Introduction: Front-line immunochemotherapy (IC) with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) is expected to cure 60-70% of newly diagnosed DLBCL. Up to one third of newly diagnosed DLBCL patients will have relapsed or refractory (r/r) disease.1 Current standard of care for these patients is salvage chemotherapy and, if chemosensitive, to be followed by high dose chemotherapy with hematopoietic cell rescue (autoHCT).2,3
Event-free survival at 24 months (EFS24) after frontline IC is associated with excellent long-term outcomes with overall survival (OS) similar to that of age- and sex-matched controls.4 In comparison, those achieving EFS24 following autoHCT for relapsed or refractory disease may have increased risk for late-mortality compared to the general population with advanced age and intensive salvage therapy contributing to the risk of late complications.5 We sought to better characterize EFS24 after autoHCT to determine the utility of this end-point for informed clinical decisions, patient management, and future clinical trials.
Methods: Patients were prospectively enrolled onto the Molecular Epidemiology Resource (MER) of the University of Iowa/ Mayo Clinic Specialized Program of Research Excellence (SPORE). Patients were followed for relapse, retreatment, and death; all events were validated by medical record review. For this analysis patients were included if they were consented within 9 months of initial diagnosis of DLBCL between 2002-2012, had received anthracycline-based IC (R-CHOP, or similar), and eventually had undergone autoHCT for r/r DLBCL. Patients with primary central nervous system (CNS) lymphoma or post-transplant lymphoproliferative disorders (PTLD) were excluded. Overall survival (OS) was defined as time from autoHCT until death due to any cause. Event-free survival (EFS) was defined as time from autoHCT until progression, relapse, retreatment, or death due to any cause. OS from achieving EFS24 after autoHCT was compared to age- and sex-matched general US population.
Results: 108 patients underwent autoHCT for relapsed DLBCL between 2002 and 2017. Median age at autoHCT of the patients was 60 years (range 20-78) and 72 (67%) were male. The most common transplant conditioning regimen was BEAM (82%). At a median follow-up after autoHCT of 85 months (range 1-171), 72 patients (67%) had an event and 64 patients (59%) had died. Kaplan-Meier estimates for EFS and OS at 24 months from the time of autoHCT were 49% (95% CI: 40-59) and 61% (95% CI: 52-71), respectively.
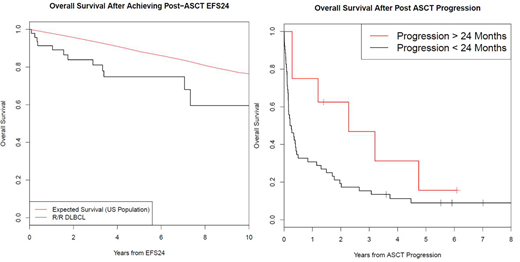
52 patients had a progression within 24 months of autoHCT; OS from progression was poor (median OS=2.8 months, 95% CI: 1.8-6.0; 5 year OS = 9%, 95% CI: 2-22). 48 patients achieved EFS24 after autoHCT; median OS from achieving EFS24 was 136 months (95% CI: 92-NE) and 5 year OS was 79% (95% CI: 68-93). This was inferior to the background population (Figure 1, SMR=3.64, 95% CI: 2.11-6.27, p<0.0001). Eight patients had a progression after achieving EFS24 from autoHCT. OS from progression in these 8 patients (median OS=27.3 months, 95% CI: 14.4-NE; 5 year OS = 16%, 95% CI: 3-93) was improved compared to progression within 24 months of autoHCT (p=0.072, figure B). Cause of death (COD) in EFS24 achievers was progression of lymphoma (n=6), infection (n=1), secondary malignancy related to therapy (n=3), heart disease (n=1), and unknown (n=1).
Conclusions: Patients achieving EFS24 after salvage chemotherapy and autoHCT have a favorable long-term prognosis; however, overall survival remained inferior to the general population. Most common COD after achieving EFS24 was progression of lymphoma. In spite of this, EFS24 remains a valuable end-point for informed clinical decisions, patient management, and future clinical trials.
Maurer:Morphosys: Membership on an entity's Board of Directors or advisory committees; Celgene: Research Funding; Nanostring: Research Funding. Ansell:Mayo Clinic Rochester: Employment; Bristol-Myers Squibb: Research Funding; Affimed: Research Funding; Trillium: Research Funding; Regeneron: Research Funding; Seattle Genetics: Research Funding; Mayo Clinic Rochester: Employment; Affimed: Research Funding; Bristol-Myers Squibb: Research Funding; LAM Therapeutics: Research Funding; Regeneron: Research Funding; Seattle Genetics: Research Funding; Trillium: Research Funding; LAM Therapeutics: Research Funding. Cerhan:Celgene: Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees; NanoString: Research Funding. Farooq:Celgene: Honoraria; Kite Pharma: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal