Abstract
Introduction: Daratumumab is an anti-CD38 IgGκ monoclonal antibody that has been combined successfully with lenalidomide and dexamethasone. The combination of daratumumab with lenalidomide and dexamethasone (DRd) has been compared with lenalidomide and dexamethasone alone (Rd) in patients (pts) with relapsed or refractory multiple myeloma (RRMM) in a randomized phase 3 study (Dimopoulos MA, et al. N Engl J Med 2016; in press). In a pre-specified interim analysis, the DRd combination demonstrated significantly longer progression-free survival (PFS) in addition to deep and durable responses compared with the Rd arm. We performed subgroup analyses to further examine these efficacy data according to prior treatment exposure.
Methods: Pts who received ≥1 prior line of therapy were randomized (1:1) to Rd (lenalidomide: 25 mg PO on Days 1-21 of each 28-day cycle; dexamethasone: 40 mg PO weekly) with or without daratumumab (16 mg/kg IV qw for 8 weeks, q2w for 16 weeks, then q4w until progression). The primary endpoint was PFS. Pts who were refractory to lenalidomide were not eligible. All analyses were performed in pts who received 1 to 3 prior lines of therapy.
Results: Median follow-up was 13.5 months. Pts who were lenalidomide-naive prior to the start of study treatment (DRd, n=226; Rd, n=219) demonstrated significantly longer PFS with DRd vs Rd (median: not reached [NR] vs 18.4 months; HR, 0.36; 95% CI, 0.25-0.52; P<0.0001), with estimated 12-month PFS rates of 83.0% vs 59.9%, respectively. ORR was significantly higher with DRd vs Rd (96% vs 79%), with ≥VGPR rates of 76% vs 47% and ≥CR rates of 44% vs 21%, respectively (P<0.0001 for all). In the lenalidomide-exposed subgroup (DRd, n=46; Rd, n=45), median PFS was NR in both treatment groups (HR, 0.49; 95% CI, 0.22-1.12; P=0.0826); estimated 12-month PFS rates were 84.1% vs 63.1%, respectively. ORR was higher with DRd vs Rd but did not reach statistical significance (87% vs 71%; P=0.0729); however, rates of ≥VGPR (78% vs 38%; P=0.0001) and ≥CR (44% vs 12%; P=0.0011) were significantly improved with DRd vs Rd, respectively.
For bortezomib-naive pts (DRd, n=44; Rd, n=45), PFS was significantly longer with DRd vs Rd (median: NR vs 15.8 months; HR, 0.34; 95% CI, 0.13-0.86; P=0.0170), with estimated 12-month PFS rates of 85.4% vs 69.2%, respectively. ORR was significantly higher with DRd vs Rd (98% vs 82%; P=0.0158), with trends toward increased rates of ≥VGPR (74% vs 55%; P=0.0544) and ≥CR (42% vs 23%; P=0.0576). In the bortezomib-exposed pts (DRd, n=228; Rd, n=219), median PFS was NR in DRd vs 18.4 months in Rd (HR, 0.35; 95% CI, 0.24-0.50 P<0.0001); estimated 12-month PFS rates were 82.8% vs 58.7%, respectively. Significant differences in ORR (93% vs 77%), rate of ≥VGPR (77% vs 43%) and rate of ≥CR (44% vs 19%) were observed with DRd vs Rd, respectively (P<0.0001 for all).
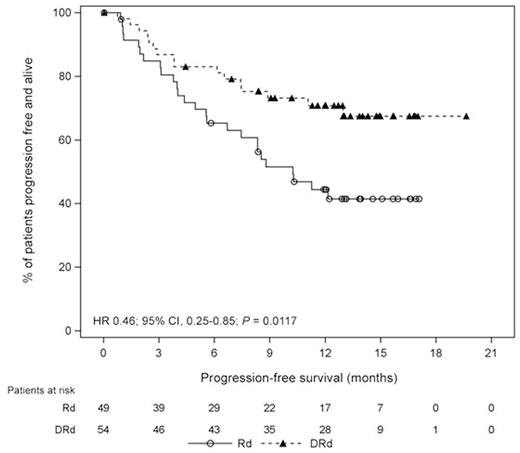
Among bortezomib-refractory patients (DRd, n=54; Rd, n=49), the PFS benefit of DRd compared with Rd was maintained (median: NR vs 10.3 mo, respectively; HR, 0.46; 95% CI, 0.25-0.85; P=0.0117; Figure). The estimated 12-month PFS rates were 70.8% vs 44.4%, respectively. Similar to bortezomib-exposed pts, ORR (92% vs 68%; P=0.0024), rate of ≥VGPR (75% vs 36%; P=0.0001), and rate of ≥CR (46% vs 13%; P=0.0003) were all significantly higher with DRd vs Rd for bortezomib-refractory pts.
Updated data will be presented at the meeting.
Conclusions: Among pts who received 1 to 3 prior lines of therapy, significantly longer PFS and higher ORR were observed with DRd vs Rd among pts who previously received bortezomib or were refractory to bortezomib or were lenalidomide-naive. Higher rates of deeper responses were observed in pts who previously received lenalidomide or bortezomib. Follow-up is ongoing to assess PFS in pts who received 1 to 3 prior lines of therapy and previously received lenalidomide. These results further strengthen the significant benefit of combining daratumumab with Rd for RRMM.
Progression-free Survival in Bortezomib-refractory Patients who Received 1 to 3 Prior Lines of Therapy
Progression-free Survival in Bortezomib-refractory Patients who Received 1 to 3 Prior Lines of Therapy
Moreau:Janssen: Honoraria, Speakers Bureau; Novartis: Honoraria; Takeda: Honoraria; Celgene: Honoraria; Amgen: Honoraria; Bristol-Myers Squibb: Honoraria. Kaufman:Pharmacyclics: Consultancy; Incyte: Consultancy; Novartis: Consultancy, Research Funding; Celgene: Consultancy, Research Funding. Sutherland:Celgene: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Janssen: Consultancy, Honoraria. Lalancette:Celgene: Honoraria; BMS: Honoraria. Iida:Celgene: Honoraria, Research Funding; Janssen Pharmaceuticals: Honoraria, Research Funding. Prince:Janssen: Honoraria; Celgene: Honoraria. Cochrane:BMS: Other: Received sponsorship to attend international meetings; Novartis: Other: Received sponsorship to attend international meetings; Celgene: Other: Received sponsorship to attend international meetings; Takeda: Other: Received sponsorship to attend international meetings. Khokhar:Janssen: Employment. Guckert:Johnson & Johnson: Equity Ownership; Janssen: Employment. Qin:Janssen: Employment. Oriol:Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal