Abstract
Background: The fully human IgG4 anti-PD-1 monoclonal antibody, nivolumab, has shown to be highly efficacious in relapsed and refractory (RR) classical Hodgkin lymphoma (cHL). Initial results of a single-arm nivolumab monotherapy in heavily pretreated young adults with RR cHL, previously treated with autologous hematopoetic stem cell transplantation (autoSCT) and brentuximab vedotin are promising. However, demonstrating similar results in real-life experience in patients not on Pharma clinical studies are of major clinical importance.
Aim: The aim of the current study was to evaluate the efficacy and safety of nivolumab in previously heavily pretreated patients with RR cHL not on Pharma clinical studies.
Methods: The records of patients with RR cHL who were treated with nivolumab in seven medical centers were reviewed. The regimen consisted of nivolumab 3 mg/kg once every 2 weeks until disease progression or unacceptable toxicity.
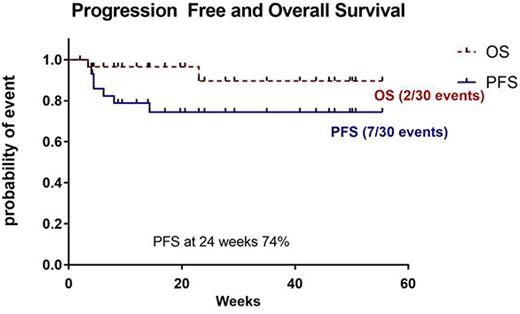
Results: Between May 2015 and April 2016, 30 patients with RR cHL received nivolumab monotherapy. The median age was 35 years (range: 21 to 88). Patients were extremely heavily pretreated with median of 6 prior therapies (range: 3 to 11); 73% of them received ≥ 5 lines of therapy. Specifically, 29/30 (97%) received ABVD, 7/30 (23%) escBEACOPP, 20/30 (67%) gemcitabine based regimens, 30/30 (100%) brentuximab vedotin, 8/30 (27%) bendamustine, 3/30 (10%) lenalidomide, 28/30 (93%) other combinations of chemotherapy and 22/30 (73%) radiotherapy. Twenty patients (67%) previously underwent autoHSCT and 4/30 (13%) allogeneic transplantation (alloSCT), two before and two after receiving nivolumab. Median number of nivolumab doses received was 8 (range 1 to 29). Twenty three patients (77%) are still on treatment. Seven patients (23%) discontinued therapy due to disease progression 3/30 (10%), adverse events 2/30 (7%) and referral to alloSCT 2/30 (7%). Overall response rate among 25 evaluable patients was 76%, complete response (CR) - 7/25 (28%), partial response - 12/25 (48%), stable disease - 3/25 (12%) and only 3/25 (12%) progressed, one of them with cHL type of Richter's transformation. Two patients underwent alloSCT in remission following nivolumab treatment. One patient was transplanted from a matched sibling donor and the second patient from a T replete haploidentical donor with post transplantation cyclophosphamide as anti-graft versus host disease prophylaxis. Both patients are in CR, ten and three months post transplantation, respectively. The median overall survival (OS) and progression free survival (PFS) for the cohort were not reached (Figure 1). At 24 weeks the PFS was 74%. Adverse events reported in 9/30 patients (30%) were usually mild. Severe nivolumab related adverse events in 3/30 (10%) included immune related pneumonitis, myelitis and uveitis. No treatment related death occurred.
Conclusions: The check point inhibitor nivolumab is a novel therapy for RR cHL patients, previously with unmet need following relapse or refractory to autoHSCT and brentuximab vedotin. Our real life experience confirms and highlights the efficacy and safety of nivolumab in very heavily pretreated young and elderly patients with cHL eligible as well as ineligible for autologous HSCT.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal