
Background: The BCL-2 family of anti-apoptotic proteins (e.g., BCL-2, BCL-XL, MCL-1) are important regulators of lymphocyte development and are therapeutic targets for hematologic malignancies, including multiple myeloma (MM). Venetoclax (Ven) is a highly selective, potent, oral BCL-2 inhibitor that induces apoptosis in MM cells and has shown synergistic activity with bortezomib (Velcade; V) and dexamethasone (Dex or d). Combination of the CD38 monoclonal antibody (mAb) daratumumab (D) with Ven is hypothesized to further increase anti-myeloma activity based upon dual mechanisms of pro-apoptotic effects on tumor cells as well as enhanced immune stimulation. Pre-clinically and in healthy human subjects, Ven treatment leads to enrichment of CD8+ T effector memory cells, while resulting in loss of CD4+ and CD8+ naïve T-cells, however the potential immunomodulatory effect of Ven in MM is unknown. Results presented herein describe the pharmacodynamic changes observed in immune cell subsets in relapsed/refractory (R/R) MM patients (pts) treated with VenDex, VenDd, and VenDVd.
Methods: Peripheral blood samples were collected at day 1 of cycles 1-5 to characterize pharmacodynamic changes in B- and T-cell sub-populations by multicolor flow cytometry in Ven MM clinical trials M13-367 (NCT01794520) and M15-654 (NCT03314181). M13-367 is a phase 1/2 study of VenDex in t(11;14) R/R MM, and M15-654 is a phase 1/2 study of VenDd in t(11;14) R/R MM (Part 1) and VenDVd in R/R MM (Part 2). As of 12 June 2019, 15 out of 31 pts treated with VenDex in phase 2 of M13-367, 19 out of 24 pts treated in Part 1 (VenDd), and 19 out of 24 pts treated in Part 2 (VenDVd) of M15-654 had baseline and post-treatment specimens available for analysis.
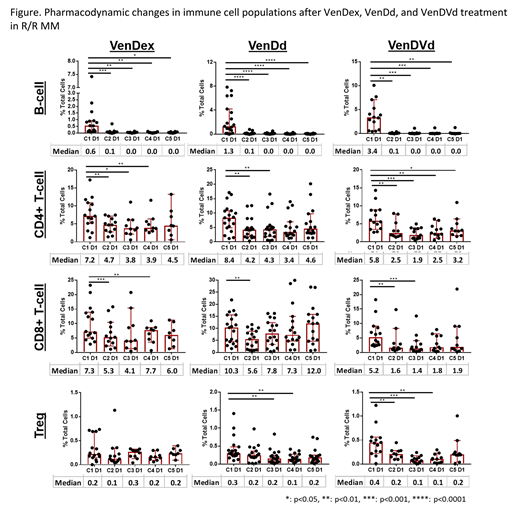
Results: Consistent with previous findings that B-cells are highly dependent upon BCL-2 for cell survival, each Ven-containing regimen (VenDex, VenDd, and VenDVd) resulted in rapid and sustained reduction (~90% decrease from baseline by end of cycle 1) in peripheral B-cell (CD19+/CD5-) counts (Figure). In subgroup analyses, naïve B-cells (CD27-/IgM+) were significantly reduced in pts treated with each regimen, however regulatory B-cells (CD27+/CD24+) remained largely unaffected. In addition, a decrease in plasmablasts (CD27+CD38+CD20-) were only observed in pts treated with D-containing regimens (VenDd and VenDVd), which is consistent with expression of CD38 in this subgroup.
In contrast to B-cells, CD3+ T-cells were minimally reduced (~20-30% reduction from baseline through end of cycle 4) with VenDex and VenDd treatment. A more prominent reduction was observed in pts treated with VenDVd (~50-60% reduction from baseline through end of cycle 4). CD4+ T-cells were more sensitive than CD8+ T-cells to VenDex and VenDd, while both CD4+ and CD8+ T-cells were reduced in VenDVd treated pts (Figure). T-cell subgroup analyses were also performed, including naïve T-cells (CD45RA+/CD197+), effector (CD45RA+/CD197-), effector memory (CD45RA-/CD197-), and central memory (CD45RA-/CD197+). Overall, there were no significant changes in the composition of the T-cell pool (Tnaive, TCM, TEM, TEMRA) or in Th1 (CD4+/CD183+/CD196-), Th2 (CD4+/CD183-/CD196-), and Th17 (CD4+/CD183-/CD196+) cells in pts treated with VenDex, VenDd, or VenDVd. Finally, a decrease in Tregs (CD4+/CD25+/CD127low/CD194+) was observed in VenDd and VenDVd treated pts, but not in VenDex treated pts (Figure). While no reduction in overall Tregs was observed with VenDex, a decrease in the relative proportion of activated Tregs (HLA-DR+/CD45RO+) to naïve (HLA-DR-/CD45RO-) and memory (HLA-DR-/CD45RO+) Tregs was observed.
Conclusions: Comprehensive immunophenotyping studies were conducted to characterize the potential effects of Ven in B- and T-cell subsets when used in combination with standard of care anti-myeloma agents of various mechanisms of action (i.e. glucocorticoid, CD38 mAb and/or proteasome inhibitor). While each Ven combination regimen resulted in significant reductions in circulating B-cells, T-cell effects were largely dependent upon the combination regimen. Minimal reduction in total T-cell counts were observed with VenDex and VenDd, which were primarily due to reduction in CD4+ T-cells. However, the extent of T-cell depletion (CD4+ and CD8+ T-cells) was greatest in pts treated with the bortezomib-containing regimen VenDVd. Correlative studies between immune profiles and pt outcomes are ongoing.
Vishwamitra:AbbVie: Employment, Other: stock or other options. Mantis:AbbVie Inc: Employment, Other: Stock/stock options. Haribhai:AbbVie: Employment, Other: stock or other options. Uziel:AbbVie: Employment, Other: stock or other options. Leverson:AbbVie Inc: Employment, Other: Stock or options. Pauff:AbbVie Inc: Employment, Other: may own stock or stock options. Bueno:AbbVie: Employment, Other: Stock/stock options. Maciag:AbbVie: Employment, Other: Stock/stock options. Ross:AbbVie: Employment, Other: Stock/stock options.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal