Abstract
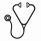
Azacitidine (AZA) is effective in high risk myelodysplastic syndromes (MDS), chronic myelomonocytic leukemia type 2 (CMML-2) and low blast count acute myeloid leukemia (AML) patients not suitable for more intensive treatment. Factors that may influence response to AZA are still under investigation. Bone marrow fibrosis is a potentially negative prognostic marker on overall survival (OS), but its clinical significance in this setting of patients remains to be clarified.
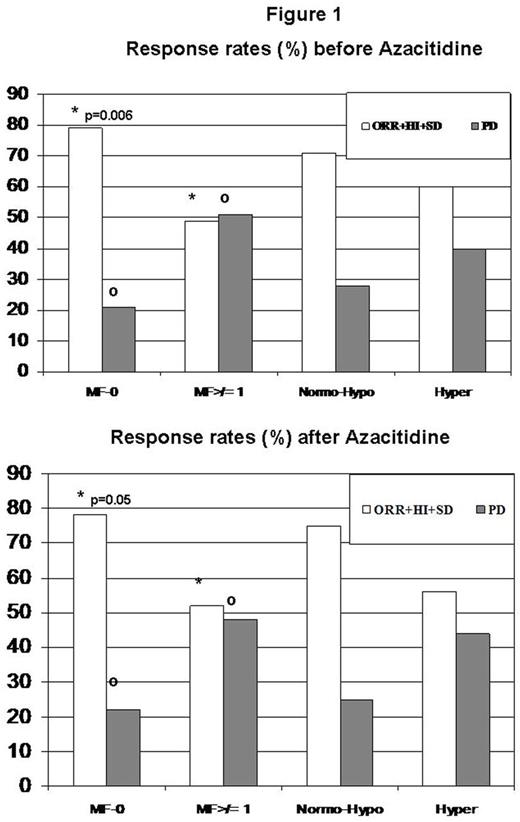
We evaluated clinical predictors of OS and overall response rate (ORR; complete/partial response CR/PR; stable/progressive disease SD/PD) to AZA in a real life cohort. We studied 94 consecutive patients, treated at two Institutions from June 2009 till February 2016 with AZA subcutaneously (5+2+2 schedule) every 28 days, outside clinical trials. We analyzed data from routine laboratory analysis, bone marrow histology, morphology and cytogenetics at diagnosis. OS was measured from the starting of AZA treatment. Table 1 shows the clinical characteristics pre- and post-AZA: most patients (68%) were AREB1 or AREB2, 13% RCUD/RCMD or MDS NOS according to WHO 2008 classification, 17% AML, 2% CMML. At the onset of AZA therapy the majority of MDS cases (68%) showed an intermediate-2 risk, according to the International Prognostic Scoring System (IPSS) and high/very high risk (78%) according to IPSS-revised. Secondary and de novo cases, as well as cytogenetics risk groups, were equally represented; 50% of patients were transfusion dependant and moderate to severe neutropenia or thrombocytopenia were present in roughly 50/70% of cases respectively. As expected, bone marrow biopsies pre-AZA showed hypercellularity in most patients (65%). Remarkably, 47,5% of cases showed bone marrow fibrosis of ≥1 grade before AZA initiation. These findings were mostly unchanged at post-AZA evaluation. On the whole, 93 patients receiving > 4 cycles of therapy were available for response evaluation according to International Working Group 2006 criteria. After a median of 6 cycles (4-44), ORR was 41.9% (CR 18.3%, PR 11.8%, SD with hematologic improvement HI 11.8%), SD was 21.5%, PD 10.7% and 25.8% failed to achieve a response. Thirteen percent of patients reached at least partial cytogenetic response and 50% a HI. ORR was not influenced by monocytosis, neutropenia or IPSS cytogenetic risk category. Interestingly, pre-AZA marrow blast percentage, cytogenetic risk, time from diagnosis to AZA and the interval from 1st to 6th cycle had no impact on response. As regards marrow characteristics, patients with MF-0 pre-AZA displayed significantly lower PD rate and higher ORR, SD and HI than those with any grade of fibrosis (21.4% vs 51.4% and 78.6% vs 48,6%, respectively p=0.006, Fig1). This observation was also confirmed at marrow evaluation after AZA (22% versus 48% for PD and 78% versus 52% for ORR/SD/HI, p=0.05, Fig1). Regarding cellularity pre- and post-AZA, higher ORR,SD and HI and lower PD were observed for patients with normo/hypo compared to those with hyper-cellularity (Fig1) although not significantly. Forty-one percent of cases presented a hematologic toxicity (33% neutropenia and 18% thrombocytopenia of any grade) occurring after a median of 2 (1-18) AZA cycles. Moreover 28.6% of patients had an infection during AZA treatment, not related to neutropenia degree. Of note, toxicities did not affect median time from the 1st to the 6th AZA cycle (170,115-240 days), nor ORR. Median OS from the beginning of therapy was 18.5 months (12.7-24.4, 95% CI). IPSS high category [HR 2.24 (1.19-4.20) p=0.01], poor cytogenetics [2.19 (1.27-3.78) p=0.005], and lower ORR [0.46 (0.26-0.80) p=0.006] significantly affected OS. Unexpectedly, a response obtained after less than 4 cycles negatively impact OS [HR 0.86 (0.80-0.92) p<0.0001]. Notably, cases with pre-AZA fibrosis ≥MF-1 showed lower OS [2.26 (1.28-3.99) p=0.005]. In conclusion we provide evidence of no relationship between neutropenia and infections and of no impact of toxicities on dose-density and ORR to AZA treatment. Moreover, high marrow fibrosis and hypercellularity may affect response to AZA therapy. Further studies are needed to disclose the clinical/biological significance of marrow fibrosis/cellularity in the era of hypomethylating agents.
Reda:Roche: Membership on an entity's Board of Directors or advisory committees; Gilead: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal