Background:
Direct oral anticoagulants (DOACS) were recently approved for anticoagulation in patients with atrial fibrillation for prevention of stroke and patient with venous thromboembolism (VTE) for the prevention of recurrent VTE. They have shown comparable efficacy and safety compared to vitamin K antagonist (VKA). DOACS have uniform pharmacokinetics and pharmacodynamics; hence, regular monitoring is not required. The morbidly obese patients may have increased volume of distribution and altered clearance which may alter the pharmacokinetics and consequently efficacy and safety of DOACS in these patients. 2016 International Society of Thrombosis and Hemostasis guideline recommends avoiding DOACS in a patient with body mass index (BMI) >40 kg/m2 or with body weight >120 kg due to lack of clinical trials. There is no sizeable randomized control trial to study the outcome of DOACS in morbidly obese patients, but subgroup analysis of a major randomized control trials and observational studies may provide the data on the efficacy and safety of DOACS. We intend to utilize this data in this meta-analysis to study the effectiveness and safety of DOACS in morbidly obese patients.
Methods:
Eligibility Criteria-
Randomized control trial, observational studies including patients above 18 years with BMI>40kg/m2, body weight>120 kg and history of atrial fibrillation or VTE who are taking DOACS or warfarin for therapeutic purpose were included in the analysis.
Study Selection-
We searched Medline and Cochrane database. 52 studies were identified and 43 remained after removing duplicates. After assessing full text for eligibility 5 studies were included in the meta-analysis.
Statistical analysis-
Statistical analysis was performed with Review manager 5.3 by the Cochrane Collaboration. Forest plot was used to analyze publication bias. Heterogeneity in the studies were analyzed with Cochran Q analysis and I2 statistics.
Results:
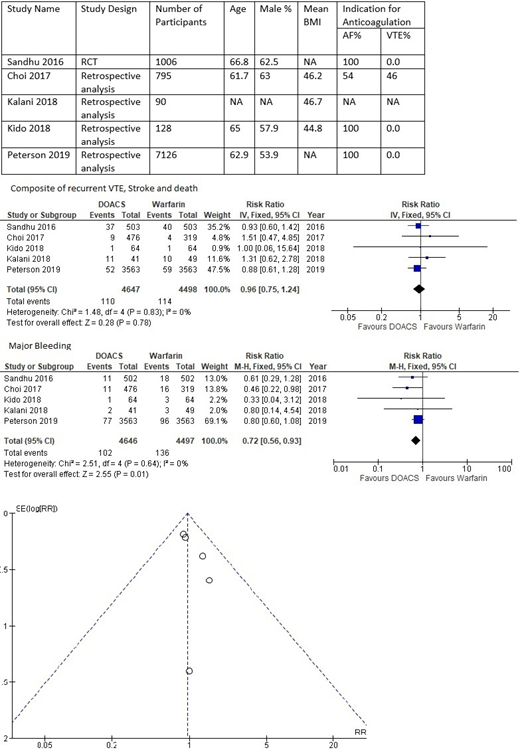
Population characteristics were available in four studies. The mean age of the population ranged from 61.7 to 66.8 years, and mean BMI ranged from 44.8 to 46.7 kg/m2 . I2 analysis showed that the studies included were homogeneous, so fixed-effect model was applied. The incidence of recurrent VTE, stroke, or death ranged from 1.5% to 7.3% in DOACS compared to 1.2% to 7.9% in warfarin except in one study by Kalani et al. where it was 26% in DOACS vs. 20% in warfarin. Relative risk was 0.96 (confidence interval 0.75 - 1.28, p 0.78) indicating there was no significant difference in the primary efficacy outcome between the two groups. The incidence of major bleeding ranged from 1.5% to 4.8% in DOACS compared to 2.6% to 6.1% in warfarin. Relative risk was 0.72 (confidence interval 0.56 - 0.93, p 0.01) indicating that the risk of major bleeding was low among patient using DOACS.
Conclusion:
Efficacy of DOACS is similar compared to VKA when used for prevention of recurrence in VTE or prevention of stroke in atrial fibrillation in the patients with BMI>40 kg/m2 or bodyweight >120 kg. There was a trend towards a lower risk of bleeding in the patients with DOACS which is similar to the risk of major bleeding reported in non-obese patients in landmark randomized clinical trials. Only two studies included the patients with VTE, and additional studies are required to assess the safety and efficacy of DOACS among morbidly obese individuals with VTE.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal