Introduction:
Carfilzomib use as a single agent or in combination has been shown to improve outcomes in relapsed refractory multiple myeloma (RRMM). Traditionally, carfilzomib has been administered in twice-weekly dosage schemes as compared to the newly approved once-weekly dosage scheme. The once-weekly dosing of carfilzomib is thought to provide same efficacy and has been evaluated in many clinical trials. We have performed a systematic review of comparative efficacy and toxicity profiles of once and twice-weekly carfilzomib dosing schemes in RRMM.
Materials and methods:
Systematic review was done according to PRISMA statement. PubMed, Embase, AdisInsight, & Clinicaltrials.gov databases were searched, without any filters, using the search terms "Multiple Myeloma" AND "Kyprolis" OR "Carfilzomib". Most recent reports of phase I/II/III clinical trials reporting the clinical efficacy and/or safety of carfilzomib in RRMM were considered for inclusion. After screening of 267 articles, 16 clinical trials (N=2393) evaluating twice-weekly carfilzomib and 4 trials (N=418) evaluating once-weekly dosing schedule were considered for inclusion.
Results
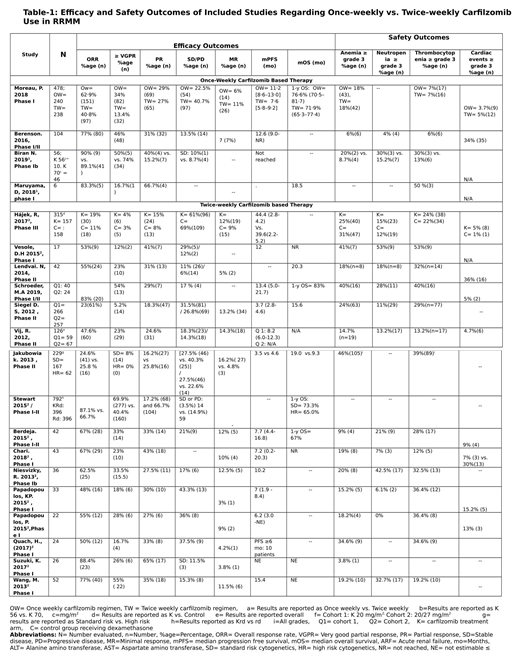
Once versus (vs.) twice-weekly carfilzomib regimens were evaluated in a total of 406 and 1869 patients, respectively. In our discussion, group-1 refers to patients receiving once weekly carfilzomib while group-2 refers to twice weekly dosing schedule group. For the evaluated population, high risk cytogenetics were reported in 15% (n=60) patients in group-1 and in 17% (n= 317) patients in group-2. The median no. of prior lines of therapy ranged 1-8 in group-1 as compared to 1-20 in group-2. All studies reported the patient responses according to International Myeloma Working Group (IWMG) response criteria. The cumulative overall response rate (ORR) was better in group 1: 70.4% (n= 286) vs. 48% (n= 896). Similarly, more patients in group-1 achieved stringent complete response (sCR) or complete response (CR): 11.5% (n= 47) patients in group 1 vs. 5.2% (n= 99) patients in group 2. The response rates were also non-inferior in group-1 for other responses as overall. In group-1, 30% (n= 123) patients achieved very good partial response (VGPR) compared to 21% (n=389) patients in group-2. The partial response (PR) rate was 28.5% (n= 116) vs. 22% (n= 412) in groups 1 and 2 respectively
The rate of minimal response (MR) achievement was comparable in both groups: 5% (n= 21) in group-1 vs. 8% (n= 154) patients in group-2. But lesser proportion of patients in group-1 went on to develop progression of myeloma disease [5.6% (n= 23) vs. 15.6% (n= 293)]. Stable disease (SD) state was achieved by 12.3% (n=50) patients in group-1 compared to 20% (n= 379) patients in group-2. Owing to phase-1 design of most clinical trials evaluating the once-weekly dosing schedule, the survival data is immature at present, but the median progression free survival (PFS) ranged from 1 day to 12.6 months in group-1 while in group 2 it ranged from 3.7-44.4 months. (Table-1)
The toxicity profiles were also comparable for both dosing schedules. The most common hematological adverse events (AEs) were anemia and thrombocytopenia and their incidences were 15.7% (n= 64) vs. 25.5% (n= 477) and 13.3% (n= 54) vs. 23.1% (n= 432) in groups 1 and 2 respectively. The most common non-hematological AEs were infections, fatigue, and nausea that were reported in 22% (n= 89), 19% (n= 77), and 17.4% (n= 71) patients in group 1, and 14.5% (n= 272), 19.3% (n= 362), and 19.1% (n= 357) patients in group 2, respectively. Overall, grade 3 or above cardiac AEs were reported in 3.4% (n= 14) patients in group 1 and 3.3% (63/1869) patients in group 2. The most common cardiac AE was hypertension which constituted 50% (n= 7) and 64% (n= 40) of all grade 3 or more cardiac AEs in groups 1 and 2, respectively.
Conclusion
Achievement of objective response rates for once-weekly carfilzomib appears to be better or at-least similar to twice-weekly carfilzomib regimens. There is paucity of prospective studies comparing once vs. twice-weekly carfilzomib in RRMM which highlights the need for more clinical trials evaluating this regimen. Apart from efficacy and safety profile, once-weekly carfilzomib dosing also carries the potential of becoming a preferred option in terms of lesser financial burden and number of patient visits to the infusion centers.
Anwer:In-Cyte: Speakers Bureau; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal