Introduction: The immune response to factor VIII (fVIII) is a CD4+ T cell dependent process initiated by fVIII recognition and processing by antigen presenting cells. The C1 and C2 domains have been proposed as the primary domains that mediate fVIII internalization by dendritic cells. Our prior studies demonstrate that fVIII pre-bound to anti-C1 and C2 domain monoclonal antibodies (MAbs) reduces fVIII internalization by murine derived dendritic cells. However, anti-A1 and A3 domain antibodies increase fVIII endocytosis by dendritic cells. In this study, we analyzed the antibody titers of hemophilia A mice immunized with binary fVIII and MAb complexes to determine whether changes in fVIII internalization observed in vitro correspond to changes in the immune response to fVIII in vivo.
Methods: Exon16 (E16) knockout mice deficient in fVIII were immunized with B-domain deleted fVIII in the presence of either anti-fVIII MAb 2-116 (anti-A1, IgG2a), 4A4 (anti-A2, IgG2a), 2-113 (anti-A3, IgG1), B136 (anti-C1, IgG2a), or 3D12 (anti-C2, IgG2b) versus fVIII alone. Mice were immunized by weekly retro-orbital injections of 0.1 µg fVIII incubated with 1 µg anti-fVIII MAb for 4 weeks followed by a boost dose of 0.2 µg fVIII and 2 µg anti-fVIII MAb one week later at week 5. Anti-fVIII ELISA titers (in arbitrary units, AU) were analyzed from plasma samples collected at week 7 to account for IgG half-life. A separate cohort of mice immunized with anti-fVIII MAbs alone served as controls to normalize ELISA titers in each fVIII/MAb group to account for residual injected MAbs in the plasma samples.
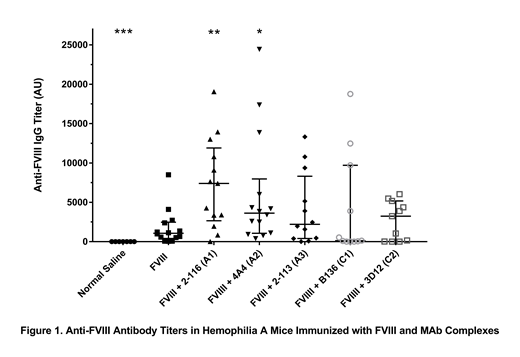
Results: Hemophilia A mice immunized with fVIII/2-116 and fVIII/4A4 complexes significantly increased anti-fVIII ELISA titers compared to mice immunized with fVIII alone (figure 1). Mice immunized with fVIII/2-116 and fVIII/4A4 had median ELISA titers of 7,401 AU (interquartile range, IQR, 2,654 - 11,910 AU) and 3,620 AU (IQR 1,062 - 7,969 AU), respectively, compared to mice immunized with fVIII (median titer 1,063 AU, IQR 402 - 2,476 AU). MAb 2-116 is a non-inhibitory antibody with a titer of <1 Bethesda Unit (BU)/mg IgG, while 4A4 is a highly inhibitory antibody with a titer of 40,000 BU/mg IgG. Neither MAb interferes with fVIII binding to von Willebrand factor (VWF) or phospholipid vesicles. Mice immunized with fVIII/2-113 (median titer 2,210 AU, IQR 402 - 8,318 AU), fVIII/B136 (median titer 123 AU, IQR 0 - 9,709 AU), and fVIII/3D12 (median titer 3,244 AU, IQR 0 - 5,180 AU) did not have significantly different anti-fVIII ELISA titers compared to mice immunized with fVIII. However there was a trend towards reduced anti-fVIII titers with fVIII/B136 and fVIII/3D12 injections. MAbs B136 and 3D12 inhibit fVIII binding to VWF and have been shown to significantly increased fVIII clearance in hemophilia A mice compared to fVIII in a VWF-dependent manner. Median titers for mice immunized with MAbs alone to account for residual injected MAbs were 13 AU (2-116), 12 AU (4A4), 18 AU (2-113), 16 AU (B136), and 4 AU (3D12).
Conclusions: Immunization of hemophilia A mice with fVIII/MAb complexes, specifically anti-A1 MAb 2-116 and anti-A2 MAb 4A4, enhance the immune response to fVIII. MAb 2-116 significantly increased anti-fVIII antibody titers in vivo, which correlates with increased fVIII internalization by immature dendritic cells observed in vitro. A better understanding of the effect of anti-fVIII antibodies on fVIII conformational changes could provide insight into whether these changes alter fVIII recognition by immune cells and subsequently propagate the immune response to fVIII at the onset of inhibitor formation or during immune tolerance induction.
Batsuli:Genentech: Other: Advisory board participant; Bayer: Other: Advisory board participant; Octapharma: Other: Advisory board participant. Meeks:HEMA Biologics: Membership on an entity's Board of Directors or advisory committees; Novo Nordisk: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees; Bioverativ: Membership on an entity's Board of Directors or advisory committees; Takeda-Shire: Membership on an entity's Board of Directors or advisory committees; Bayer: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal