Abstract
Introduction:
Both HIV+ Hodgkin and non-Hodgkin lymphomas have higher rates of latent infection by the gamma-herpesviruses (GHVs), Epstein-Barr virus (EBV) and Kaposi sarcoma-associated herpes virus (KSHV), than corresponding lymphomas in the HIV-seronegative population. Bortezomib, a proteasome inhibitor, induces lytic activation of both EBV and KSHV. Lytic activation of GHVs latently infecting lymphoma cells is hypothesized to beneficial both for direct tumor cell lysis as well as increased cytotoxic immune response due to viral lytic gene products. Furthermore, preclinical studies found proteasome inhibition impairs HIV infectivity via preservation of human anti-retroviral APOBEC3G, suggesting a novel therapeutic strategy to control HIV. Given that therapy of relapsed or refractory HIV-associated lymphoma (R/R-HAL) results in modest rates of remission, we sought to capitalize on the high viral association within HAL using an oncolytic strategy with bortezomib.
Objectives:
The primary objective of this study was to evaluate safety and overall response rate (ORR) of R/R-HAL to bortezomib combined with ifosfamide, carboplatin, etoposide +/- rituximab (ICE/R).
The secondary objectives of this study were to estimate the impact of bortezomib on lytic activation of EBV and KSHV, using peripheral blood mononuclear cell (PBMC) viral loads, and on HIV using single copy plasma viral loads; to report overall survival at 1 year (1yr-OS); and to correlate EBV and KSHV viral load changes with lymphoma response.
Methods:
A 3+3 dose escalation design with a 7-day lead-in period of bortezomib alone prior to bortezomib + ICE/R allowed for assessment of early effects of bortezomib on viral loads. Bortezomib was given intravenously on day 1 and 8 of each cycle at one of 4 dose levels: 0.7, 1, 1.3 or 1.5 mg/m2. Standard dose ICE +/- R began day 8 of cycle 1 (28-day cycle); ICE +/- R began day 1 of all subsequent cycles (21-day cycle). Rituximab was included in the regimen only for CD20+ lymphoma.
Binomial proportions were used to estimate ORR. The product-limit (Kaplan-Meier) method was used to estimate 1yr-OS. The Wilcoxon signed rank test was used to evaluate changes in viral loads.
Results:
Twenty-three subjects were enrolled from 7 sites within the AIDS Malignancy Consortium (AMC). More than 90% of enrolled subjects were men and half were minorities; at baseline, 20/23 were on antiretroviral therapy, median CD4 count was
315/µL and median HIV viral load was undetectable. Mean age was 50 years. Over half of subjects had stage IV HAL; the majority had diffuse large B-cell lymphoma (DLBCL) (n=15), 2 had primary effusion (PEL), 3 had plasmablastic and 2 had Hodgkin lymphoma.
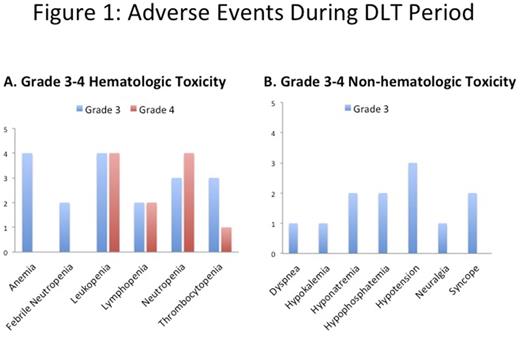
Figure 1 summarizes grade 3-4 toxicities of the 22 subjects evaluable for adverse events during the dose-limiting toxicity period (cycles 1+2). The maximum tolerated dose was not reached at the highest dose cohort studied (bortezomib 1.5mg/m2).
Responses occurred in 14/22 subjects initiating protocol therapy: 5 complete and 9 partial responses (PR). Of the 20 subjects who completed 2 or more cycles, the ORR was 80%. Nine of the responders underwent auto-hematopoietic stem cell transplant after protocol therapy. 1yr-OS was 55%.
After bortezomib alone, median values of EBV PBMC viral load measured on day 8 were 2x greater than baseline. However, paired analysis did not confirm significant change in this small sample (n=16 evaluable), and there was no correlation found between change in EBV viral load and response. The 2 subjects with known KSHV+ lymphoma (PEL) each had more than a 1-log increase in day 8 KSHV viral load compared with baseline. Both of these subjects attained a PR from protocol therapy.
Conclusions:
Addition of bortezomib to ICE/R in R/R HAL is feasible with ORR (80%) and 1yr-OS (55%) comparing very favorably with a prior AMC retrospective report of ICE/R in R/R HAL (n=31, ORR 32%, 1yr-OS 38%, Bayraktar 2012). Evaluation of data collected from individual subjects suggests GHV lytic activation may occur with bortezomib alone. However, the lower bortezomib dose levels used to treat the bulk of study subjects and the limited study sample size limit our power to confirm this conclusion. We plan to further explore the effects of proteasome inhibition on GHVs. Evaluation of the impact of bortezomib on HIV replication is pending and will be presented at the meeting.
Reid:Millennium: Research Funding. Sparano:Takeda: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal