Abstract
Background: Philadelphia chromosome-positive (Ph+) CML is a myeloproliferative disease characterized by the presence of the abnormal Ph+ in hematopoietic cells. Imatinib, dasatinib and nilotinib are BCR-ABL TKIs commonly used in the treatment of CML-CP. Many patients on BCR-ABL TKI therapy will experience adverse events (AEs). Some of the more common AEs associated with first- and second-generation BCR-ABL TKIs include fluid retention, diarrhea, rash, musculoskeletal pain, nausea, vomiting, muscle cramps, and headache. Some patients will be unable to tolerate these AEs and will discontinue therapy. The current study aimed to assess the efficacy and safety of nilotinib in patients with CML-CP who are responsive to but intolerant of treatment with imatinib or dasatinib. Although the study was stopped early due to low recruitment, here we present results on cross-intolerance and molecular response in the patients who were switched from imatinib or dasatinib to nilotinib.
Methods: Eligible adult patients had: Ph+ CML-CP associated with BCR-ABL quantifiable by real-time quantitative reverse transcriptase-polymerase chain reaction (RQ-PCR); received ≥3 months imatinib or dasatinib or both; were <1% (IS) BCR-ABL level in the blood during imatinib or dasatinib treatment; and were experiencing any non-hematological AEs (any grade) that persisted for ≥1 month or recurred at least once despite supportive care. After a washout period of ≥3 days, patients were switched to nilotinib 300 mg BD and treated for up to 24 months. The dose could be reduced to 450 mg QD for safety reasons. Treatment interruptions, dose reductions and dose re-escalation to 300 mg BD were allowed for management of AEs. The primary outcome was achievement of MR4.5 (BCR-ABL ≤0.0032%) by 24 months. Major molecular response (MMR; BCR-ABL ≤0.1%) and MR4.0 (BCR-ABL ≤0.01%) were also assessed in an exploratory capacity at each visit (month 1, 2, 3, then every 3 months to month 24). Secondary endpoints included the kinetics of molecular response. Preliminary results are summarized descriptively. Planned enrolment was 130 patients.
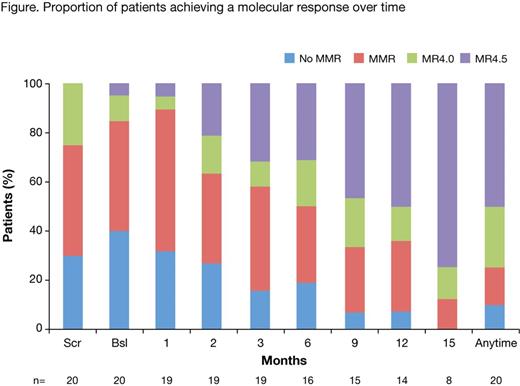
Results: The study was stopped early due to low recruitment; 20 patients were enrolled (mean age 53.9 years [range 31-77]; 14 female). 16 patients had received prior imatinib therapy, 4 patients prior dasatinib. Median nilotinib treatment duration was 494 days (mean 480, SD 167.6 days). At screening, 30% of patients were not in MMR, 45% had MMR and 25% had MR4.0. By month 3 and 24 of nilotinib treatment, 55% (11/20) and 65% (13/20) of patients, respectively, achieved at least a 1 log reduction in BCR-ABL levels. 35% (7/20) of patients achieved MR4.5 between baseline and month 3 of nilotinib treatment, and 50% (10/20) achieved MR4.5 at any time up to month 24. The proportion of patients with a molecular response at each visit up to month 15 is shown in the Figure. AEs during prior treatment with imatinib and dasatinib included gastrointestinal events (nausea, vomiting, diarrhea), superficial edema, myalgia, fatigue, rash, and headache, among others. 68% of AEs had resolved by month 3 of nilotinib treatment. Among the 13 evaluable patients on prior imatinib, 7 (54%) had resolution of all AEs during treatment with nilotinib; of 3 evaluable patients on prior dasatinib, 3 (100%) had AE resolution on nilotinib. Grade 3/4 AEs during nilotinib therapy occurred in 3 patients: diabetes mellitus, fatigue, neutropenia, pneumonia, osteoarthritis, and hyperuricemia.
Conclusions: Although early termination of the study has not allowed for a robust analysis, these results suggest that nilotinib is effective and well tolerated in most patients intolerant of imatinib or dasatinib. During the first 3 months of switching to nilotinib, 55% of patients had achieved at least a 1 log reduction in BCR-ABL levels, and 35% of patients had achieved MR4.5. The cumulative rate of MR4.5 by 24 months was 50%. Achievement of this endpoint has been linked to favorable long-term outcomes, such as treatment-free remission. Furthermore, the majority of the AEs had resolved by month 3 of nilotinib therapy. This improved tolerance to nilotinib may result in improved treatment adherence. As such, although further study in a larger population is needed for confirmation, these results provide further evidence that nilotinib is a favorable option to establish a molecular response in patients intolerant of imatinib or dasatinib.
D'Rozario:BMS: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Roche: Consultancy, Honoraria. Branford:Qiagen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Research Funding; Ariad: Research Funding; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Cepheid: Consultancy. Yeung:Ariad: Research Funding; Novartis Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Anderson:Novartis Pharmaceuticals: Employment. Gervasio:Novartis Pharmaceuticals: Employment. Hughes:Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Ariad: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal