Abstract
Purpose
To investigate safety and efficacy of high-dose chemotherapy followed by autologous stem cell transplantation (HCT-ASCT) in patients with relapsed or refractory primary CNS lymphoma (PCNSL).
Patients and methods
We conducted a single-arm multicentre phase 2 study for immunocompetent patients (<66 years of age) with PCNSL failing prior HD-MTX based chemotherapy. Induction treatment consisted of 2 courses of rituximab (rituximab 375mg/m2), high-dose cytarabine (2 x 3g/m2) and thiotepa (40mg/m2) with collection of autologous stem cells in between. Conditioning treatment for HCT-ASCT consisted of rituximab 375mg/m2, carmustine 400mg/m2 and thiotepa (4 x 5mg/kg). Patients commenced HCT-ASCT irrespective of response status after induction. Only patients not achieving complete remission (CR) after HCT-ASCT received whole brain radiotherapy (WBRT). The primary endpoint was CR after HCT-ASCT by intention-to-treat (ITT). Secondary endpoints included safety, progression free survival (PFS, time to progression or death) and overall survival (OS, time to death due to any cause).
Results
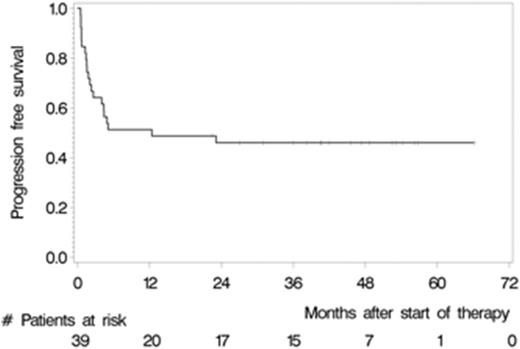
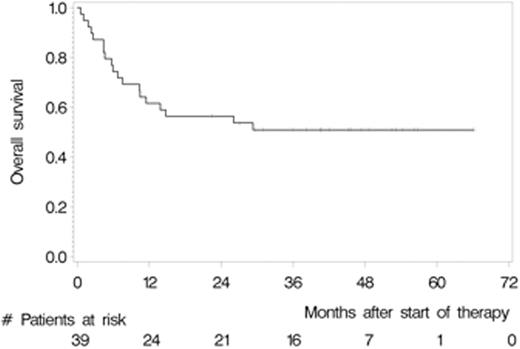
Between May 2007 and July 2012, we enrolled 39 patients from 12 German centres. The median age and Karnofsky performance score was 57 years (range 37 to 65) and 90% (range 60% to 100%), respectively. 28 (71.8%) patients had relapsed and 8 (28.2%) refractory disease. 22 (56.4%) patients responded to induction (4 CR, 18 partial remissions [PR]) and 32 (82.1%) patients commenced HCT-ASCT. 22 patients (56.4%, 95% CI 39.6% to 72.2%) achieved CR after HCT-ASCT, 6 (15.4%) achieved PR, and 1 (2.6%) had stable disease. In 9 (17.8%) patients the final scan was not done, because 7 (18.0%) did not undergo HCT-ASCT and 2 died (5.1%) during HCT-ASCT procedure. After a median follow-up of 45.2 months, the respective 2-year PFS and OS rates were 46.0% (95% CI 30.3% to 61.7%, median PFS 12.4 months, Figure 1) and 56.4% (95% CI 40.8% to 72.0%); median OS not reached (Figure 2). The non-relapse mortality rate was 10.3% (95% CI 4.1% to 26.0%) at 1 year without any further increase afterwards. In the subset of 32 patients who received HCT-ASCT, 14 (56.3%) experienced progression or died translating into 1 and 2-year PFS rates (calculated from date of HCT-ASCT) of 62.5% (95% CI 45.7% to 79.3%) and 56.1% (95% CI 38.8% to 73.3%) with no further decrease afterwards. Main grade 3 or higher toxicities were hematological as expected. We recorded four (10.3%) treatment-related deaths, 2 during induction and 2 during HCT-ASCT.
Conclusions
In eligible PCNSL patients failing HD-MTX based chemotherapy, a short induction with high-dose cytarabine and thiotepa followed by HCT-ASCT is an effective treatment option. Our data provide a reliable benchmark for future comparative studies needed to further scrutinize the role of HCT-ASCT in the salvage setting for PCNSL.
Kasenda:Riemser: Other: Travel Support. Schmidt-Wolf:Janssen: Research Funding; Novartis: Research Funding. Röth:Alexion Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Honoraria. Stilgenbauer:Amgen: Consultancy, Honoraria, Other: Travel grants, Research Funding; Genentech: Consultancy, Honoraria, Other: Travel grants , Research Funding; Boehringer Ingelheim: Consultancy, Honoraria, Other: Travel grants , Research Funding; Janssen: Consultancy, Honoraria, Other: Travel grants , Research Funding; Hoffmann-La Roche: Consultancy, Honoraria, Other: Travel grants , Research Funding; Pharmacyclics: Consultancy, Honoraria, Other: Travel grants , Research Funding; Novartis: Consultancy, Honoraria, Other: Travel grants , Research Funding; Sanofi: Consultancy, Honoraria, Other: Travel grants , Research Funding; Gilead: Consultancy, Honoraria, Other: Travel grants , Research Funding; AbbVie: Consultancy, Honoraria, Other: Travel grants, Research Funding; Mundipharma: Consultancy, Honoraria, Other: Travel grants , Research Funding; Genzyme: Consultancy, Honoraria, Other: Travel grants , Research Funding; GSK: Consultancy, Honoraria, Other: Travel grants , Research Funding; Celgene: Consultancy, Honoraria, Other: Travel grants , Research Funding. Illerhaus:Riemser: Honoraria; Amgen: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal