Abstract
INTRODUCTION:
Diffuse large B-cell lymphoma (DLBCL) is the most frequently observed hematologic malignancy with 45,000 patients newly diagnosed globally each year. DLBCL patients can be cured if successfully treated with first line therapy, but patients who relapse face a poor prognosis.
OBJECTIVES:
The aim of this analysis was to assess the prognosis for patients who relapse after first line treatment and to assess the impact of patient age on outcomes through a systematic review of the literature.
METHODS:
A systematic review of observational and interventional studies that describe survival outcomes in DLBCL was conducted. The review included studies published internationally since 2000 that enrolled adult patients with DLBCL who had relapsed or progressed following first line treatment with rituximab. Phase I interventional studies, case reports, and studies assessing novel therapies in second line treatment were excluded. Data were collected on median overall survival (OS), and 1-yr and 5-yr survival rates. OS data were reported according to the time point from which they were calculated: from the point of relapse (studies including patients who may or may not have received second line treatment), from initiation of treatment post relapse (studies including patients who received active treatment post relapse), and from the time of autologous stem cell transplant (ASCT) post relapse. Results were further analysed by patient age.
RESULTS:
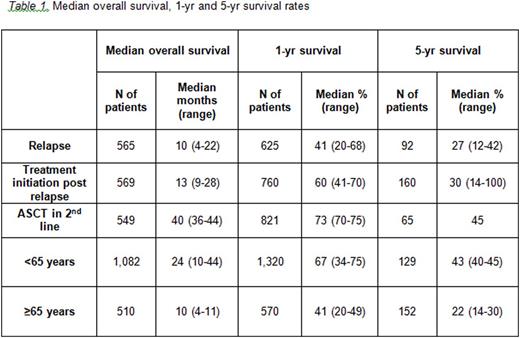
The search retrieved 2,275 citations, of which 28 studies met the inclusion criteria. In studies reporting OS from the time of relapse, median survival was 10 months (range 4 - 22) (Table 1). The proportions of patients alive at 1 and 5 years post relapse were on average (range) 41% (20% - 68%) and 27% (12% - 42%) respectively. The patients included in these studies were a mix of those who went on to receive a second line treatment and those who did not. In studies of patients who relapsed and received a subsequent line of treatment, median survival from the initiation of treatment post relapse was 13 months (9 - 28) and the proportions of patients alive at 1 and 5 years post second line treatment initiation were on average 60% (41% - 70%) and 30% (14% - 100%) respectively. In studies where survival was calculated from the time of ASCT post relapse, median survival was 40 months (36 - 44), while the proportions of patients alive at 1 and 5 years post ASCT were 73% (70% - 75%) and 45% respectively. The longer survival times for patients who received treatment post relapse likely reflects the better prognostic profile of patients deemed suitable for subsequent treatment versus those post relapse who may not have received second line treatment. Survival after ASCT is significantly influenced by the usual practice of selecting for transplant only patients showing chemo-responsiveness to salvage therapy. In studies that reported survival by patient age, median survival was more than twice as long for patients aged <65 years than those aged ≥65 years (24 and 10 months, respectively), and the proportions of patients alive at 5 years were 43% and 22%, respectively.
CONCLUSIONS:
The prognosis for patients with DLBCL who relapse is poor, with median survival of less than one year and less than half of patients who relapse still alive at one year post relapse. Despite the use of ASCT treatment, less than half of patients who met the eligibility criteria for and underwent ASCT were alive at 5 years post ASCT. Age was seen to be an important prognostic indicator in DLBCL patients who relapse, with poorer prognosis in patients aged ≥65 years than in those aged <65 years.
McMillan:Celgene: Honoraria; Roche: Honoraria. Martín:Sevier: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria. Haioun:Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sandoz: Honoraria, Membership on an entity's Board of Directors or advisory committees. Chiappella:Pfizer: Speakers Bureau; Teva: Speakers Bureau; Janssen-Cilag: Speakers Bureau; Celgene: Speakers Bureau; Roche: Speakers Bureau; Amgen: Speakers Bureau. Di Rocco:Celgene: Honoraria. Rueda:Roche: Speakers Bureau; Celgene: Honoraria, Speakers Bureau; Takeda: Speakers Bureau; Janssen: Speakers Bureau; Mundipharma: Speakers Bureau. Palaska:Celgene: Consultancy. Davies:Pfizer: Research Funding; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel to scientific conferences, Research Funding; Janssen: Honoraria; Mundipharma: Honoraria; GSK: Research Funding; Karyopharma: Honoraria, Research Funding; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, accommodation, expenses, Research Funding; Gilead: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Bayer: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal