Abstract
INTRODUCTION
Follicular lymphoma (FL) is considered a chronic, immune responsive and still incurable tumour. Recently two papers have shown the efficacy of active immunotherapy by repeated administration of autologous dendritic cells (DC) in Follicular Lymphoma (FL). Notably, in both studies a third of patients achieved long lasting remission. A novel DC population (IFN-DC), differentiated from human monocytes in the presence of GM-CSF and IFN-alpha has been developed in the laboratory of the Istituto Superiore di Sanità (ISS). IFN-DC have been demonstrated highly efficient in internalizing tumour-cell antigens, recovering "in vitro" T-cell responses from FL patients, as well as in mediating NK cell activation and enhanced cytotoxic effector function toward autologous FL cells. We have recently started a phase I study (approved by EC in 2014, EudraCT: 2013-003158-25) of intranodal immunotherapy based on sequential injection of low-dose anti-CD20 antibody (Ab) and IFN-D, in patients with advanced FL. Here we report early results of the ongoing trial.
STUDY DESIGN
IFN-DC Trial (EudraCT: 2013-003158-25) is a phase I trial aiming at evaluating safety and tolerability as well as immune and clinical responses of a IFN-DC based therapy in combination with rituximab for the treatment of patients with advanced FL.
· The Primary endpoints are: I) Evaluation of safety and tolerability of treatment II) Evaluation of tumor-specific immune responses. by determining: i) Tumor-specific immune response in peripheral blood; ii) Intratumoral infiltration of immune cells; iii) DTH test.
· The secondary endpoint is clinical response. The study population consists of patients with relapsed/refractory FL, aged 18-75 years, not needing immediate retreatment basing on the GELF Criteria. The
· Treatment: the regimen foresees eight injection cycles, each consisting of the combined administration of rituximab and IFN-DC. The first four administered every two-weeks and the remaining four administered monthly. Rituximab (5-10mg) and low or high doses of IFN-DC (20±5 x106 or 40±5x106cells respectively), are administered by intranodal direct injection (IDI).
RESULTS
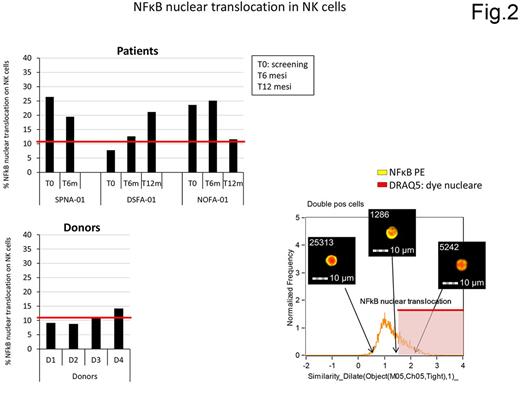
Five patients have been enrolled so far. The treatment proved to be safe, feasible and well tolerated. Three patients have completed treatment and two experienced complete remission with disappearance of both proximal and distal lesions, as confirmed by PET scan. Notably in both patients the distal lymph nodes, which had not been directly treated by intranodal injections, reduced to normal size earlier than those treated by IDI (abscopal effect). One of these two is still in durable complete clinical and molecular remission after 17 months while the latter patient, who had progressed after completing eight cycles of treatment, experienced a complete clinical remission after a second course of immunotherapy ( Fig.1). Two out of three patients performed the DHT test and, despite one of the two achieved remission, they both resulted non-responsive. The characterization of IFN-DC and lymphoma nodes in FL patients have shown high expression of PD-1 and PD-L1 in FL microenvironment and on IFN-DC respectively. Immunomonitoring is currently underway. However, quantification of NF-kB (p65) nuclear translocation by multispectral image flow cytometry, as a marker of NK cell activation, showed the persistence of higher percentages of activated NK cells in the two responding patients (Fig.2).
CONCLUSION
Currently no definitive data prove the benefit of IDI treatment, however the trial is ongoing and other patients are under screening. Notably both the responding patients showed the abscopal effect. The possible set-up of an effective and comparatively economic treatment, that exploits patients' immune system against this chronic and immune responsive tumour, is worth of exploring. Furthermore it can be envisaged that the combination of immune checkpoint inhibitor antibodies to DC-based therapies, might improve clinical activity of DC-based strategies.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal