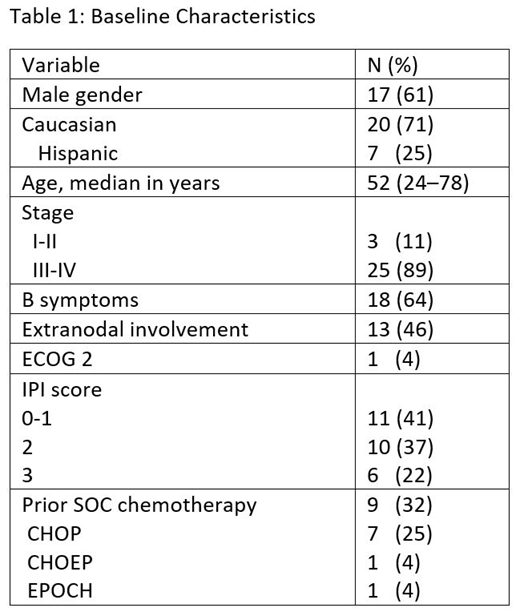
Results: 28 pts were enrolled and were evaluable for toxicity, 25 were evaluated for efficacy. We observed only 1/6 DLTs in cycle 1 (platelet < 10k) during the safety-lead-in and open enrollment proceeded. 12 pts had AITL, 11 had ALCL (1 ALK+, 10 ALK-), 4 had PTCL NOS, and 1 had T-follicular helper PTCL. Baseline characteristics are in shown in Table 1. 20 pts completed all cycles of CHEP-BV, 1 pt discontinued CHEP-BV early (MD discretion), and 7 remain on CHEP-BV. Of 20 pts who completed CHEP-BV, 1 pt had progressive disease (PD) at end of induction, 10 proceeded to ASCT and 9 did not. Of 19 consolidation-eligible pts, 1 pt discontinued prior to consolidation at MD discretion (DUSP22 rearrangement) and 18 pts are planned for or started BV consolidation. 1 pt reached EOT and 1 pt discontinued BV consolidation after 9 cycles due to pneumonia and grade (gr) 2 neuropathy. The most frequent CHEP-BV related adverse events (AE) include fatigue (75%), nausea (71%), anemia (46%), peripheral neuropathy (39%, all gr 1), thrombocytopenia (36%), neutropenia (32%), vomiting (29%), lymphopenia (25%), constipation (25%), and oral mucositis (25%). The most common gr 3-4 AEs were neutropenia (29%, 21% gr 4), anemia (21%, 7% gr 4), febrile neutropenia (21%, all gr 3), thrombocytopenia (14%, all gr 4). After 3 cycles of CHEP-BV the ORR was 100% with 58% CR (14 CR, 10 PR), and at completion of CHEP-BV the ORR was 95% with 90% CR (19 CR, 1 PR, 1 PD). Follow-up data are not yet mature, but only 2 patients have had PD to date.
Conclusions: In pts with CD30-expressing PTCL, induction therapy with CHEP-BV is tolerable and associated with a high CR rate. Longer follow-up time is needed to assess the safety and efficacy of BV consolidation after CHEP-BV (+/- ASCT).
Herrera:Kite Pharma: Consultancy, Research Funding; Immune Design: Research Funding; Pharmacyclics: Research Funding; Adaptive Biotechnologies: Consultancy; Bristol-Myers Squibb: Consultancy, Research Funding; Gilead Sciences: Consultancy, Research Funding; Seattle Genetics: Consultancy, Research Funding; AstraZeneca: Research Funding; Merck: Consultancy, Research Funding; Genentech, Inc.: Consultancy, Research Funding. Zain:Seattle Genetics: Consultancy; Spectrum: Consultancy. Savage:Seattle Genetics, Inc.: Consultancy, Honoraria, Research Funding; BMS, Merck, Novartis, Verastem, Abbvie, Servier, and Seattle Genetics: Consultancy, Honoraria. Feldman:AbbVie: Honoraria, Other: Travel expenses, Speakers Bureau; Pharmacyclics: Honoraria, Other: Travel expenses, Speakers Bureau; Takeda: Honoraria, Speakers Bureau; Celgene: Honoraria, Research Funding, Speakers Bureau; Kite Pharma: Honoraria, Other: Travel expenses, Speakers Bureau; Cell Medica: Research Funding; Trillium: Research Funding; Amgen: Research Funding; Bayer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Kyowa Hakko Kirin: Research Funding; Eisai: Research Funding; Pfizer: Research Funding; Portola Pharma: Research Funding; Roche: Research Funding; Janssen: Honoraria, Speakers Bureau; Seattle Genetics: Consultancy, Honoraria, Other: Travel expenses, Speakers Bureau; Viracta: Research Funding; Roche: Research Funding; Corvus: Research Funding. Brammer:Verastem, Inc: Research Funding; Viracta Therapeutics, Inc.: Research Funding; Bioniz Therapeutics, Inc.: Research Funding. Popplewell:City of Hope: Employment. Budde:F. Hoffmann-La Roche Ltd: Consultancy. Kwak:Pepromene Bio: Consultancy, Equity Ownership, Research Funding; InnoLifes: Consultancy, Equity Ownership; Xeme BioPharma, Inc: Consultancy, Equity Ownership; Enzychem LifeSciences: Consultancy; Celltrion Healthcare: Consultancy; Celltrion, Inc.: Consultancy. Iyer:Novartis: Research Funding; Seattle Genetics, Inc.: Research Funding; Genentech/Roche: Research Funding; Bristol-Myers Squibb: Research Funding; Arog: Research Funding; Incyte: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal