
BACKGROUND: Venous thromboembolism (VTE) is the second leading cause of death in cancer patients. The treatment of cancer-associated VTE carries a significantly greater risk of major bleeding (MB) and recurrent VTE compared to that in non-cancer patients. CHEST Guidelines suggest the use of low molecular weight heparin (LMWH) over a vitamin K antagonist (VKA) in patients diagnosed with VTE and cancer. The last decade has seen an emergence of non-VKA anticoagulants (NOACs) for the treatment of VTE. Despite completed and ongoing clinical trials, there is a lack of real-world evidence comparing the effectiveness and safety of LWMH with VKAs and NOACs among VTE patients with active cancer. Therefore, this study evaluates the risk of MB, clinically relevant non-MB (CRNMB), and recurrent VTE (fatal or non-fatal) among VTE patients with active cancer prescribed apixaban, LMWH, or warfarin in routine clinical practice.
METHODS: Four US commercial insurance claims databases were used to identify VTE patients with active cancer (defined as cancer diagnosis or cancer treatment [chemotherapy, radiation and cancer-related surgery] within 6 months before or 30 days after VTE diagnosis) who initiated apixaban, LMWH, or warfarin within 30-days following the first VTE event (01SEP2014-31MAR2018). Patients who used LMWH as bridging therapy for warfarin (≤14 days before or after warfarin initiation) were classified as warfarin users. Patients were followed to the earliest of: health plan disenrollment, death, index therapy discontinuation, switch to another anticoagulant, study end, or a maximum of 6 months. Additional analysis using all available follow-up was also conducted. Stabilized inverse probability treatment weighting (IPTW) was used to balance treatment cohorts. Cox proportional hazard models were used to evaluate the risk of MB, CRNMB, and recurrent VTE.
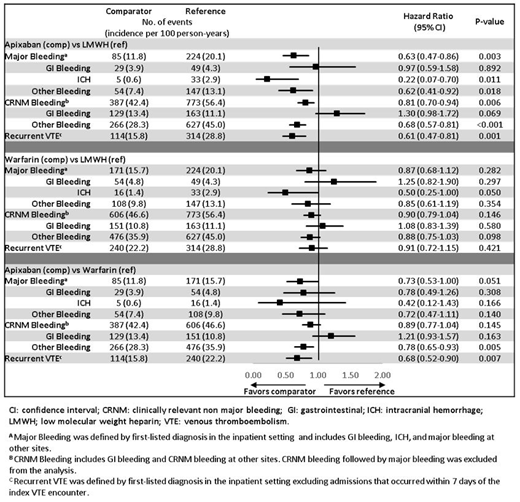
RESULTS: After applying all eligibility criteria, 3,393 apixaban, 6,108 LMWH, and 4,585 warfarin patients were identified with mean ages of 65, 64, and 64 years, respectively. After IPTW, all patient characteristics were balanced. The mean follow-up was 105, 88, and 113 days for apixaban, LMWH, and warfarin, respectively. Among the weighted VTE cancer population, 51% of the patients had metastatic cancer and 77% of patients received cancer treatment. Further, 15% of patients had very high-risk cancer (brain, stomach, or pancreas), and 40% patients had high-risk cancer (lung, lymphoma, gynecologic, bladder, testicular, renal cell carcinoma). Apixaban patients had a lower risk of MB, CRNM bleeding, and recurrent VTE compared to LMWH. Apixaban patients also had a lower risk of recurrent VTE and a similar risk of MB and CRNM bleeding compared to warfarin. Warfarin patients had a similar risk of MB, CRNM bleeding, and recurrent VTE compared to LMWH (Figure). When the entire available follow-up period (mean follow-up was 137, 106, and 166 days for apixaban, LMWH, and warfarin, respectively) was used, the trends were similar to the 6-month analysis for apixaban vs. LMWH and warfarin vs. LMWH. However, apixaban patients were associated with a lower risk of both MB and recurrent VTE compared to warfarin patients.
CONCLUSION: VTE patients with active cancer initiating apixaban had significantly lower risk of MB, CRNM bleeding, and recurrent VTE compared to LMWH patients. Apixaban patients also had a lower risk of recurrent VTE compared to warfarin patients. These results may be helpful for clinicians in evaluating different anticoagulation treatments for VTE patients with active cancer. Further studies are needed to evaluate these outcomes between different anticoagulation treatment options.
Cohen:Lifeblood: Other: advisor to Lifeblood: the thrombosis charity and is the founder of the European educational charity the Coalition to Prevent Venous Thromboembolism; ONO: Consultancy, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Portola: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy; TRN: Consultancy; UK Government Health Select Committee: Other: advised the UK Government Health Select Committee, the all-party working group on thrombosis, the Department of Health, and the NHS, on the prevention of VTE; Temasek Capital: Consultancy; ACI Clinical: Consultancy; Navigant: Consultancy; McKinsey: Consultancy; Bayer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; AbbVie: Consultancy; Aspen: Consultancy, Speakers Bureau; Bristol-Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Boston Scientific: Consultancy; Boehringer-Ingelheim: Consultancy, Speakers Bureau; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; CSL Behring: Consultancy; GlaxoSmithKline: Consultancy, Speakers Bureau; GLG: Consultancy; Guidepoint Global: Consultancy; Johnson and Johnson: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Leo Pharma: Consultancy; Medscape: Consultancy, Speakers Bureau. Keshishian:STATinMED Research: Other: I am a paid employee of STATinMED Research which is a paid consultant to Bristol-Myers Squibb Company and Pfizer Inc.. Lee:Pfizer Inc.: Employment, Equity Ownership. Wygant:Bristol-Myers Squibb Company: Employment. Rosenblatt:Bristol-Myers Squibb: Other: Stock Owner ; Bristol-Myers Squibb Company: Employment. Hlavacek:Pfizer Inc.: Employment. Mardekian:Pfizer Inc.: Employment. Wiederkehr:Pfizer Inc.: Employment. Sah:STATinMED Research: Other: I am a paid employee of STATinMED Research which is a paid consultant to Bristol-Myers Squibb Company and Pfizer Inc.. Luo:Pfizer Inc.: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal