Abstract
Introduction
Tyrosine kinase inhibitor (TKi) has become the standard of care in patients (pts) with chronic myeloid leukemia (CML) and an unavoidable tool in the combined therapy for pts with Philadelphia chromosome positive (Ph+) acute lymphoblastic leukemia (ALL).
Nevertheless, because of resistance to TKI and side-effects, allogeneic stem cell transplantation (HSCT) remains the standard therapy of ALL Ph+ and of CML pts failing 1st line therapy with TKi, with failure or insufficient response or intolerance or mutations resistant to 2nd generation TKI, or in the advanced phase at diagnosis (accelerated phase and blast crisis).
Unfortunately, despite greater remission with the use of TKi pre-transplant, HSCT transplant outcome have not improved largely due to high incidence of relapse after transplant.
In the past decade several multi-institutional studies confirmed the feasibility and safety of post-HSCT imatinib administration as prophylactic or therapeutic strategy. Second and 3rd generation TKi administration after HSCT - targeting mutational status and according to pre-HSCT activity - is today under investigation.
Methods
Here we are reporting our experience in post-HSCT treatment with the 3rd generation TKi ponatinib in 5 pts (4 CML, 1 ALL Ph+) treated between 2011 and 2016 at our Institution.
Pts data and information were collected from Institutional database and chapters revision. A written consent was given by pts allowing the use of medical records for research in accordance with the Declaration of Helsinki.
Results
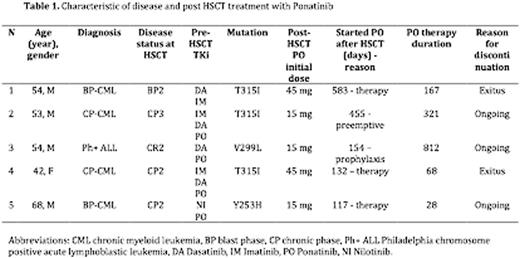
Pts and diseases features are reported in table 1. Stem cell source was peripheral blood in all cases, 3 pts were transplanted from a family mismatched donor (haplo), 1 from a family matched donor, 1 from a matched unrelated donor (MUD). The 3 haplo-transplanted pts previously underwent a MUD HSCT. All pts received a treosulfan based conditioning regimen and GvHD prophylaxis consisted on co-administration of MMF and rapamycin.
Pre-transplant treatment for the ALL Ph+ consisted of chemotherapy combined with dasatinib, followed by a first MUD HSCT and dasatinib in maintenance. The patient relapsed 1 year after HSCT with documentation of mutation V299L. Ponatinib was introduced as salvage treatment to bridge second haplo HSCT.
Pre-transplant treatment for the CML patients consisted of TKi therapy with combination of chemotherapy in case of uncontrolled progression of disease. Two pts received a first MUD HSCT but relapsed respectively 5 months and 4 years later.
Four pts received ponatinib 45 mg daily before the last HSCT: one patient achieved sustained major molecular response, 3 pts obtained transient response. All pts were presenting 2nd generation TKi resistant mutation (ref table 1).
Ponatinib was started at a median time of 157 days after HSCT (range, 117-583): in 3 cases as salvage treatment in overt relapse, while in one case as prophylaxis and one case as preemptive therapy.
Acute GvHD was diagnosed in 4 pts before ponatinib administration, 2 of them also experienced chronic GvHD. No new cases of GvHD were observed after initiation of ponatinib.
Immunosuppressive treatment and azoles treatment were discontinued before ponatinib in all but one patient who was under combined treatment for chronic GvHD: therapeutic drug monitoring was closely performed without evidence drug-drug interaction.
Pts were regularly evaluated for toxicities. No serious adverse events were reported in our experience: we administered ponatinib at a median maximum dosage of 30 mg daily (range, 15-45 mg), for a median of 24 weeks (range, 4 - 116 weeks). Two pts required anti hypertension drugs. One patient was closely monitored for multifactorial liver cholestasis never requiring ponatinib discontinuation.
At last evaluation one patient maintained the status of molecularly undetectable leukemia (follow-up post HSCT 30 months) and two pts obtained molecular response (follow-up post HSCT 25 months and 5 months). Two patients who received therapeutic ponatinib in overt relapse didn't respond and died for progressive disease.
Conclusions
Ponatinib is safe and well tolerated as bridge to HSCT and to maintain the disease control after transplant. Prophylaxis targeted therapy and pre-emptive therapy with ponatinib may lead the reduction of disease relapse for high-risk Ph+ leukemia.
Bonini:Molmed SpA: Consultancy; TxCell: Membership on an entity's Board of Directors or advisory committees. Ciceri:MolMed SpA: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal