Introduction:
Six cycles of rituximab plus infusional EPOCH is considered a preferred regimen for first-line treatment of HIV-associated diffuse large B-cell lymphoma (DLBCL), HHV8-positive DLBCL, and primary effusion lymphoma, and is among the preferred regimens for HIV-associated Burkitt's lymphoma in the 2019 NCCN guidelines. A phase III trial demonstrated non-inferiority of 4 cycles of R-CHOP (followed by 2 additional doses of rituximab) compared with 6 cycles of R-CHOP in immunocompetent patients with low-risk DLBCL (stage I-II, age 18-60 years, and an age-adjusted International Prognostic Index score of 0), indicating that de-escalation of treatment duration may be safely achieved without compromising curability in an appropriately selected patient population. Here we report the outcomes for patients with HIV-associated DLBCL and high-grade non-Hodgkin lymphoma (NHL) treated with 4-6 cycles of EPOCH plus rituximab based a response-adapted treatment strategy under a completed prospective clinical trial (AMC-034).
Methods:
One hundred-six patients with HIV-associated DLBCL or high-grade CD20-positive NHL enrolled at multiple centers across the U.S. were randomized to receive rituximab (375 mg/m2) given either concurrently prior to each infusional EPOCH cycle, or sequentially (weekly for 6 weeks) following completion of dose-adjusted EPOCH regimen tailored for HIV+ patients. EPOCH consisted of a 96-hour IV infusion of etoposide, doxorubicin, and vincristine plus oral prednisone followed by IV bolus cyclophosphamide every 21 days for 4 to 6 cycles. Patients received 2 additional cycles of therapy after documentation of a complete response (CR) by computerized tomography after cycles 2 and 4.
Results:
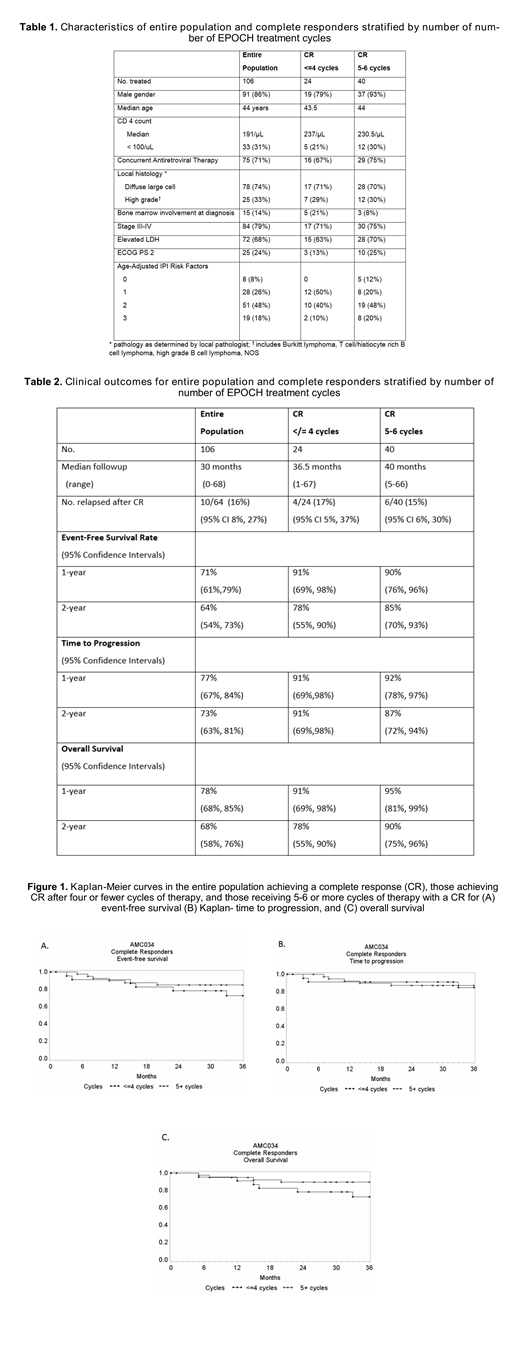
Sixty-four of 106 patients (60%, 95% CI 50%, 70%) had a CR in both treatment arms; characteristics of entire population and CR proportions stratified by number of EPOCH treatment cycles were similar (Table 1).The 2-year event-free survival (EFS) rates were similar in the 24 patients with CR who received 4 or fewer EPOCH cycles (78%, 95% confidence intervals [55%, 90%]) due to achieving a CR after 2 cycles, compared with those who received 5-6 cycles of EPOCH (85%, 95% CI 70%, 93%) due to failure to achieve a CR after 2 cycles (Table 2 and Figure 1). Time to disease progression and overall survival wer also similar for those treated with 4 or fewer cycles compared with 5-6 cycles (91% vs. 87%, and 78% vs. 90%, respectively) (Table 2 and Figure 1).
Conclusion:
A response-adapted strategy may permit a shorter treatment duration without compromising therapeutic efficacy in an appropriately selected population of patients with HIV-associated NHL, which merits further evaluation in additional prospective trials.
Noy:Janssen: Consultancy; Medscape: Honoraria; Prime Oncology: Honoraria; NIH: Research Funding; Pharamcyclics: Research Funding; Raphael Pharma: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal