Abstract
Background: R-maintenance for 2 years is part of a standard treatment approach for previously untreated follicular lymphoma. In this study we compared the efficacy and safety of 4 versus 2 years of R-maintenance following first-line treatment with B-R.
Methods: Patients included in the study were required to have stage II (bulky disease >7 cm), III, or IV disease. Patients were treated with up to 6 cycles of B-R plus 2 additional cycles of Rituximab. All responding patients received 2 years R-maintenance (375 mg/m2 every 2 months). Patients who tolerated treatment for the entire 2 years and who were still in remission hereafter were subsequently randomized to 2 more years of R-maintenance or observation. The primary endpoint was progression-free survival (PFS). Secondary endpoints included response rates, overall survival (OS), and toxicity.
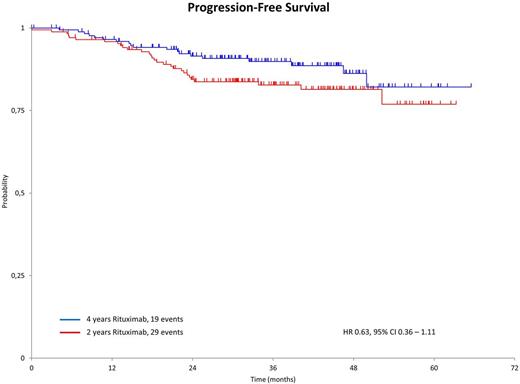
Results: A total of 612 patients with follicular lymphoma were enrolled in the study. Of enrolled patients, 555 were evaluable for response and 497 (90%) responded to B-R induction, with 181 patients (33%) achieving a complete remission. A total of 261 patients discontinued treatment during induction or the first 2 years of R-maintenance. The most frequent reasons for discontinuation included progression in 85 of 261 pts (33%), patient's or doctor's choice in 55 pts (21%), toxicity or infections in 29 pts (11%), Rituximab intolerance in 14 pts (5%), or death in 11 pts (4%) with a median age of 70 years and 5 pts who died from infections. Of 351 patients randomized to 2-years or 4-years of R-maintenance, 350 were evaluable (n = 172 and n = 178, respectively). The median age was 60 years and patient characteristics were comparable between groups. The median observation time was 36 months following randomization. Median PFS and OS have not yet been reached in either arm. PFS appears superior with 4 years versus 2 years of R-maintenance with a hazard ratio (HR) of 0.63 (95% CI 0.36-1.11; fig. 1). There was no difference in OS between groups. One patient died from a progressive multifocal leukoencephalopathy (PML, randomized to 4 years of Rituximab). A historical comparison for PFS between responding patients given 2 years of R-maintenance in this MAINTAIN trial and subjects from the former StiL NHL1 study (B-R versus CHOP-R) who received B-R only appears to favor R-maintenance with a HR of 0.78 (95% CI 0.54-1.04). Data analysis is ongoing and updated results will be presented at the ASH meeting.
Conclusions: Results at the time of this analysis appear to favor 4-years over 2-years of R-maintenance and 2-years over observation compared to a historical control. Further analysis including the updated dataset will be presented at the ASH meeting and should provide more definitive evidence regarding the benefit of prolonged R-maintenance.
Rummel: Roche: Honoraria, Other: PrefMab is sponsored by F. Hoffmann-La Roche Ltd. Third-party Medical Writing assistance, under the direction of Mathias Rummel, was provided by Lynda McEvoy of Gardiner-Caldwell Communications, and was funded by F. Hoffmann-La Roche Ltd, Research Funding. Buske: Hexal: Honoraria; Celltrion, Inc.: Consultancy, Honoraria; Pfizer: Honoraria; Gilead: Honoraria; Janssen: Honoraria, Research Funding; Roche: Honoraria, Research Funding. Balser: Sanofi-Aventis: Other: Travel support; IOMEDICO: Other: Travel support. Behringer: Roche: Consultancy, Honoraria; BMS: Consultancy, Honoraria. Dürig: Lead Discovery Center: Research Funding. Maschmeyer: Gilead: Honoraria; Pfizer: Honoraria, Travel support; Merck-Serono: Honoraria; Celgene: Honoraria; Bristol-Meyers Squibb: Honoraria; Basilea: Honoraria; Janssen-Cilag: Honoraria; Astellas: Honoraria; AstraZeneca: Honoraria; Boehringer-Ingelheim: Honoraria; Amgen: Honoraria; Bristol-Meyers Squibb: Other: Travel support. Schliesser: Bayer: Equity Ownership; Roche: Equity Ownership. Burchardt: Celgene: Honoraria; Roche: Honoraria; CSL: Honoraria; Medac: Honoraria. Barth: Celegene: Honoraria; CSL: Honoraria, Other: Travel support; BMS: Honoraria; Hexal: Honoraria; Johnson & Johnson: Honoraria; Medac: Honoraria; Mundipharma: Honoraria; Novartis: Honoraria; Roche: Honoraria; Takeda: Honoraria; Gilead: Other: Travel support; Amgen: Other: Travel support. Greil: Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel support, Research Funding; Novartis, Celgene: Research Funding; Takeda: Honoraria, Research Funding; BMS, Amgen: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal