Background: Immune thrombotic thrombocytopenic purpura (iTTP) is a potentially fatal hematologic disorder characterized by thrombocytopenia, microangiopathic hemolytic anemia, and ischemic organ impairment. The incidence of iTTP is higher among African-Americans (AA), however, differences in presentation and outcomes have not been fully investigated. In a multi-center cohort of patients with iTTP from the United States Thrombotic Microangiopathy (USTMA) Consortium, we tested the hypothesis that AA race is an independent predictor of poor outcomes including iTTP related mortality and relapse.
Methods: We queried data from the USTMA iTTP registry, which currently includes data from 785 individual patients from 15 institutions across the United States. Data from at least one iTTP episode are available for 734 patients. The cohort is 35.1% (N = 272) White, 58.7% (N = 455) African American, 0.4% (N=3) Asian, 1.8% (N=14) Hispanic, and 4.0 % (N=31) other/unknown race. We restricted our analyses to AA and White participants because of small numbers in the other groups. We compared presenting features and treatments using the chi-squared test and t-test for categorical and continuous variables, respectively. A relapse was defined as a recurrent iTTP episode occurring at least 30 days after last therapeutic plasma exchange. To evaluate relapse-free survival, we included only patients enrolled in the registry at their first TTP episode (144 White and 246 AA) since patients presenting with a relapse as their index episode are already confirmed to have relapsing iTTP. Kaplan Meier analysis was used to compare relapse-free survival in White and AA patients, and a Cox regression model was developed to evaluate the independent effect of race on relapse, adjusting for potential confounders including age, sex, and the use of rituximab.
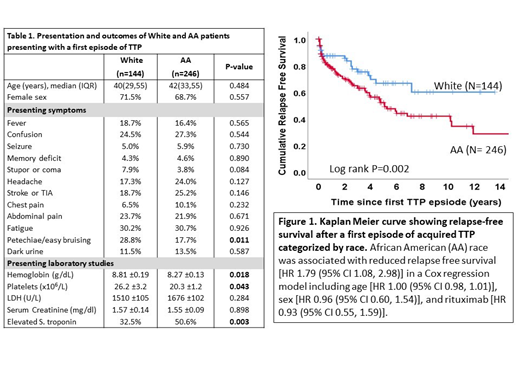
Results: Demographics and presenting features of 390 individuals (144 White and 246 AA) presenting with a first episode of iTTP are shown in Table 1. Presenting symptoms including fever, confusion, seizure, memory deficits, stupor, headache, stroke, chest pain, abdominal pain, fatigue, and dark urine were similar between Whites and AA except for petechiae, which were more frequently documented in Whites (28.8% vs 17.7%, p=0.011). Presenting laboratory studies were also comparable though AA had a higher rate of elevated serum troponin (50.6% vs 32.5%, p=0.003), lower hemoglobin level (8.27 ± 0.13 vs 8.81 ± 0.19, p=0.0176) and platelet count (20.3 ± 1.2 vs 26.2 ± 3.2, p=0.0432). In addition to therapeutic plasma exchange and corticosteroids, rituximab was administered to 23.7% of White patients and 22.7% of AA during their first iTTP episode (P=0.815). Median time to platelet count recovery (days of daily plasma exchange until normal platelet count for two consecutive days) was shorter in AA compared with White patients [5 (IQR 4, 10) vs. 8 (IQR 5, 14), log rank P = 0.004]. AA race remained a significant predictor of the shorter time to platelet count recovery [HR 1.44 (95% CI 1.12, 1.85), P=0.004] after adjusting for rituximab therapy [HR 0.60 (95% CI 0.0.46, 0.80), P<0.001], female sex [HR 0.95 (95% CI 0.73, 1.22), P=0.669], age [HR 0.99 (95% CI 0.99, 1.01), P=0.682], platelet count [HR 1.00 (95% CI 0.99, 1.04), P=0.820] and LDH at presentation [HR 1.00 (95% CI 1.00, 1.00), P=0.525]. Death during the first episode occurred in 8.9% of White patients and 5.5% of AA patients (P=0.206).
Relapse-free survival after the first episode of iTTP was lower in AA than White patients (Figure 1). AA race was associated with the reduced relapse free survival [HR 1.79 (95% CI 1.08, 2.98), P=0.024] in a Cox regression model adjusted for age [HR 1.00 (95% CI 0.98, 1.01), P=0.683], sex [HR 0.96 (95% CI 0.60, 1.54), P=0.867], and rituximab therapy [HR 0.93 (95% CI 0.55, 1.59), P=0.806].
Conclusion: African Americans with iTTP have a higher relapse rate and shorter relapse free survival after the first episode of the disease compared with Caucasian patients, which is independent of age, sex and rituximab therapy. Contrary to our hypothesis, acute outcomes of iTTP (time to platelet count recovery and mortality) were not worse in AA patients. The factors contributing to the higher relapse rate in AA with iTTP need to be further investigated. Our findings suggest that AA patients may also benefit from closer follow up.
Farland:Sanofi: Membership on an entity's Board of Directors or advisory committees. Metjian:Sanofi: Membership on an entity's Board of Directors or advisory committees. Raval:Bayer, Inc: Research Funding; Sanofi: Membership on an entity's Board of Directors or advisory committees. Liles:Shire: Other: PI on clinical trial Sickle cell ; Imara: Other: PI on Clinical trial- Sickle cell ; Novartis: Other: PI on clinical trial Sickle cell . Baumann Kreuziger:CSL Behring: Consultancy; Vaccine Injury Compensation Program: Consultancy. McCrae:Dova Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Pfizer Pharmaceutical: Membership on an entity's Board of Directors or advisory committees; Rigel Pharmaceutical: Membership on an entity's Board of Directors or advisory committees; Sanofi Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. Zheng:Alexion: Speakers Bureau; Ablynx/Sanofi: Consultancy, Speakers Bureau; Clotsolution: Other: Co-Founder; Shire/Takeda: Research Funding. Cataland:Alexion: Consultancy, Research Funding; Ablynx/Sanofi: Consultancy, Research Funding. Chaturvedi:Shire/Takeda: Research Funding; Sanofi: Consultancy; Alexion: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal