Background:
Solitary plasmacytoma (SPC) is a rare plasma cells disorder, which may present as a single bone lesion or an extramedullary plasmacytoma. High dose radiotherapy may be curative, yet most patients eventually develop Multiple Myeloma (MM). Due to rarity of the disease, there is uncertainty regarding the optimal treatment strategy, especially in patients that present with known risk factors. The role of adjuvant anti-myeloma therapy is still under debate, whereas few studies have reported efficacy of novel agents.
We aimed to explore clinical characteristics and treatment patterns and to identify predictors for relapse and progression to myeloma in a real world setting.
Methods:
Consecutive patients, diagnosed and treated for solitary plasmacytoma in 13 centers, between 2008 and 2019 (defined by the IMWG criteria) were identified, based on medical centers patient registries and pathological reports. Patient's medical charts were reviewed, and data regarding patient and disease characteristics, treatment regimen, and clinical endpoints, focusing on response to treatment and time to disease progression were documented. The treatment response categories were defined according to the IMWG criteria.
Results:
62 patients (pts) were identified. Baseline characteristics and workup are detailed in the table. Median follow-up was 39 months (mo) (IQR: 13.5, 63.3). Fifty nine pts (95.2%) were treated with radiotherapy, with a median dose of 45.0 Gy (range: 30-54 Gy); radiotherapy dose was 45-54 in 31 pts, < 45Gy in 11 pts. Twenty pts (33.9%) underwent surgery in addition to radiotherapy, 3 more pts underwent a surgery alone. The median time to radiotherapy since diagnosis was 2.7 months (range: 1day-18.2 months), 8 pts received any anti-myeloma therapy for treatment of plasmacytoma including Melphalan/dexamethasone (n=1), bortezomib based induction followed by ASCT (n=2), VD/VTD (n=2) and KRD (n=1).
Among 25 patients with evaluable post-therapy PET-CT response (performed 12 months post radiotherapy), 26% had no remaining FDG uptake, 20% had a 50% or greater decrease in FDG uptake. Among 40 patients with CT or MRI post therapy assessment, 20% had disappearance of SPC, 27% had ≥50% reduction, 7% had <50% reduction and 25% remained without change. Non-bone SPC were more likely to achieve CR than bone SPCs, 86% vs 29%, respectively, p=0.021.
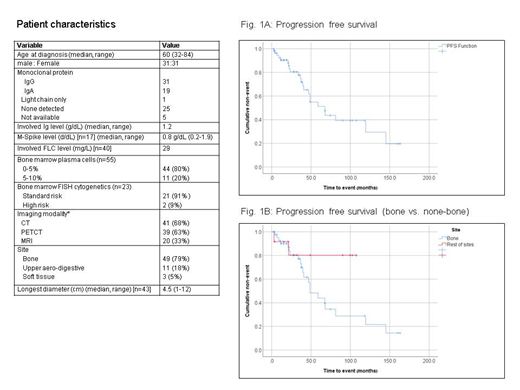
During the follow-up period, 16 pts (25.8%) experienced evolution to MM (15 pts had bony and 1 soft tissue plasmacytoma in diagnosis). Among patients with bone SPC (n=49), 30.5% evolved to MM. A total of 6 pts had a recurrence of SPC (5 pts had a bony and 1 patient upper aerodigestive plasmacytoma in diagnosis). 5 pts died (3 without recurrence or evolution to MM). Median time to plasmacytoma recurrence in pts that achieved CR was 36.7 mo (range: 3 -67.9 mo); Median time of evolution to MM was 37.5 mo (range: 6.7-87.2 mo). Progression free survival (PFS) was 67.7 mo for all pts (Fig1A), 48 mo for bone SPC and was not reached for non-bone SPC (p=0.11) (Fig1B). Median overall survival was not reached. Overall survival at median follow-up was 93.6%.
Notably, all 8 pts who were treated with anti-myeloma therapy had a bony plasmacytoma and received radiotherapy in addition. None of these pts had recurrence of plasmacytoma or evolution to MM. Of the 11 pts that had SPC in an upper aerodigestive tract site, none evolved to MM; 10 of these pts were treated with only radiotherapy, one had a plasmacytoma recurrence in the same site.
Conclusions:
We present real world data of a multicenter patient cohort with SPC. As previously suggested, prognosis is more favorable for none bone SPCs. Though numbers are modest, our analysis suggests a benefit of the early addition of systemic therapy to standard radiotherapy for patients with solitary bone plasmacytoma.
Yael:Janssen: Consultancy; Amgen: Consultancy, Research Funding; Neopharm: Consultancy; Takeda: Consultancy; Madison: Consultancy; VBL Therapeutics: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal