Background
HLA donor-specific antibodies (DSAs) are preformed antibodies found in recipients against donor's HLA antigens. HLA DSAs are generated through the activation of B cells, which differentiate into specific plasma cells for a given recipient HLA determinant. The existence of HLA DSAs forms a significant barrier to the success of allogenic hematopoietic stem cell transplantation (AHSCT) because HLA DSAs are known to cause primary graft failure (PGF). Rates of primary graft failure (PGF) with DSAs have been reported to be between 24 to 83%, and the highest rates are seen in haplo-identical and cord blood transplantation recipients. Currently, there has not been an established method of depleting DSAs due to the long half life of plasma cells. Since there has been an increasing number of alternative or HLA-mismatched AHSCT performed in hematologic malignancies, it is crucial to develop a feasible way of eliminating DSAs in recipients of AHSCT.
CD19 CAR T cells, which have achieved great success in treating B cell malignancies, were shown to have profound efficacy of treating B cell related autoimmune disorders such as lupus in recent mouse model preclinical studies. However, plasma cells were spared in the treatment of lupus with single target CD19 CAR T cells. Additionally, peripheral circulating anti-DNA IgG and IgM autoantibodies remain elevated or increased in treated mice.
Here we present the efficacy of BCMA-CD19 compound CAR (cCAR), which target both B cells and plasma cells, in preclinical study and in our first-in-human phase 1 clinical trial.
Method
We constructed a BCMA-CD19 cCAR, which is a 2-unit CAR composed of a complete BCMA-CAR fused to a complete CD19-CAR by a self-cleaving P2A peptide, enabling independent expression of both CAR receptors separately on the T-cell surface. We then assessed the functional activity of cCAR in co-culture assay with multiple cell lines. We also verified cCAR efficacy with two mouse models each injected with either BCMA-expressing MM.1S cells or CD19-expressing REH tumor cells. In our phase 1 clinical trial, we enrolled children and adults with B-ALL planning to undergo stem cell transplant but were excluded due to high titers of DSAs.
Results
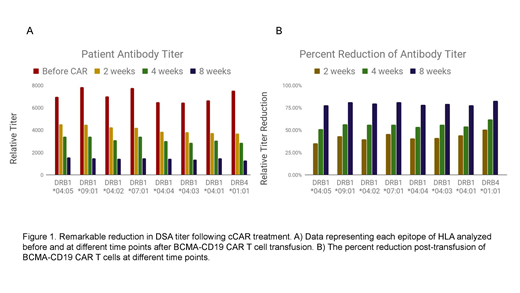
BCMA-CD19 CAR T-cells exhibited robust cytotoxic activity against the K562 tumor cell line, which is synthetically expressing CD19 or BCMA surface antigen, in co-culture assays. These results indicate the ability of each complete CAR domain to specifically lyse target cells. In mouse model study, BCMA-CD19 CAR T cells were able to eliminate myeloma cells in mice injected with MM.1S cells (multiple myeloma cell line) and deplete REH tumor burden in mice injected with REH cells (B acute lymphoblastic cell line). Our preclinical results demonstrated that both components of the compound CAR, BCMA and CD19, are specifically and equally lysing B cells and plasma cells in vivo, making BCMA-CD19 cCAR a candidate for clinical use. In our first-in-human clinical trial, a 48 yr old female patient having treatment resistant B-ALL with high DSA titers achieved remarkable reduction of DSA titer levels following a single dose of BCMA-CD19 cCAR T cells. Patient exhibited complete remission of B-ALL at day 14 post-CAR T cells. Leukemic cells, normal B cells and plasma cells in bone marrow were undetectable by flow cytometry analysis. Total IgM dropped by 80% 2 weeks post-CAR. Eight different DSA antibody titers were also significantly decreased. At 8 weeks post-CAR, all DSA antibodies titers that we examined were reduced by approximately 80% (Figure 1). Updated results in our phase 1 clinical trial will be presented.
Conclusion
Our first in human clinical trial on BCMA-CD19 cCAR demonstrated profound efficacy in reducing DSA levels in patients with B-ALL, which made them eligible candidates for stem cell transplant. Our results further suggested that BCMA-CD19 cCAR has the potential to be applied beyond the realm of hematological diseases and can benefit patients receiving solid organ transplants or those with other antibody-mediated diseases such as lupus, multiple sclerosis and ANCA related autoimmune disorders including microscopic polyangiitis (MPA), granulomatosis with polyangiitis (GPA), or eosinophilic granulomatosis with polyangiitis (EGPA).
Ma:iCAR Bio Therapeutics Ltd: Employment. Wada:iCell Gene Therapeutics LLC: Employment. Ma:iCell Gene Therapeutics LLC: Consultancy, Equity Ownership, Research Funding; iCAR Bio Therapeutics Ltd: Consultancy, Equity Ownership, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal