Abstract
Background: Pediatric hospital-acquired venous thromboembolism (HA-VTE) has increased over the past ten years (Raffini et al. Pediatrics 2009; 124; 1001-1008), with an associated increase in both VTE-associated complications and treatment-related adverse events. Children with VTE have a four-fold increase in length of admission compared to children without VTE. There are many known clinical risk factors but the presence of central venous lines (CVLs) has been shown to be among the strongest risk factors in children (Mahajerin et al. Haematologica 2015 Aug; 100(8): 1045-50). Current tools, including clinical risk-prediction scoring systems as well as genetic risk factors, are not sufficient to predict VTE risk at this time. Identification of dependable, rapid and cost-effective biomarkers for the prediction of VTE in children with CVLs is essential in guiding anti-coagulation prophylaxis, thus reducing VTE occurrence among hospitalized children with CVLs.
Objectives: Determine the ability of thrombin generation (TG) and microparticle-based phospholipid-dependent procoagulant potential (MP-PPL) to predict VTE occurrence in hospitalized children with CVLs.
Methods: Plasma sample were collected from 34 hospitalized acutely ill children between the age of 1 month and 21 years, who are at risk for VTE due to multiple known clinical risk factors, including a newly placed non-tunneled CVL. Children that had undergone a recent cardiac catheterization or on dialysis, plasmapheresis or ECMO were excluded. Samples from 16 age matched healthy controls were also collected. Venous samples were collected prior to CVL placement. Platelet poor plasma (PPP) was obtained according to a strict protocol to minimize pre-analytical variable. All pertinent clinical and laboratory information was extracted, including CVL-related variables and details of VTE in affected subjects. TG was measured by means of the calibrated automated thrombogram (CAT). MP-PPL was measured using two methods: 1) Functional assay that detects the ability of circulating MPs to indirectly facilitate thrombin generation (Zymuphen MP-Activity kit, Hyphen BioMed, France), 2) Clot-based assay that measures phospholipid-dependent, factor Xa initiated clotting time in relation to normal controls (STA-Procoag-PPL, Diagnostica Stago).
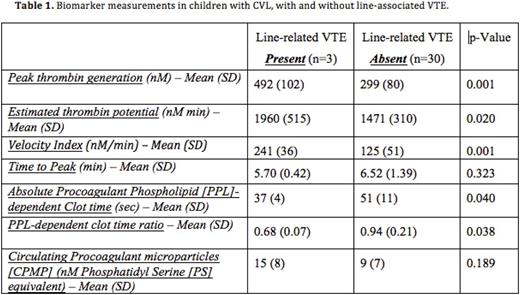
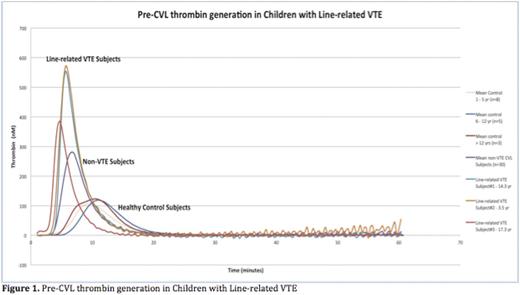
Results: The median age of our cohort was 10 yrs (IQR 12, range 0 - 19), with a slight male predominance (53%) and increased representation of adolescents (41%). CVLs were placed in an intensive care setting in 38% of subjects and 39% of subjects had an underlying condition associated with chronic inflammation. The median duration of CVL placement from admission was 2 days (IQR 5, range 1 - 19) and the median total duration of CVLs were 14 days (IQR 20, range 5 - 84). Line-associated VTE occurred in 9% (3/34) of children with CVL and one subject had a VTE unrelated to CVL. There was no observed significant difference between children with or without line-related VTE in terms of age, gender, lag of CVL placement from admission, duration of CVLs, caliber of initial vessel of insertion, number of lumens, size of CVL and internal length of the CVL. However, all subjects with line-associated VTE were admitted for acute infection to an intensive care setting, with the majority of VTE occurring within 48 hours from line placement (2/3). Correlation analysis of the two MP-PPL assays was significant (r = -0.659). Even though the MP-PPL functional assay did not show a significant difference between subjects with and without line-associated VTE (p-value 0.189), the MP-PPL clot-based assay showed measurable difference between the two groups, both as an absolute value and as a ratio (p-value: 0.04 and 0.038 respectively, Table 1). Peak thrombin generation (p-value 0.001), estimated thrombin potential (ETP) - (p-value 0.02) and velocity index (p-value 0.001) were significantly higher in subjects with line-associated VTE (see Figure 1). Subject recruitment and sample analyses, involving these assays and other potential biomarker candidates, are ongoing.
Conclusion: Given the rising incidence of pediatric VTE, the ability to use biomarkers to stratify patients according to their VTE risk will provide a vital tool to guide preventative efforts, and minimizing unnecessary expense and toxicities. TGA and MP-PPL have shown great potential for differentiating children with CVLs that may develop a line-related VTE as a HA-VTE.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal