Abstract
Background: Somatic gene mutations occur in the majority of MDS pts; specific mutations and high mutation frequency have prognostic relevance (Papaemmanuil et al. Blood. 2013;122:3616-27). Evaluation of somatic mutations may support the diagnosis of MDS and guide treatment (Tx) selection. The phase 3 randomized MDS-005 study compared LEN and placebo (PBO) Tx in red blood cell transfusion-dependent (RBC-TD) non-del(5q) lower-risk MDS pts ineligible for or refractory to ESAs. Deletions in chromosome 5q are associated with a high response rate to LEN in MDS pts; however, no mutations have been definitively associated with a predictable clinical response to LEN in non-del(5q) MDS.
Aim:To investigate the relationship between somatic gene mutations detected by targeted next-generation sequencing (NGS) and response and overall survival (OS) in lower-risk non-del(5q) MDS pts treated with LEN in the MDS-005 study.
Methods: Eligible pts were: RBC-TD (≥ 2 units packed RBCs/28 days 112 days immediately prior to randomization) with International Prognostic Scoring System defined Low-/Intermediate-1-risk non-del(5q) MDS; ineligible for ESA Tx (serum erythropoietin > 500 mU/mL); or unresponsive or refractory to ESAs (RBC-TD despite ESA Tx with adequate dose and duration). 239 pts were randomized 2:1 to oral LEN 10 mg once daily (5 mg for pts with creatinine clearance 40-60 mL/min) or PBO. DNA was isolated from bone marrow mononuclear cells or whole blood collected at screening from a subset of pts who gave informed consent for this exploratory biomarker analysis and had adequate tissue for analysis. Targeted NGS of 56 genes was performed at Munich Leukemia Laboratory; average sequencing coverage was 2,000-5,000-foldand the variant allele frequency detection cutoff was 3%. Target regions varied by gene, including all exons to hotspots. For association tests, mutant variants (heterozygous or homozygous) were scored as 1 (mutant) or 0 (wildtype) for gene-level analyses. A Fisher exact test was used to test association of mutation status with response. Median OS was calculated by the Kaplan-Meier method. Hazard ratios and 95% confidence intervals were determined by a non-stratified Cox proportional hazards model. A log-rank test was used to test treatment effect with OS for single gene mutation status.
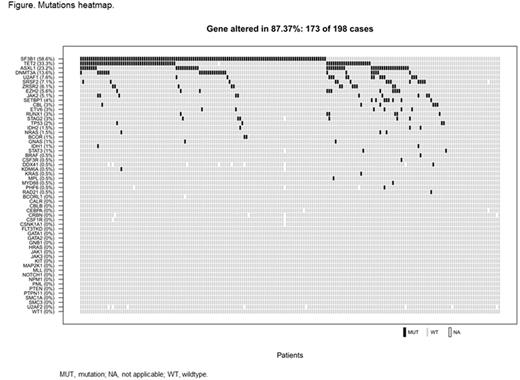
Results: The biomarker cohort included 198 of 239 pts (83%; LEN n = 130, PBO n = 68). At least 1 mutation was detected in 30/56 (54%) genes and 173/198 (87%) pts. The most frequently mutated genes were SF3B1 (59%), TET2 (33%), ASXL1 (23%), and DNMT3A (14%); the most frequent co-mutations were SF3B1/TET2 (23%), SF3B1/DNMT3A (10%), SF3B1/ASXL1 (10%), and TET2/ASXL1 (9%) (Figure). Of 116 pts with SF3B1 mutations, 115 (99%) had ≥ 5% ring sideroblasts. The 56-day RBC transfusion-independence (RBC-TI) response rate was significantly lower in LEN-treated ASXL1 mutant pts vs wildtype pts (10% vs 32%, respectively; P = 0.031). At 168 days, the RBC-TI response rate was still lower in LEN-treated ASXL1 mutant pts vs wildtype pts (7% vs 22%); however, the difference was not significant (P = 0.101). LEN-treated DNMT3A mutant pts had a higher 56-day RBC-TI response rate vs wildtype pts (44% vs 25%); however, this difference did not reach significance (P = 0.133) due to the small sample size. RBC-TI response rate with LEN was similar regardless of total number of mutations per pt. Higher numbers of mutations were significantly associated (P = 0.0005) with worse median OS. Mutation in any of the genes associated with a negative prognosis reported by Bejar et al. (N Engl J Med. 2011;346:2496-506) was also significantly associated (P = 0.0003) with worse median OS.However, OS was not significantly different in LEN- vs PBO-treated pts based on any single gene mutation status.
Conclusions: In this group of lower-risk RBC-TD non-del(5q) MDS pts, somatic mutations in genes recurrently mutated in myeloid cancers were detected in 87% of pts. SF3B1 mutations (alone or in combination) were most frequent and not associated with response to LEN. ASXL1 mutant pts had a significantly lower LEN response rate vs wildtype pts, whereas DNMT3A mutant pts had a trend for improved LEN response. Median OS was influenced by mutations, but not significantly modified by LEN. Determining predictive clinical markers for Tx response in non-del(5q) MDS pts remains challenging; nevertheless, there is a significant need to identify pt subsets who may be responsive to LEN Tx.
Santini:Novartis: Consultancy, Honoraria; Amgen: Other: advisory board; Onconova: Other: advisory board; Celgene: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria; Astex: Other: advisory board. Fenaux:Celgene, Janssen, Novartis, Astex, Teva: Research Funding; Celgene, Novartis, Teva: Honoraria. Giagounidis:Celgene Corporation: Consultancy. Platzbecker:Janssen-Cilag: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Celgene Corporation: Honoraria, Research Funding; Amgen: Honoraria, Research Funding; TEVA Pharmaceutical Industries: Honoraria, Research Funding. Zhong:Celgene Corporation: Employment, Equity Ownership. Wu:Celgene Corporation: Employment, Equity Ownership. Mavrommatis:Discitis DX: Membership on an entity's Board of Directors or advisory committees; Celgene Corporation: Employment, Equity Ownership. Beach:Celgene Corporation: Employment, Equity Ownership. Hoenekopp:Celgene Corporation: Employment, Equity Ownership. MacBeth:Celgene Corporation: Employment, Equity Ownership, Patents & Royalties, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal