Introduction: Idelalisib (IDELA, Zydelig®) is the first-in-class PI3Kδ inhibitor and is approved in the U.S. as an oral monotherapy for relapsed / refractory follicular lymphoma (R/R FL) after at least two prior lines of systemic therapy. IDELA's regulatory approval was based on a phase 2, open-label clinical trial in 125 patients with R/R indolent non-Hodgkin's lymphoma (Gopal et al., NEJM, 2014) and outcomes in the FL subgroup were published by Salles et al. (Haematologica, 2017). The current study evaluates the characteristics and treatment patterns of patients treated with IDELA for R/R FL in a real-world setting.
Methods: Adult patients diagnosed with R/R FL (grades 1, 2, and 3a) and treated with IDELA within the US Oncology Network (USON) between 7/1/2014 to 6/30/2018 were analyzed retrospectively. Patient data were obtained from USON's structured electronic health records' system, iKnowMed (iKM)TM. Manual chart review (ChR) was used to determine physician response and to confirm IDELA treatment patterns. Overall survival (OS) and progression-free survival (PFS) were estimated using Kaplan-Meier methods. Descriptive statistics were generated for outcomes of interest, including duration of therapy (DoT), median follow-up, and adverse event (AE) frequency.
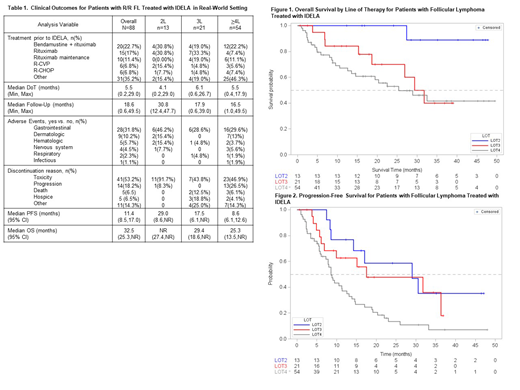
Results: A total of 124 patients with FL and prescribed IDELA were identified in iKM TM. After Chr confirming the diagnosis of follicular lymphoma diagnosis and initiation of IDELA, 88 patients were retained for analysis. Median age of patients was 68.9 years, with 52.3% female and the majority white and non-Hispanic (90.9% and 93.2%, respectively, Table 1). The most common regimens immediately prior to IDELA initiation were bendamustine + rituximab (22.7%), rituximab (17%), and rituximab maintenance (11.4%). Eighty-six (97.7%) patients had co-morbidities categorized as vascular (50%), endocrine (33%), respiratory (13.6%), or cardiac (12.5%). Thirteen (14.8%), 21 (23.9%), and 54 (61.4%) patients initiated IDELA in second line (2L), 3L, and >4L, respectively. Baseline lab values at IDELA initiation were similar regardless of line of therapy (LOT). mDOT was 5.5 mos. for the entire population and was similar across all LOTs (4.1 mos., 6.1 mos., and 5.5 mos. in 2L, 3L, and >4L, respectively). AEs were noted in 45.5% with the most common being gastrointestinal (31.8%) and dermatologic (10.2%). Respiratory and infectious AEs were noted in 2.3% and 1.1%, respectively, although Pneumocystis jirovecii pneumonia (PJP) prophylaxis was rarely prescribed (2.3%). Toxicity as a reason for IDELA discontinuation varied in frequency across LOT and was more common in 2L compared to 3L and >4L (91.7% compared to 43.8% and 46.9%, respectively). With a median follow-up of 18.6 months for the population, the mPFS was 11.4 mos. [95%CI: 8.5,17.0] and mOS was 32.5 mos. [95% CI: 25.3,NR]. Stratified by LOT, median follow-up time, mOS, and mPFS were greater in 2L (30.8 mos., NR [95% CI: 27.37,NR], and 29.0 mos. [95% CI: 8.6,NR], respectively) than in 3L or >4L (3L: 17.9 mos., 29.4 mos. [95%CI: 18.6,NR], and 17.5 mos. [95% CI: 6.1,NR]; >4L: 16.5 mos., 25.3 mos. [95%CI: 13.5.,NR], and 8.6 mos. [95% CI: 6.1,12.6], respectively, Figures 1 and 2).
Conclusion: Findings from this analysis suggest that R/R FL patients treated with IDELA in a real-world setting experience a similar mDOT and mPFS as those treated in the clinical trial setting (Salles et al., Haematologica, 2017). Patients treated in 2L demonstrated longer PFS and OS compared to later lines, but also experienced increased IDELA discontinuation due to toxicity, perhaps reflecting a lower incidence of progressive disease in earlier treatment lines, or a more immunocompetent population leading to higher rates of autoimmune AEs. Use of PJP prophylaxis in IDELA-treated patients was uncommon, an observation suggesting an opportunity for provider education. Our findings enhance available data on relapsed FL patient outcomes in real-world clinical practice and support the use of IDELA in patients with R/R FL after at least 2 systemic therapies.
Andorsky:Gilead: Research Funding; Genetech: Research Funding; CTI: Research Funding; AstraZeneca: Consultancy; Celgene: Research Funding. Chan:Gilead Sciences, Inc.: Employment, Equity Ownership. Clark:McKesson: Consultancy, Employment, Equity Ownership. Ruzicka:Gilead Sciences, Inc.: Employment. Robert:McKesson: Employment. Awan:Pharmacyclics: Consultancy, Research Funding; AstraZeneca: Consultancy, Speakers Bureau; Abbvie: Consultancy, Speakers Bureau; Janssen: Consultancy; Genentech: Consultancy; Sunesis: Consultancy; Gilead: Consultancy.
Idelalisib is a PI3 kinase inhibitor indicated for the treatment of patients with relapsed follicular B-cell lymphoma who have received at least two prior systemic therapies. Some patients in this observational study used Idelalisib after one prior systemic treatment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal