Background: Chimeric antigen receptor (CAR) T-cell therapy has been a major innovative breakthrough for hematologic malignancies with 2 currently FDA approved CAR T-cell products (tisagenlecleucel and axicabtagene ciloleucel) and several others in different stages of clinical investigation. However, these therapies are associated with unique safety profiles and potentially serious toxicities, including cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity (ICANS), that require vigilant monitoring and prompt recognition and management to ensure patient safety and optimal therapeutic benefit. We developed an online interactive decision support tool at www.clinicaloptions.com/carttool to give healthcare providers (HCPs) case-specific, evidence-based guidance from experts on the management of adverse events (AEs) due to CAR T-cell therapy. Here, we report a comparison of CAR T-cell toxicity management among HCPs using the tool vs the expert consensus recommendations in the tool.
Methods: In March 2019, a panel of 5 experts provided consensus guidance for the screening, prophylaxis, monitoring, and management of CRS and ICANS in patients for which CAR T-cell therapy was either planned or started. This information was used to build the interactive online tool. To use the online tool, HCPs enter the AE that the patient is experiencing, either CRS or ICANS; the grade or severity of the event, per the American Society for Transplantation and Cellular Therapy consensus grading for CRS and ICANS (Lee DW, et al. Biol Blood Marrow Transplant. 2019;25:625-638); and their planned management approach. The HCPs were then shown the expert management recommendation for that specific AE scenario. After viewing the expert management recommendation, HCPs were asked if it impacted their intended management approach.
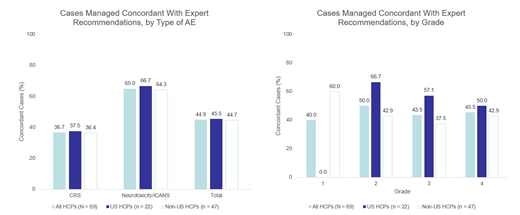
Results: Between May and July 2019, 115 HCPs entered 166 unique case scenarios into the tool. The majority of cases (58%) entered were for patients who were planned for CAR T-cell therapy or who had started therapy without yet experiencing an AE, for which users received expert recommendations on pretreatment screening along with AE prophylaxis and monitoring. Of the 69 cases entered for patients who had received CAR T-cell therapy and were experiencing an AE, 71% were CRS and 29% were neurotoxicity/ICANS. The majority of CRS cases (67%) were intermediate grade (2/3) whereas the ICANS cases were evenly distributed across all grades (1-4). Overall the planned toxicity management strategy of HCPs matched the expert recommendations in 45% of cases, with the greatest discordance for CRS management, where the rate of agreement was 37% (Figure). The proportion of cases in which the planned management strategies of HCPs matched expert recommendations also varied by syndrome grade/severity between US and non-US HCPs. There was no concordance (0%) among US HCPs compared with non-US HCPs (60%) for grade 1 AEs, whereas greater concordance was found in the management of grade 2 and grade 3 AEs among US HCPs (67% and 57%, respectively) compared with non-US HCPs (43% and 44%, respectively; Figure). Of the 15 grade 1 AEs entered by users, only 5 came from US HCPs. Among the 48% of HCPs who answered the optional survey on the impact of the tool on their intended management plan, 48% indicated that the expert recommendations changed their approach, and 80% reported practicing at a treatment center authorized to administer CAR T-cell therapy.
Conclusions: These data suggest that many HCPs are challenged to optimally manage toxicities related to CAR T-cell therapy and are not managing their patients in concordance with expert recommendations. Use of an online tool providing easy access to evidence-based consensus expert recommendations may improve patient care and safety in patients treated with CAR T-cell therapy. A detailed analysis of the tool, including case entries and planned management vs expert consensus recommendations for each toxicity and grade, will be presented.
Figure. Planned Management of HCPs Compared With Expert Recommendations
Frigault:Novartis: Patents & Royalties: Royalty; Arcellx, Celgene, Foundation Medicine, Kite/Gilead, Nkarta, Novartis, and Xenetic: Consultancy. DeAngelo:GlycoMimetics: Research Funding; Abbvie: Research Funding; Blue Print Medicines: Consultancy, Research Funding; Novartis: Consultancy, Patents & Royalties: Royalty, Research Funding; Celgene: Consultancy; Amgen: Consultancy; Shire: Consultancy; Jazz Pharmaceuticals Inc: Consultancy; Incyte: Consultancy; Pfizer: Consultancy; Takeda Pharmaceuticals: Consultancy. Galinsky:Pfizer Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Merus Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; ABIM: Other: Member on specialty oncology board; AbbVie Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. Paul:Agios: Consultancy; Pfizer: Consultancy. Park:Allogene: Consultancy; Amgen: Consultancy; AstraZeneca: Consultancy; Autolus: Consultancy; GSK: Consultancy; Incyte: Consultancy; Kite Pharma: Consultancy; Novartis: Consultancy; Takeda: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal