INTRODUCTION
Multiple myeloma (MM) is considered an incurable disease. Despite the introduction of novel agents allowed deeper response, high-dose chemotherapy and autologous stem cell transplantation (ASCT) remain the standard of care for patients (pts) in good clinical conditions. The most used strategies to mobilize stem cells from bone marrow (BM) into peripheral blood are high-dose cyclophosphamide (HD-CTX) plus G-CSF and G-CSF plus plerixafor (G-CSF+P). The goal of this retrospective study is to investigate whether the two different mobilization strategies have an impact on the clearance of monoclonal PCs in the apheresis products and on pts' outcome.
PATIENTS AND METHODS
We analyzed 62 pts (median age 61, range 41-75, 37 males and 25 women) diagnosed with MM and treated with ASCT between Mar 2014 and Mar 2018 at our Hematology Division (Pisa, Italy). All pts received induction therapy with at least 4 cycles of bortezomib, thalidomide and dexamethasone (VTD). 9/62 pts obtained a less than partial response (PR) and received lenalidomide-based regimens. After induction, 8 (12,9%) pts achieved complete remission (CR), 26 (41,9%) were in PR, 28 (45,2%) obtained a very good partial response (VGPR). 43/62 fit pts received HD-CTX (2-3 g/sqm) on day 1 followed by G-CSF (30 MU/day) started on day 4 until day 7, increased to 60 MU/day from day 8 until the end of apheresis. In 19/62 pts, after 4 days of G-CSF (60 MU/day) administration and not sufficient mobilization, we added plerixafor (0,24 mg/kgbw) for up to 4 consecutive days. In 43/62 pts we collected apheresis samples (10μl) analyzed through flow citometry to enumerate clonal residual PCs. The panel used to asses clonality included: CD138 Per-Cp, CD38 APC, CD19 PE-Cy7, CD45 APC-Cy7, cytoplasmic immunoglobulin K chain and L chain.
RESULTS
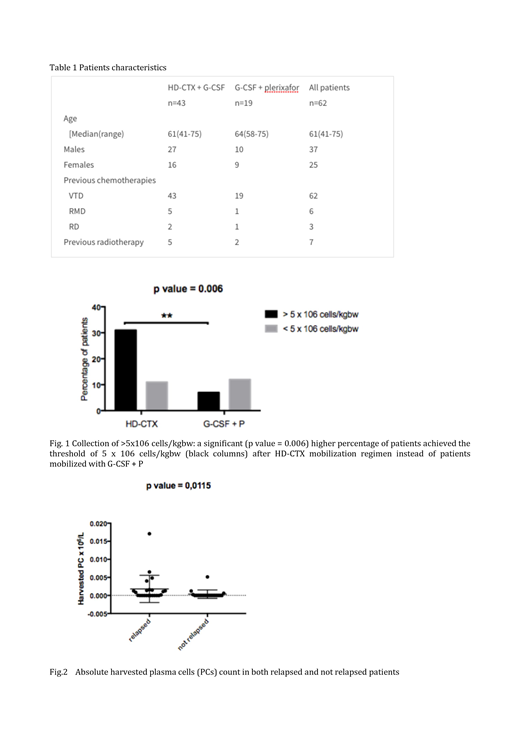
At the end of the peripheral blood stem cell (PBSC) collection, pts treated with HD-CTX presented a higher CD34+ absolute count (p=0.0489) and achieved the threshold of 5x106 CD34+ cells/kgbw in a significantly (p=0.006) higher percentage. We found a nearly significant (p=0.0517) lower count of CD34+ PBSCs in pts who received lenalidomide-based regimens before the mobilization. Performing flow citometry on apheresis samples, we observed that the number of the harvested clonal PCs showed a significant correlation (p=0.0115) with the occurrence of post-ASCT relapse. ROC curve analysis investigating the predictive effect of the number of pathological PCs on disease relapse showed an area under the curve of 0,6978 (95% CI 0.5392-0.8564; p=0.0267). Neither BM residual PCs detectable on BM biopsies performed before apheresis (r=-0.1323; p=0.609) nor the type of mobilization scheme (p=0.707) had an impact on the proportion of clonal PCs in the graft. Additionally, we did not observe any statistically significant difference in progression free- (PFS) (p=0.8276) and overall survival (OS) (p=0.2475) between the HD-CTX and G-CSF+P groups.
DISCUSSION
PBSC mobilization has a succession rate > 85%. Despite the use of HD-CTX to increase PBSC yields and decrease tumor burden, there is not clear evidence of a superior mobilization strategy. Additionally, HD-CTX has a not negligible toxicity and approximately 10% of the pts require hospitalization. Conversely, G-CSF+P is a safe and effective approach also in poor mobilizers. In our study, we observed a significative difference in the apheresis yields (p=0.0489) and in the percentage of pts who achieved the threshold of 5x106 CD34+ cells/kgbw (p=0.006) in favor of HD-CTX. Additionally, the detection of harvested residual clonal PCs could be a promising strategy to recognise pts more likely to relapse after ASCT. Nonetheless, we failed to demonstrate a superior effect of HD-CTX in the clearance of harvested clonal PCs, in agreement with the absence of a different pts' outcome amongst the two mobilization strategies. In conclusion, the choice between the two regimens is challenging and requires careful consideration of multiple factors. Overall, young fit pts, especially in the high-risk setting, should be treated with all appropriate modalities including chemiomobilization followed by double-ASCT. Conversely, in pts candidate to a single-ASCT it is reasonable to use G-CSF+P, since HD-CTX does not improve PFS and OS and add toxicity. The absence of an in-vivo purging effect on apheresis products of chemiomobilization further strengthens a chemotherapy-free mobilization.
Galimberti:Roche: Speakers Bureau; Celgene: Speakers Bureau; Novartis: Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal