Introduction: The RESPONSE trial (NCT01243944) compared ruxolitinib (Rux) and best available therapy (BAT) in pts with polycythemia vera (PV) who were intolerant of or resistant to hydroxyurea (HU) according to modified European LeukemiaNET criteria. In the primary analysis, at week (Wk) 32, 60% of (pts) randomized to Rux achieved HCT control (HCT <45%). The present analysis evaluated the effect of baseline characteristics on HCT response at Wk 32, and aimed to determine the long-term clinical efficacy of Rux in pts who did and did not achieve the protocol-defined HCT control (i.e., HCT control responders and non-responders at Wk 32) in RESPONSE at Wk 256.
Methods: Adult pts with phlebotomy-dependent PV with splenomegaly, and resistant to or intolerant of HU were enrolled. Pts were randomized to receive Rux (at a starting dose of 10 mg BID) or single-agent BAT (1:1). HCT control was defined as lack of phlebotomy eligibility between Wks 8−32 with no more than 1 phlebotomy eligibility between randomization and Wk 8. Phlebotomy eligibility was based on protocol-defined HCT values (HCT > 45% and ≥ 3 percentage points higher than baseline or > 48%, whichever was lower; regardless of receipt of phlebotomy), and pts with missing data or assessments outside of protocol-defined time windows were considered non-responders. In this analysis, a logistic regression model was fitted to identify the significant baseline factors to predict HCT control response at Wk 32. Time to phlebotomy eligibility in the HCT control responders and time from the first phlebotomy eligibility to the second phlebotomy eligibility in the HCT control non-responders were plotted, and the changes in hematological parameters (HCT, WBC and platelet count), spleen volume and allele burden over time, up to Wk 256, were studied in HCT control responders and non-responders who were randomized to Rux treatment arm in RESPONSE.
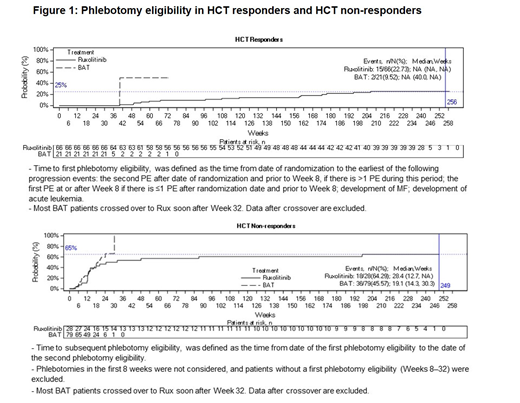
Results: A total of 222 pts were randomized to receive either Rux (n = 110) or BAT (n = 112). Baseline WBC (P=0.0198) and baseline JAK2 V617F allele burden (P=0.0159), were found to be predictors of the HCT response within Rux treated pt group (n = 110). In the HCT responder subgroup of the Rux arm, 23% (15/66) pts needed their first phlebotomy by Wk 256. In the HCT non-responder subgroup of the Rux arm, out of 28 patients who experienced their first phlebotomy between Wk 8 and Wk 32, 64% (18/28) of pts required subsequent phlebotomy by Wk 256, with a median duration of 28.4 Wks (12.7, NA). Pts receiving Rux demonstrated controlled hematologic parameters (HCT, WBC, and platelets) over the course of study, regardless of whether they were HCT control responders and HCT control non-responders at Wk 32. From Wk 48 to Wk 80, 97% HCT control responder pts and 84% HCT control non-responder pts of the Rux treatment arm required no phlebotomies. From Wk 80 to Wk 256, 91% and 68% of the evaluable pts in the Rux treatment arm remained phlebotomy-free for HCT control responders and non-responders, respectively. By Wk 256, spleen volume on an average was reduced from baseline by approximately 35% and 50% for HCT control responders and non-responders, respectively. In pts with available assessments, allele burden on an average was reduced approximately from 80% at baseline to 55% at Wk 256 in the HCT control responders, and approximately from 70% at baseline to 40% at Wk 256 in the HCT control non-responders.
Conclusions: The results from present analysis demonstrated that the benefits of the Rux treatment were not limited to pts who achieved HCT control at Wk 32. Patients treated with Rux were able to maintain hematological parameters, spleen volume reduction, and JAK2 V617F allele burden reduction for a longer duration (up to 5 years), regardless of whether they were HCT control responders or non-responders at Wk 32.
Verstovsek:Constellation: Consultancy; Pragmatist: Consultancy; Incyte: Research Funding; Roche: Research Funding; NS Pharma: Research Funding; Celgene: Consultancy, Research Funding; Gilead: Research Funding; Promedior: Research Funding; CTI BioPharma Corp: Research Funding; Genetech: Research Funding; Blueprint Medicines Corp: Research Funding; Novartis: Consultancy, Research Funding; Sierra Oncology: Research Funding; Pharma Essentia: Research Funding; Astrazeneca: Research Funding; Ital Pharma: Research Funding; Protaganist Therapeutics: Research Funding. Kiladjian:Novartis: Honoraria, Research Funding; Celgene: Consultancy; AOP Orphan: Honoraria, Research Funding. Wroclawska:Novartis Pharma AG: Employment. Dong:Novartis: Employment. Vannucchi:Celgene: Membership on an entity's Board of Directors or advisory committees; CTI BioPharma: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Incyte: Membership on an entity's Board of Directors or advisory committees; Italfarmaco: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal